Canine SCF (stem cell factor; also known as c‑kit ligand) is a type I transmembrane (TM) glycoprotein that plays an important role in a number of fetal and adult developmental processes (1‑4). It is synthesized as a 274 amino acid (aa) precursor that contains a 25 aa signal sequence, a 190 aa extracellular region, a 23 aa TM segment and a 36 aa cytoplasmic tail (5). Within the extracellular region there are four potential N‑linked glycosylation sites, two intrachain disulfide bonds, and four alpha ‑helices. Although the predicted molecular weight is 19 kDa, the native molecule is anywhere from 28‑40 kDa in size and reflects both N‑ and O‑linked glycosylation (1). Glycosylation is not necessary for bioactivity (6). The transmembrane form of SCF can be cleaved proteolytically, generating a 165 aa soluble form. Circulating SCF exists as both a monomer and nondisulfide‑linked homodimer, with monomer predominating (50% to 75%) (6). Both the soluble and TM forms have bioactivity. Their principal targets may be different, however (7). A second, alternate splice short form of SCF has been identified in other species (1). It is membrane bound but lacks the proteolytic cleavage site found in the long form. Thus, it cannot give rise to a soluble molecule. No such isoform has been reported for canine, but it could be assumed to exist. The ratio of long form to short form varies from tissue to tissue (1). Soluble canine SCF shares 88%, 93%, 86%, 83%, 76%, 76%, 86% and 88% aa sequence identity with porcine, feline, bovine, human, mouse, rat, goat and equine SCF, respectively. Cells known to express SCF include endothelial cells, fibroblasts and keratinocytes (1).


Key Product Details
Species Reactivity
Canine
Applications
Western Blot, ELISA Capture (Matched Antibody Pair), Neutralization, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant canine SCF/c‑kit Ligand
Lys26-Ala190
Accession # Q06220
Lys26-Ala190
Accession # Q06220
Specificity
Detects canine SCF/c‑kit Ligand in ELISAs and Western blots. In sandwich immunoassays, less than 20% cross-reactivity with recombinant feline SCF is observed, less than 8% cross-reactivity with recombinant human SCF is observed, and less than 0.5% cross-reactivity with recombinant mouse SCF is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Canine SCF/c‑kit Ligand Antibody
Cell Proliferation Induced by SCF/c‑kit Ligand and Neutralization by Canine SCF/c‑kit Ligand Antibody.
Recombinant Canine SCF/c-kit Ligand (Catalog # 2278-SC) stimulates pro-liferation in the TF-1 human erythroleukemic cell line in a dose-dependent manner (orange line). Proliferation elicited by Recombinant Canine SCF/c-kit Ligand (5 ng/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Canine SCF/ c-kit Ligand Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2278). The ND50 is typically 0.1-0.3 µg/mL.SCF/c‑kit Ligand in Canine PBMCs.
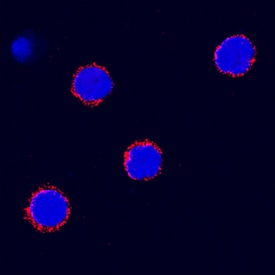
SCF/c-kit Ligand was detected in immersion fixed canine peripheral blood mononuclear cells (PBMCs) using Goat Anti-Canine SCF/c-kit Ligand Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2278) at 15 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to plasma membrane. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Applications for Canine SCF/c‑kit Ligand Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed canine peripheral blood mononuclear cells
Sample: Immersion fixed canine peripheral blood mononuclear cells
Western Blot
0.1 µg/mL
Sample: Recombinant Canine SCF/c-kit Ligand (Catalog # 2278-SC)
Sample: Recombinant Canine SCF/c-kit Ligand (Catalog # 2278-SC)
Neutralization
Measured by its ability to neutralize SCF/c‑kit Ligand-induced proliferation in the TF‑1 human erythroleukemic cell line. Kitamura, T. et al. (1989) J. Cell Physiol. 140:323. The Neutralization Dose (ND50) is typically 0.1-0.3 µg/mL in the presence of 5 ng/mL Recombinant Canine SCF/c‑kit Ligand.
Canine SCF/c-kit Ligand Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: SCF/c-kit Ligand
References
- Broudy, V.C. (1997) Blood 90:1345.
- Nakagawa, S. and T. Kitoh (2000) Curr. Opin. Hematol. 7:133.
- Yoshida, H. et al. (2001) J. Invest. Dermatol. Symp. Proc. 6:1.
- Kang, J. and S.D. Der (2004) Curr. Opin. Immunol. 16:180.
- Dunham, S.P. and D.E. Onions (1996) DNA Seq. 6:233.
- Hsu, Y-R. et al. (1997) J. Biol. Chem. 272:6406.
- Kapur, R. et al. (1998) Blood 91:879.
Long Name
Stem Cell Factor
Alternate Names
c-kit Ligand, DCUA, DFNA69, FPH2, FPHH, KITLG, KL-1, MGF, SHEP7, SLF
Gene Symbol
KITLG
UniProt
Additional SCF/c-kit Ligand Products
Product Documents for Canine SCF/c‑kit Ligand Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Canine SCF/c‑kit Ligand Antibody
For research use only
Related Research Areas
Customer Reviews for Canine SCF/c‑kit Ligand Antibody
There are currently no reviews for this product. Be the first to review Canine SCF/c‑kit Ligand Antibody and earn rewards!
Have you used Canine SCF/c‑kit Ligand Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars