534 results for "TSLP" in Products
TSLP Products
TSLP (Thymic Stromal Lymphopoietin) is a secreted cytokine that signals through a complex of TSLP R and IL-7 R alpha/CD127. It acts on thymic dendritic cells which convert potentially autoreactive T cells into regulatory T cells. TSLP promotes fetal liver pro-B cell proliferation as well as large pre-B cell differentiation and B cell lymphopoiesis in adult bone marrow. It is produced by allergen challenged keratinocytes and mucosal epithelial cells. It enhances Th2 biased allergic inflammation by limiting Th1/Th17 inflammation, promoting Th2 differentiation, and enhancing the function of basophils, eosinophils, and mast cells.
| Reactivity: | Human |
Build your own Luminex Assay with our Luminex Assay Customization Tool
| Reactivity: | Human |
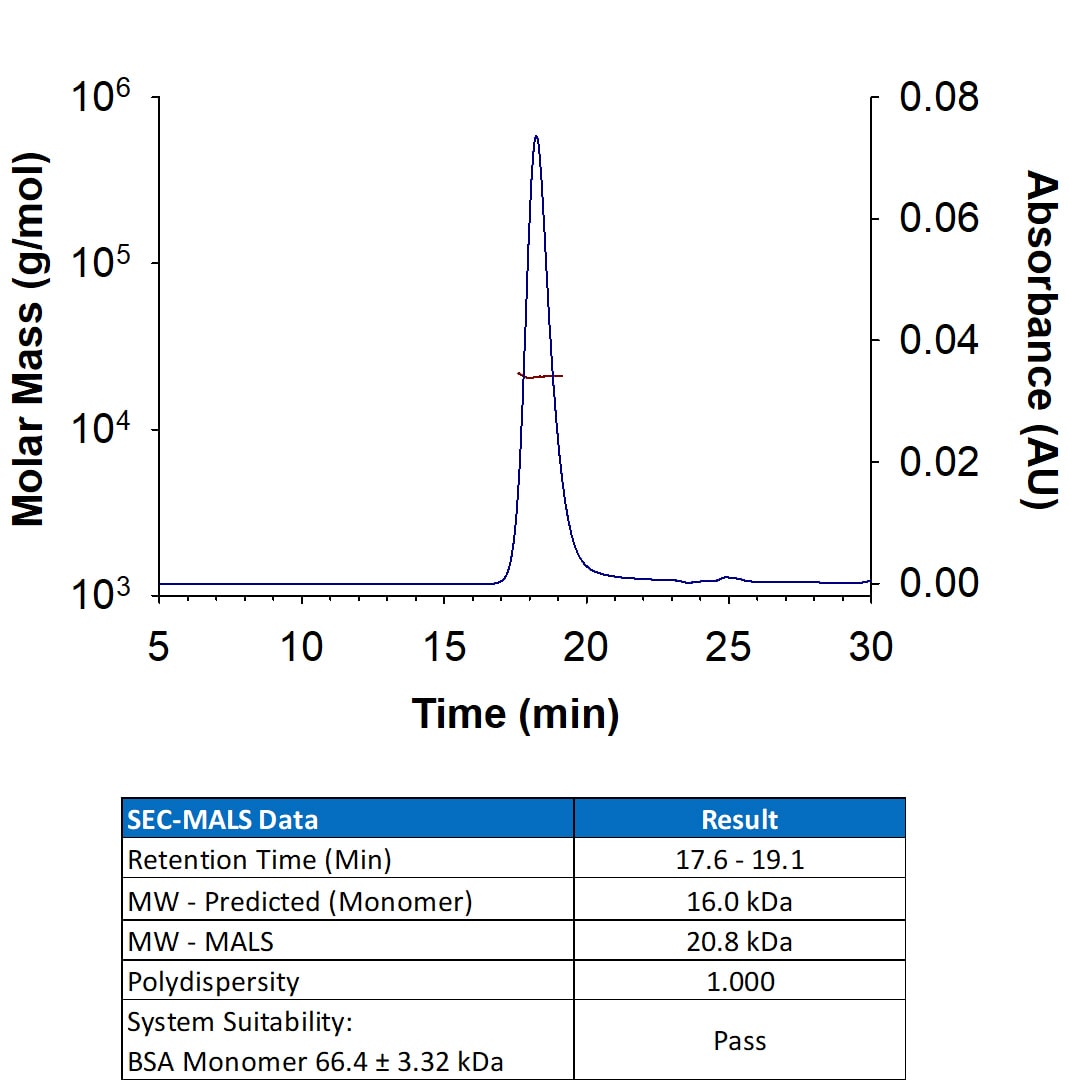
| Source: | E. coli |
| Accession #: | Q969D9.1 |
| Applications: | Bioactivity |
| Reactivity: | Mouse |
| Source: | NS0 |
| Accession #: | NP_067342 |
| Applications: | Bioactivity |
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, B/N |
| Reactivity: | Mouse |
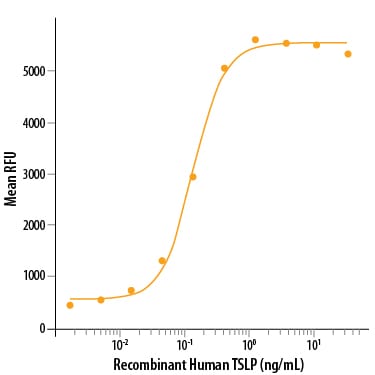
| Sensitivity: | 6.3 pg/mL |
| Assay Range: | 7.8-500 pg/mL (Cell Culture Supernates, Serum, EDTA Plasma, Heparin Plasma) |
| Applications: | ELISA |
| Reactivity: | Mouse |
| Details: | Rat IgG2A Monoclonal Clone #152614 |
| Applications: | WB, B/N |
| Reactivity: | Human |
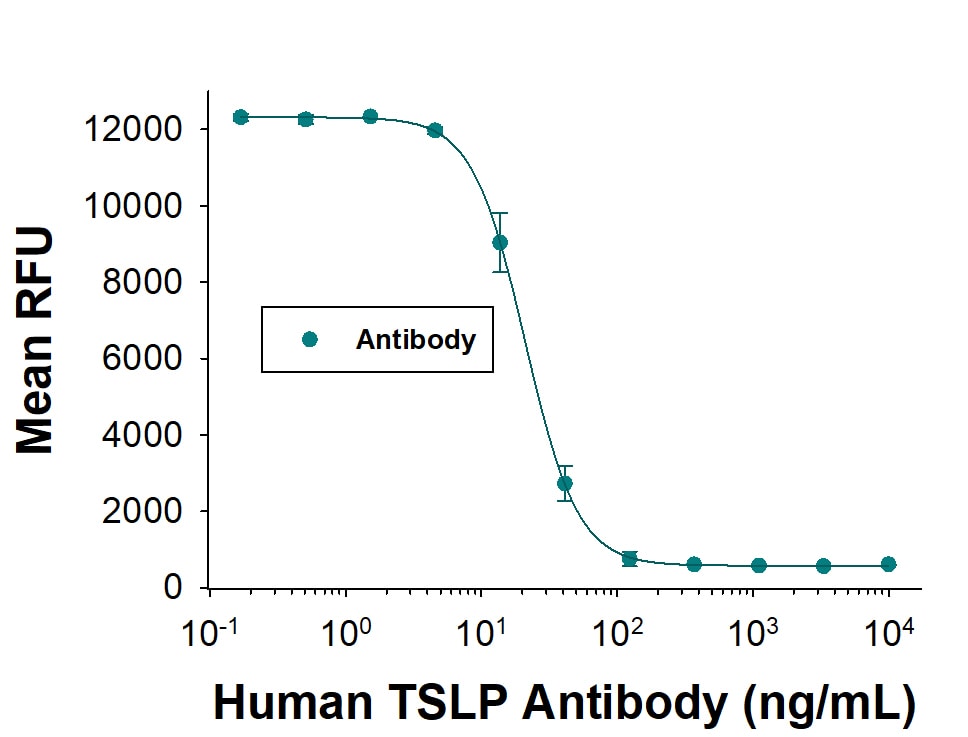
| Sensitivity: | 9.87 pg/mL |
| Assay Range: | 31.2-2000 pg/mL (Cell Culture Supernates, Serum, EDTA Plasma, Heparin Plasma) |
| Applications: | ELISA |
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB, ELISA(Cap), B/N |
| Reactivity: | Mouse |
| Assay Range: | 15.6-1000 pg/mL |
| Applications: | ELISA |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ELISA, ICC/IF |
| Reactivity: | Human |
| Assay Range: | 31.2-2000 pg/mL |
| Applications: | ELISA |
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #258136 |
| Applications: | WB |
| Reactivity: | Human, Rabbit |
| Details: | Mouse IgG2a Kappa Monoclonal Clone #55N1D10 |
| Applications: | IHC, WB |
| Reactivity: | Human, Rabbit |
| Details: | Mouse IgG2a Kappa Monoclonal Clone #55N1D10 |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ELISA |
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, ELISA(Det) |
| Reactivity: | Human |
| Details: | Mouse IgG2a Kappa Monoclonal Clone #55N2D4 |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Mouse IgG2a Kappa Monoclonal Clone #55N2D4 |
| Applications: | IHC, WB |
| Reactivity: | Mouse |
| Details: | Rat IgG2A Monoclonal Clone #152640 |
| Applications: | WB, ELISA(Cap) |
| Reactivity: | Human |
| Source: | HEK293 |
| Accession #: | Q969D9.1 |
| Applications: | Bioactivity |
| Reactivity: | Mouse, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ELISA, ICC/IF |
| Reactivity: | Human |
| Details: | Mouse IgG1 kappa Monoclonal Clone #55N1E3 |
| Applications: | WB |
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #933822 |
| Applications: | ELISA, B/N |
| Reactivity: | Human |
| Details: | Mouse IgG1 kappa Monoclonal Clone #55N1E3 |
| Applications: | WB |
| Reactivity: | Human |
| Source: | Synthetic |
| Applications: | AC |











![Western Blot: TSLP AntibodyBSA Free [NB110-55234] Western Blot: TSLP AntibodyBSA Free [NB110-55234]](https://resources.rndsystems.com/images/products/TSLP-Antibody-Western-Blot-NB110-55234-img0013.jpg)


![Western Blot: TSLP Antibody (55N1D10)BSA Free [NBP2-27326] Western Blot: TSLP Antibody (55N1D10)BSA Free [NBP2-27326]](https://resources.rndsystems.com/images/products/TSLP-Antibody-55N1D10-Western-Blot-NBP2-27326-img0003.jpg)
![Western Blot: TSLP Antibody (55N1D10)Azide and BSA Free [NBP2-80999] Western Blot: TSLP Antibody (55N1D10)Azide and BSA Free [NBP2-80999]](https://resources.rndsystems.com/images/products/TSLP-Antibody-55N1D10-Azide-and-BSA-Free-Western-Blot-NBP2-80999-img0001.jpg)
![Western Blot: TSLP AntibodyBSA Free [NB110-55233] Western Blot: TSLP AntibodyBSA Free [NB110-55233]](https://resources.rndsystems.com/images/products/TSLP-Antibody-Western-Blot-NB110-55233-img0004.jpg)
![Western Blot: TSLP Antibody (55N2D4)BSA Free [NBP2-27325] Western Blot: TSLP Antibody (55N2D4)BSA Free [NBP2-27325]](https://resources.rndsystems.com/images/products/TSLP-Antibody-55N2D4-Western-Blot-NBP2-27325-img0003.jpg)
![Western Blot: TSLP Antibody (55N2D4)Azide and BSA Free [NBP2-80998] Western Blot: TSLP Antibody (55N2D4)Azide and BSA Free [NBP2-80998]](https://resources.rndsystems.com/images/products/TSLP-Antibody-55N2D4-Azide-and-BSA-Free-Western-Blot-NBP2-80998-img0001.jpg)
![Western Blot: TSLP AntibodyBSA Free [NBP1-76754] Western Blot: TSLP AntibodyBSA Free [NBP1-76754]](https://resources.rndsystems.com/images/products/TSLP-Antibody-Western-Blot-NBP1-76754-img0002.jpg)
![Western Blot: TSLP Antibody (55N1E3) [NBP2-27323] Western Blot: TSLP Antibody (55N1E3) [NBP2-27323]](https://resources.rndsystems.com/images/products/TSLP-Antibody-55N1E3-Western-Blot-NBP2-27323-img0001.jpg)
![Western Blot: TSLP Antibody (55N1E3)Azide and BSA Free [NBP2-80997] Western Blot: TSLP Antibody (55N1E3)Azide and BSA Free [NBP2-80997]](https://resources.rndsystems.com/images/products/TSLP-Antibody-55N1E3-Azide-and-BSA-Free-Western-Blot-NBP2-80997-img0001.jpg)