Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
257 results for "MAP2" in Products
MAP2 Products
Microtubule-associated Protein 2 (MAP2) is an 1827 amino acid (aa) cytoskeletal associated protein that contains C-terminal microtubule-binding repeats and a conserved N-terminal domain, which has been shown to interact with the RII regulatory subunit of Protein Kinase A. Human MAP2 shares 84% and 79% aa identity with the mouse and rat orthologs, respectively. Multiple splice forms exist, resulting in four distinct isoforms with differing N-terminal domains. Mammalian MAP2 is mainly ...
| Reactivity: | Human, Mouse, Rat, Feline |
| Details: | Chicken IgY Polyclonal |
| Applications: | IHC, WB, ICC/IF, Mycoplasma |
Loading...
| Reactivity: | Human, Mouse, Rat, Bovine |
| Details: | Mouse IgG1 Monoclonal Clone #4H5 |
| Applications: | IHC, WB, ICC/IF |
Loading...
| Reactivity: | Human, Mouse, Rat, Monkey |
| Details: | Mouse IgG2B Monoclonal Clone #5H11 |
| Applications: | IHC, WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG3 Monoclonal Clone #885232 |
| Applications: | mIF, IHC, ICC/IF, COMET |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #CL5420 |
| Applications: | IHC, WB, ICC/IF, Single-Cell Western |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #2C4 |
| Applications: | mIF, IHC, WB, ICC/IF, COMET |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB, ICC/IF |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #CL5420 |
| Applications: | IHC, WB, ICC/IF |
Loading...
| Reactivity: | Human, Mouse, Rat, Avian, Bovine, +1 More |
| Details: | Mouse IgG1 Monoclonal Clone #HM-2 |
| Applications: | IHC, WB, ICC/IF, MA |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Goat IgG1 Polyclonal |
| Applications: | IHC, WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #CL5420 |
| Applications: | IHC, WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #HL1655 |
| Applications: | IHC, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #5B7 |
| Applications: | WB, ELISA, Flow, CyTOF-ready |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #HL1656 |
| Applications: | IHC, ICC/IF |
Loading...
| Reactivity: | Mouse, Rat |
| Details: | Sheep IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF |
Loading...
| Reactivity: | Human, Mouse, Porcine, Bovine, Feline |
| Details: | Mouse IgG1 Monoclonal Clone #MT-01 |
| Applications: | WB, ICC/IF, IP |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Source: | Wheat germ |
| Applications: | WB, ELISA, AP, MA, PAGE |
Loading...
| Reactivity: | Human |
| Sensitivity: | 0.188 ng/mL |
| Assay Range: | 0.313-20 ng/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Sensitivity: | 37.5 pg/mL |
| Assay Range: | 62.5-4000 pg/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG3 Monoclonal Clone #885232 |
| Applications: | IHC, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG3 Monoclonal Clone #885232 |
| Applications: | IHC, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG3 Monoclonal Clone #885232 |
| Applications: | IHC, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG3 Monoclonal Clone #885232 |
| Applications: | IHC, ICC/IF |
Loading...


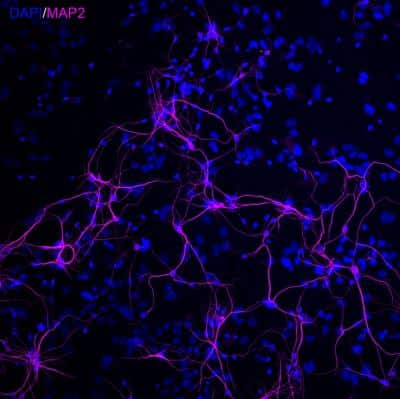
![Immunocytochemistry/ Immunofluorescence: MAP2 Antibody (4H5) [NBP2-25156] Immunocytochemistry/ Immunofluorescence: MAP2 Antibody (4H5) [NBP2-25156]](https://resources.rndsystems.com/images/products/MAP2-Antibody-4H5-Immunocytochemistry-Immunofluorescence-NBP2-25156-img0003.jpg)
![Immunohistochemistry Free-Floating: MAP2 Antibody (5H11) [NBP1-92711] Immunohistochemistry Free-Floating: MAP2 Antibody (5H11) [NBP1-92711]](https://resources.rndsystems.com/images/products/MAP2-Antibody-5H11-Immunocytochemistry-Immunofluorescence-NBP1-92711-img0001.jpg)

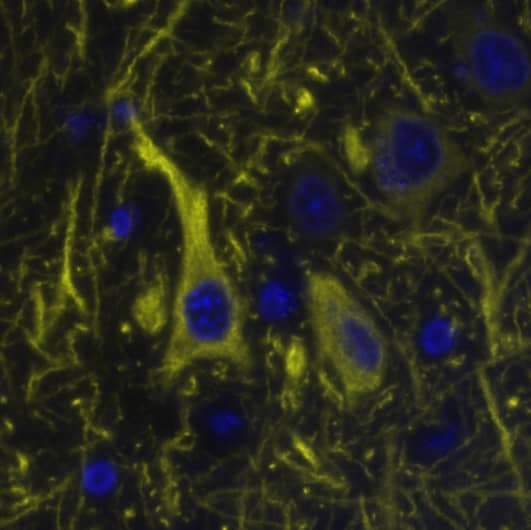
![Western Blot: MAP2 Antibody (CL5420) [NBP2-61417] Western Blot: MAP2 Antibody (CL5420) [NBP2-61417]](https://resources.rndsystems.com/images/products/MAP2-Antibody-CL5420-Western-Blot-NBP2-61417-img0007.jpg)
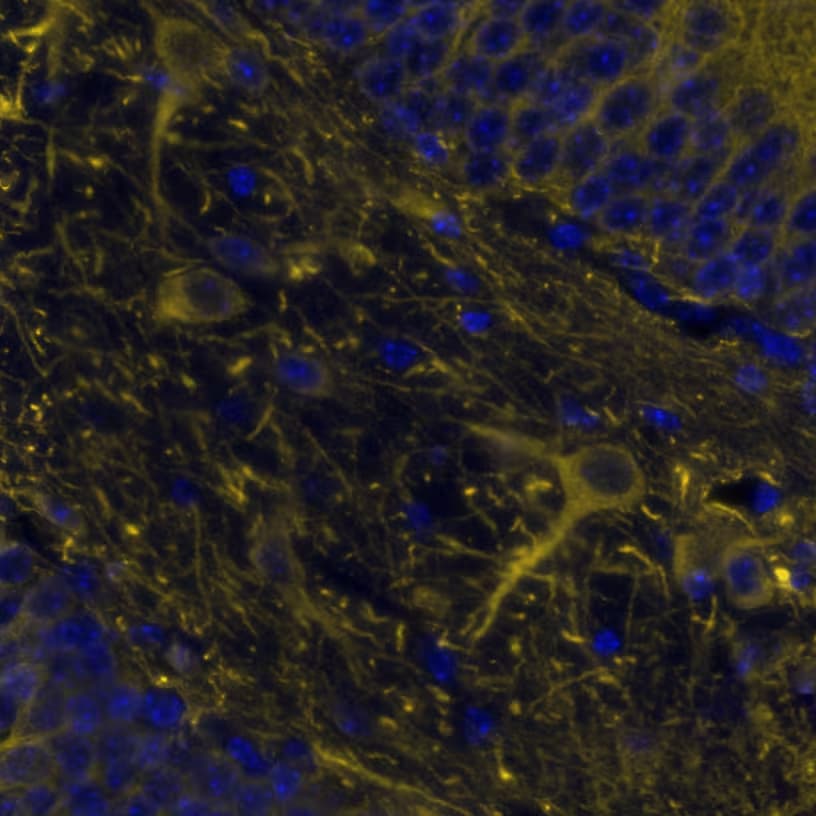
![Immunohistochemistry: MAP2 Antibody [NBP1-81264] Immunohistochemistry: MAP2 Antibody [NBP1-81264]](https://resources.rndsystems.com/images/products/MAP2-Antibody-Immunohistochemistry-NBP1-81264-img0006.jpg)
![Western Blot: MAP2 Antibody (CL5420) [NBP3-52055] MAP2 Antibody (CL5420) Western Blot: MAP2 Antibody (CL5420) [NBP3-52055]](https://resources.rndsystems.com/images/products/nbp3-52055_mouse-map2-mab-cl5420-western-blot-273202615211923.jpg)
![Western Blot: MAP2 Antibody (HM-2)BSA Free [NB120-11267] Western Blot: MAP2 Antibody (HM-2)BSA Free [NB120-11267]](https://resources.rndsystems.com/images/products/MAP2-Antibody-HM-2-Western-Blot-NB120-11267-img0003.jpg)
![Western Blot: MAP2 Antibody [NBP3-05552] Western Blot: MAP2 Antibody [NBP3-05552]](https://resources.rndsystems.com/images/products/MAP2-Antibody-Western-Blot-NBP3-05552-img0002.jpg)
![Immunohistochemistry-Paraffin: MAP2 Antibody [NBP3-43783] MAP2 Antibody (CL5420) - Azide and BSA Free Immunohistochemistry-Paraffin: MAP2 Antibody [NBP3-43783]](https://resources.rndsystems.com/images/products/nbp3-43783_mouse-map2-mab-cl5420-azide-and-bsa-free-282202512121921.jpg)
![Immunocytochemistry/ Immunofluorescence: MAP2 Antibody [NBP1-81263] Immunocytochemistry/ Immunofluorescence: MAP2 Antibody [NBP1-81263]](https://resources.rndsystems.com/images/products/MAP2-Antibody-Immunocytochemistry-Immunofluorescence-NBP1-81263-img0001.jpg)
![Immunocytochemistry/Immunofluorescence: MAP2 Antibody (HL1655) - Azide and BSA Free [NBP3-25554] - MAP2 Antibody (HL1655) - Azide and BSA Free](https://resources.rndsystems.com/images/products/nbp3-25554_rabbit-map2-mab-hl1655-azide-and-bsa-free-2120249403852.jpg)
![Western Blot: MAP2 Antibody (5B7)BSA Free [NBP2-37734] Western Blot: MAP2 Antibody (5B7)BSA Free [NBP2-37734]](https://resources.rndsystems.com/images/products/MAP2-Antibody-5B7-Western-Blot-NBP2-37734-img0002.jpg)
![Immunohistochemistry-Paraffin: MAP2 Antibody (HL1656) - Azide and BSA Free [NBP3-25555] - MAP2 Antibody (HL1656) - Azide and BSA Free](https://resources.rndsystems.com/images/products/nbp3-25555_rabbit-map2-mab-hl1656-azide-and-bsa-free-212024939165.jpg)
![Immunocytochemistry/ Immunofluorescence: MAP2 Antibody [NB100-98717] Immunocytochemistry/ Immunofluorescence: MAP2 Antibody [NB100-98717]](https://resources.rndsystems.com/images/products/MAP2-Antibody-Immunocytochemistry-Immunofluorescence-NB100-98717-img0003.jpg)
![Western Blot: MAP2 Antibody (MT-01)BSA Free [NB500-415] Western Blot: MAP2 Antibody (MT-01)BSA Free [NB500-415]](https://resources.rndsystems.com/images/products/MAP2-Antibody-MT-01-Western-Blot-NB500-415-img0001.jpg)
![Western Blot: MAP2 Antibody [NBP3-21231] - Western Blot: MAP2 Antibody [NBP3-21231] -](https://resources.rndsystems.com/images/products/nbp3-21231_rabbit-map2-pab-452023155034.jpg)
![SDS-PAGE: Recombinant Human MAP2 GST (N-Term) Protein [H00004133-P01] SDS-PAGE: Recombinant Human MAP2 GST (N-Term) Protein [H00004133-P01]](https://resources.rndsystems.com/images/products/qc_test-H00004133-P01-1.jpg)
![ELISA: Human MAP2 ELISA Kit (Colorimetric) [NBP2-80250] - Human MAP2 ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp2-80250_human-map2-elisa-kit-colorimetric-132202416201952.jpg)
![ELISA: Human MAP2 ELISA Kit (Chemiluminescence) [NBP2-80251] - Human MAP2 ELISA Kit (Chemiluminescence)](https://resources.rndsystems.com/images/products/nbp2-80251_human-map2-elisa-kit-chemiluminescence-13220241692279.jpg)