ABCG1 Antibody - BSA Free
Novus Biologicals | Catalog # NB400-132

![Western Blot: ABCG1 AntibodyBSA Free [NB400-132] Western Blot: ABCG1 AntibodyBSA Free [NB400-132]](https://resources.rndsystems.com/images/products/ABCG1-Antibody---BSA-Free-Western-Blot-NB400-132-img0016.jpg)
Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Clonality
Host
Isotype
Theoretical MW
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for ABCG1 Antibody - BSA Free
Immunohistochemistry-Paraffin: ABCG1 Antibody - BSA Free [NB400-132]
Immunohistochemistry-Paraffin: ABCG1 Antibody - BSA Free [NB400-132] - Staining of human lung, alveolar macrophages.Flow (Intracellular): ABCG1 Antibody - BSA Free [NB400-132]
Flow (Intracellular): ABCG1 Antibody - BSA Free [NB400-132] - RAW 246.7 cells were either untreated (A) or serum starved for 24 hours, and then treated with 1uM TO9 for 24 hours (B). An intracellular stain was performed with NB400-132 (blue) and a matched isotype control NB810-56910 (orange). Cells were fixed with 4% paraformaldehyde, following fixation, cells were permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 5 ug/mL for 30 minutes at room temperature, followed by Goat Anti-Rabbit Dylight 550-conjugated antibody.Western Blot: ABCG1 AntibodyBSA Free [NB400-132]
Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - Detection of ABCG1 in J774 mouse macrophages. Image provided by Dr. Angeliki Chroni of the National Centre for Scientific Research Demokritos.Western Blot: ABCG1 AntibodyBSA Free [NB400-132]
Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - Raw 264.7 (A), and Raw 264.7 + 10uM T09 18 hr (B) cell lysate using ABCG1 antibody at 2 ug/mL.Western Blot: ABCG1 AntibodyBSA Free [NB400-132]
Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - Detection of ABCG1 in mouse peritoneal macrophages. Lane 1: untreated mouse peritoneal macrophages, Lane 2: 22-(R)-hydrocholesterol treated macrophages, Lane 3: T0901713 treated macrophages, Lane 4: T091713 treated macrophages.Western Blot: ABCG1 AntibodyBSA Free [NB400-132]
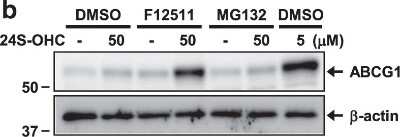
ABCG1-Antibody---BSA-Free-Western-Blot-NB400-132-img0014.jpgWestern Blot: ABCG1 Antibody - BSA Free [NB400-132] -
miR-33 deficiency improved cholesterol efflux in macrophages. A, Quantitative real-time PCR analysis of Abca1 & Abcg1 in macrophages from miR-33+/+Apoe−/− & miR-33−/−Apoe−/− mice. Values from miR-33+/+Apoe−/− were set at 100%. Values are mean±SE (n=7 each); ***P<0.001. B, Western blotting analysis of ABCA1 & ABCG1 in thioglycollate-elicited peritoneal macrophages from miR-33+/+Apoe−/− & miR-33−/−Apoe−/− mice. GAPDH was used as a loading control. C, Cholesterol efflux from thioglycollate-elicited peritoneal macrophages in the presence or absence of apoA-I (5 or 10 μg/mL). Values are mean±SE (n=6 each); **P<0.01. D, Cholesterol efflux from thioglycollate-elicited peritoneal macrophages in the presence or absence of HDL-C (50 or 100 μg/mL). Values are mean±SE (n=6 each); **P<0.01. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23316322), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: ABCG1 Antibody - BSA Free [NB400-132] -
ABCA1, ABCG1 & SR-BI protein expressions. a & b Hepatic protein expressions of ABCA1, ABCG1 & SR-BI were significantly decreased in COMT−/− mice at GD 18.5, compared to C57BL/6 J mice. Decreased hepatic ABCA1 expression was also observed at 10 days postpartum. ATI-5261 increased ABCA1 & ABCG1 expression in the liver at 10 days postpartum. c Placental protein expressions of ABCA1 & ABCG1 was reduced in COMT−/− mice, compared to C57BL/6 J mice. ATI-5261 treatment significantly increased ABCA1 levels in the placenta of COMT−/− mice. d Representative immunoblots of the corresponding proteins in the placenta with mouse RAW264.7 cell lysate included as positive control. Similar results were obtained when the experiment was repeated with lysates prepared from three batches of tissues. Data are presented as mean ± SEM. Groups (n = 8 in all groups) were compared using one-way ANOVA with post-hoc analysis (Tukey’s procedure). *, p < 0.05 Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30237900), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: ABCG1 Antibody - BSA Free [NB400-132] -
6‐Dihydroparadol increases A) ABCA1 & B) ABCG1, but not C) SR‐BI protein levels in cholesterol‐loaded THP‐1‐derived macrophages. THP‐1 cells were differentiated as described in Figure 2, & then loaded with unlabeled cholesterol for 24 h. Cells were treated with increasing concentrations of 6‐dihydroparadol (1–30 μm) for another 24 h. The protein levels of ABCA1, ABCG1, & SR‐B1 were detected by Western blot analysis. The control was treated with solvent vehicle (0.1% DMSO). As a positive control, TO901317 (5 μm, 24 h) was used. The bar graphs represent mean ± SD from three independent experiments. *p < 0.05, **p < 0.01, & ***p < 0.001 versus control (determined by Student's t‐test or ANOVA with Bonferroni's post hoc test). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29802792), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: ABCG1 Antibody - BSA Free [NB400-132] -
(A) Chemical structure, (B) cytotoxicity of alpha -asarone, (C) enhancement of cholesterol efflux by alpha -asarone, (D & E) upregulation of ABCA1 & ABCG1 by alpha -asarone & beta -asarone, & (F) elevation of retinoid X receptor (RXR) alpha transcription. J774A.1 murine macrophages were exposed to 50 μg/ml oxidized low-density lipoprotein (LDL) & treated with 1–10 μM purple Perilla frutescens extracts (PPE)-alpha -asarone & 5–10 μM beta -asarone. (B) MTT assay was performed for the measurement of alpha -asarone toxicity. Graph data represent 1 of 4 independent experiments with multiple estimations. Values are expressed as the percentage cell survival relative to the untreated control cells (cell viability, 100%). (C) Cholesterol efflux was expressed as the percentage fluorescence in the medium relative to total fluorescence. (D & E) For the measurement of ABCA1 & ABCG1 expression, total cell lysates were subjected to western blot analysis with a primary antibody against ABCG1 or ABCG1. beta -actin was used as an internal control. Bar graphs (means ± SEM, n=3) represent quantitative densitometric results of the upper bands. Bar graphs denoted without a common letter indicate significant difference, P<0.05. (F) RXR alpha mRNA expression was measured by RT-PCR. GAPDH was used as a housekeeping gene for the co-amplification with RXR alpha. Image collected & cropped by CiteAb from the following publication (https://www.spandidos-publications.com/10.3892/ijmm.2015.2101), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: ABCG1 Antibody - BSA Free [NB400-132] -
6‐Dihydroparadol increases A) ABCA1 & B) ABCG1 protein stability. THP‐1 cells were differentiated as described in Figure 2. Then cells were loaded with unlabeled cholesterol & treated with 6‐dihydroparadol (10 μm), or solvent vehicle (0.1% DMSO, control) for 24 h. Cells were lysed at different time points (0, 1, 2, 3, 4, & 6 h) after treatment with the protein synthesis inhibitor cycloheximide (CHX, 100 μm). The protein levels of both ABCA1 & ABCG1 were detected by Western blot analysis. The data points represent mean ± SD from three independent experiments. *p < 0.05 & **p < 0.01 versus control at the same time point (determined by Student's t‐test). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29802792), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: ABCG1 Antibody - BSA Free [NB400-132] -
(A) Upregulation of ABCA1 & ABCG1 & (B) enhancement of cholesterol efflux by purple Perilla frutescens extracts (PPE) in 50 μg/ml Cu2+-oxidized low-density lipoproteins (LDL)-exposed J774A.1 murine macrophages. (A) For the measurement of ABCA1 & ABCG1 expression, total cell lysates were subjected to western blot analysis with a primary antibody against ABCA1 or ABCG1. beta -actin was used as an internal control. Bar graphs (means ± SEM, n=3) represent quantitative densitometric results of the upper bands. (B) Cholesterol efflux was expressed as the percentage of fluorescence in the medium relative to the total fluorescence. Bar graphs denoted without a common letter indicate significant difference, P<0.05. Image collected & cropped by CiteAb from the following publication (https://www.spandidos-publications.com/10.3892/ijmm.2015.2101), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: ABCG1 Antibody - BSA Free [NB400-132] -
Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - 6‐Dihydroparadol increases A) ABCA1 & B) ABCG1 protein stability. THP‐1 cells were differentiated as described in Figure 2. Then cells were loaded with unlabeled cholesterol & treated with 6‐dihydroparadol (10 μm), or solvent vehicle (0.1% DMSO, control) for 24 h. Cells were lysed at different time points (0, 1, 2, 3, 4, & 6 h) after treatment with the protein synthesis inhibitor cycloheximide (CHX, 100 μm). The protein levels of both ABCA1 & ABCG1 were detected by Western blot analysis. The data points represent mean ± SD from three independent experiments. *p < 0.05 & **p < 0.01 versus control at the same time point (determined by Student's t‐test). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29802792), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: ABCG1 Antibody - BSA Free [NB400-132] -
Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - Evodiamine enhances apo A1-mediated ChE from THP-1 macrophages & increases ABCA1 protein level. (a) Differentiated THP-1 cells were loaded with [3H]-cholesterol together with the indicated treatments for 24 h. On the next day, the cells were washed twice with PBS & incubated with the same compounds [solvent vehicle control (Veh; ≤0.1% DMSO), evodiamine (1–20 μM), & the PPAR gamma agonist pioglitazone (10 μM) as positive control] with or without 10 µg/mL apo A1. Extracellular as well as intracellular radioactivities were quantified with scintillation counter. Differentiated THP-1-derived macrophages were treated with solvent vehicle control (Veh; ≤0.1% DMSO), evodiamine (10 μM), & the PPAR gamma agonist pioglitazone (10 μM) as positive control. After 24 h incubation, the cells were lysed & 20 μg protein was resolved via SDS-PAGE. Immunodetection was performed with antibodies against the indicated proteins, ABCA1 (b), ABCG1 (c), & SR-B1 (d), & visualized by chemiluminescence detection. All experiments were performed at least three times & data are presented as means ± S.D. vs. solvent vehicle control, *p < 0.05, **p < 0.01, ***p < 0.001, n.s. no significance (ANOVA/Bonferroni). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30038271), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for ABCG1 Antibody - BSA Free
Immunoblotting
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Paraffin
Immunoprecipitation
In vivo assay
Simple Western
Western Blot
Reviewed Applications
Read 2 reviews rated 5 using NB400-132 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: ABCG1
A variety of cardiovascular and cardiometabolic diseases are associated with ABCG1 dysfunction (5-7). Macrophages can become cholesterol-containing foam cells that are generated by the uptake of low-density lipoproteins (LDL), cholesterol esterification, and compromised cholesterol efflux machinery in transporters like ABCG1 and ABCA1 (2, 5, 6, 7). Foam cells are associated with the chronic, inflammatory disease atherosclerosis which is characterized by arterial buildup of plaques that can ultimately lead to cardiovascular disease (5, 6, 7). Additionally, ABCG1 has a critical role in cardiometabolic disorders. Studies have found that diabetic mice have decreased ABCG1 expression (8). Furthermore, loss of ABCG1 in mouse pancreatic beta cells ultimately leads to impaired insulin secretion, suggesting that inhibition or modulation of ABCG1 may contribute to development of diabetes and obesity (8). Finally, other related ATP-binding cassette transporter family members, such as ABCA1 and ABCG5/8, have been associated with genetically-inherited syndromes like Tangier disease, characterized by reduced levels of HDL in the blood, and Sitosterolemia, characterized by elevated plant sterol lipid accumulation in blood and tissues (7)..
Alternate names for ABCG1 includes ABC transporter 8 (ABC8), ATP-binding cassette transporter, anti-, sub-family G (WHITE), homolog of Drosophila white, and MGC34313..
References
1. Tarling E. J. (2013). Expanding roles of ABCG1 and sterol transport. Current opinion in lipidology. https://doi.org/10.1097/MOL.0b013e32835da122.
2. Tarr, P. T., Tarling, E. J., Bojanic, D. D., Edwards, P. A., & Baldan, A. (2009). Emerging new paradigms for ABCG transporters. Biochimica et biophysica acta.https://doi.org/10.1016/j.bbalip.2009.01.007.
3. Tarling, E. J., & Edwards, P. A. (2011). ATP binding cassette transporter G1 (ABCG1) is an intracellular sterol transporter. Proceedings of the National Academy of Sciences of the United States of America. https://doi.org/10.1073/pnas.1113021108.
4. Phillips M. C. (2014). Molecular mechanisms of cellular cholesterol efflux. The Journal of biological chemistry, 289(35), 24020-24029. https://doi.org/10.1074/jbc.R114.583658.
5. Ouimet, M., Barrett, T. J., & Fisher, E. A. (2019). HDL and Reverse Cholesterol Transport. Circulation research. https://doi.org/10.1161/CIRCRESAHA.119.312617.
6. Yu, X. H., Fu, Y. C., Zhang, D. W., Yin, K., & Tang, C. K. (2013). Foam cells in atherosclerosis. Clinica chimica acta; international journal of clinical chemistry. https://doi.org/10.1016/j.cca.2013.06.006
Long Name
Alternate Names
Gene Symbol
Additional ABCG1 Products
Product Documents for ABCG1 Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for ABCG1 Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Related Research Areas
Citations for ABCG1 Antibody - BSA Free
Customer Reviews for ABCG1 Antibody - BSA Free (2)
Have you used ABCG1 Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
-
Application: Western BlotSample Tested: ABCG1 transfected HEK293 cell lysate and controls (untransfected cells)Species: HumanVerified Customer | Posted 10/08/2014
-
Application: Western BlotSample Tested:Species: MouseVerified Customer | Posted 08/26/2013
There are no reviews that match your criteria.
Protocols
View specific protocols for ABCG1 Antibody - BSA Free (NB400-132):
IHC-FFPE sections
I. Deparaffinization:
A. Treat slides with Xylene: 3 changes for 5 minutes each. Drain slides for 10 seconds between changes.
B. Treat slides with 100% Reagent Alcohol: 3 changes for 5 minutes each. Drain slides for 10 seconds between changes.
II. Quench Endogenous Peroxidase:
A. Place slides in peroxidase quenching solution: 15-30 minutes.
To Prepare 200 ml of Quenching Solution:
Add 3 ml of 30% Hydrogen Peroxide to 200 ml of Methanol.
Use within 4 hours of preparation
B. Place slides in distilled water: 2 changes for 2 minutes each.
III. Retrieve Epitopes:
A. Preheat Citrate Buffer. Place 200 ml of Citrate Buffer Working Solution into container, cover and place into steamer. Heat to 90-96 degrees Celcius.
B. Place rack of slides into hot Citrate Buffer for 20 minutes. Cover.
C. Carefully remove container with slides from steamer and cool on bench, uncovered, for 20 minutes.
D. Slowly add distilled water to further cool for 5 minutes.
E. Rinse slides with distilled water. 2 changes for 2 minutes each.
IV. Immunostaining Procedure:
A. Remove each slide from rack and circle tissue section with a hydrophobic barrier pen (e.g. Liquid Blocker-Super Pap Pen).
B. Flood slide with Wash Solution. Do not allow tissue sections to dry for the rest of the procedure.
C. Drain wash solution and apply 4 drops of Blocking Reagent to each slide and incubate for 15 minutes.
D. Drain Blocking Reagent (do not wash off the Blocking Reagent), apply 200 ul of Primary Antibody solution to each slide, and incubate for 1 hour.
E. Wash slides with Wash Solution: 3 changes for 5 minutes each.
F. Drain wash solution, apply 4 drops of Secondary antibody to each slide and incubate for 1 hour.
G. Wash slides with Wash Solution: 3 changes for 5 minutes each.
H. Drain wash solution, apply 4 drops of DAB Substrate to each slide and develop for 5-10 minutes. Check development with microscope.
I. Wash slides with Wash Solution: 3 changes for 5 minutes each.
J. Drain wash solution, apply 4 drops of Hematoxylin to each slide and stain for 1-3 minutes. Increase time if darker counterstaining is desired.
K. Wash slides with Wash Solution: 2-3 changes for 2 minutes each.
L. Drain wash solution and apply 4 drops of Bluing Solution to each slide for 1-2 minutes.
M. Rinse slides in distilled water.
N. Soak slides in 70% reagent alcohol: 3 minutes with intermittent agitation.
O. Soak slides in 95% reagent alcohol: 2 changes for 3 minutes each with intermittent agitation.
P. Soak slides in 100% reagent alcohol: 3 changes for 3 minutes each with intermittent agitation. Drain slides for 10 seconds between each change.
Q. Soak slides in Xylene: 3 changes for 3 minutes each with intermittent agitation. Drain slides for 10 seconds between each change.
R. Apply 2-3 drops of non-aqueous mounting media to each slide and mount coverslip.
S. Lay slides on a flat surface to dry prior to viewing under microscope.
NOTES:
-Use treated slides (e.g. HistoBond) to assure adherence of FFPE sections to slide.
-Prior to deparaffinization, heat slides overnight in a 60 degrees Celcius oven.
-All steps in which Xylene is used should be performed in a fume hood.
-For Epitope Retrieval, a microwave or pressure cooker may be substituted for the steamer method. Adjust times as necessary depending on conditions.
-For the initial IHC run with a new primary antibody, test tissues with and without Epitope Retrieval. In some instances, Epitope Retrieval may not be necessary.
-200 ul is the recommended maximum volume to apply to a slide for full coverage. Using more than 200 ul may allow solutions to wick off the slide and create drying artifacts. For small tissue sections less than 200 ul may be used.
-5 minutes of development with DAB Substrate should be sufficient. Do not develop for more than 10 minutes. If 5 minutes of development causes background staining, further dilution of the primary antibody may be necessary.
-Hematoxylin should produce a light nuclear counterstain so as not to obscure the DAB staining. Counterstain for 1-1 1/2 minutes for nuclear antigens. Counterstain for 2-3 minutes for cytoplasmic and membranous antigens. If darker counterstaining is desired increase time (up to 10 minutes).
Western Blot Protocol for NB 400-132
Protein Extraction:
1. After washing with PBS, cells (mouse peritoneal macrophages grown in a 60 mm dish) in 300 ml of cold lysis buffer [50 mM Tris, pH 7.5, 100 mM NaCl, 1% Triton x-100, 1% NaC24H39O4, 50 mM NaF, 1 mM Na3VO4, 1 mM PMSF, 50 ug/ml of aprotinin and 50 ug/ml of leupeptin] are lysed by scraping and sonicating for 25 seconds on ice.
2. Spin cellular lysate for 10 min. at 13,000 rpm at 4 degrees Celcius.
3. Save supernatant and store at -20 degrees Celcius.
Western blotting:
1. Determine protein content by Lowry method.
2. Load 40 ug of cellular protein [pre-boiled for 5 min. in sample buffer] on a 7.5% SDS-PAGE separating gel.
3. Run electrophoresis for 90 min. at RT in 1x electrophoresis buffer.
4. After electrophoresis, equilibrate the gel and nitrocellulose membrane in transfer buffer.
5. Transfer proteins in 1x transfer buffer for 1 hour at 100 volts and RT.
6. Block the membrane in 10 ml of TBS with 5% NFDM for 1 hour at RT.
7. After a quick rinse with TBS-T (0.5% Tween-20), membrane is incubated in diluted anti-ABCG1 (cat# NB 400-132) in 1% NFDM/TBS for 1.5 hours at RT.
8. Wash the membrane in 25 ml of TBS-T for 3x 5 minutes at RT.
9. Incubate the membrane in 10 ml of diluted secondary antibody (Anti-Rabbit IgG-HRP Conjugate) in 1% NFDM/TBS for 1 hour at RT.
10. Wash the membrane with 25 ml of TBS-T for 3x 5 minutes at RT.
11. Incubate the membrane in ECL Western blotting detection reagents for 1 minute.
12. Expose to film for ~2 minutes (adjust time as needed for best image).
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for ABCG1 Antibody - BSA Free
-
Q: What secondary antibodies would be recommended for immunofluorescent or Western blot assays when using NB400-132 as the primary antibody?
A: Under the "Support and Research" tab on the NB400-132 product page on the Novus website, there are links to Anti-Rabbit labeled antibodies and isotype controls that may be useful for your experiments.

![Immunohistochemistry-Paraffin: ABCG1 Antibody - BSA Free [NB400-132] Immunohistochemistry-Paraffin: ABCG1 Antibody - BSA Free [NB400-132]](https://resources.rndsystems.com/images/products/ABCG1-Antibody---BSA-Free-Immunohistochemistry-Paraffin-NB400-132-img0013.jpg)
![Flow (Intracellular): ABCG1 Antibody - BSA Free [NB400-132] Flow (Intracellular): ABCG1 Antibody - BSA Free [NB400-132]](https://resources.rndsystems.com/images/products/ABCG1-Antibody-BSA-Free-Flow-Intracellular-NB400-132-img0012.jpg)
![Western Blot: ABCG1 AntibodyBSA Free [NB400-132] Western Blot: ABCG1 AntibodyBSA Free [NB400-132]](https://resources.rndsystems.com/images/products/ABCG1-Antibody---BSA-Free-Western-Blot-NB400-132-img0005.jpg)
![Western Blot: ABCG1 AntibodyBSA Free [NB400-132] Western Blot: ABCG1 AntibodyBSA Free [NB400-132]](https://resources.rndsystems.com/images/products/ABCG1-Antibody---BSA-Free-Western-Blot-NB400-132-img0009.jpg)
![Western Blot: ABCG1 AntibodyBSA Free [NB400-132] Western Blot: ABCG1 AntibodyBSA Free [NB400-132]](https://resources.rndsystems.com/images/products/ABCG1-Antibody---BSA-Free-Western-Blot-NB400-132-img0004.jpg)
![Western Blot: ABCG1 AntibodyBSA Free [NB400-132] Western Blot: ABCG1 AntibodyBSA Free [NB400-132]](https://resources.rndsystems.com/images/products/ABCG1-Antibody---BSA-Free-Western-Blot-NB400-132-img0014.jpg)
![Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - ABCG1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb400-132_rabbit-polyclonal-abcg1-antibody-210202423454887.jpg)
![Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - ABCG1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb400-132_rabbit-polyclonal-abcg1-antibody-210202423454895.jpg)
![Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - ABCG1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb400-132_rabbit-polyclonal-abcg1-antibody-210202423452427.jpg)
![Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - ABCG1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb400-132_rabbit-polyclonal-abcg1-antibody-210202423452419.jpg)
![Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - ABCG1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb400-132_rabbit-polyclonal-abcg1-antibody-21020242345242.jpg)
![Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - ABCG1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb400-132_rabbit-polyclonal-abcg1-antibody-210202423454874.jpg)
![Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - ABCG1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb400-132_rabbit-polyclonal-abcg1-antibody-310202415291917.jpg)
![Western Blot: ABCG1 Antibody - BSA Free [NB400-132] - ABCG1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb400-132_rabbit-polyclonal-abcg1-antibody-31020241618932.jpg)
