Canine MCP-1 (monocyte chemotactic protein-1) is an 8 kDa member of the CC chemokine family of chemotactic factors (1, 2). It is synthesized as a 101 amino acid (aa) precursor that contains a 23 aa signal sequence and a 78 aa mature segment (3). It contains no potential N-linked glycosylation sites and is not known for any posttranslational modifications. Based on human studies, MCP-1 will primarily circulate as a monomer. Noncovalent dimers are likely to be found, however. MCP‑1 activity has been localized to the N-terminus (1). Cell types known to secrete MCP-1 are considerable in number, and include keratinocytes, fibroblasts, endothelium, osteoblasts, macrophages, mast cells, smooth muscle cells and astrocytes (1, 2). In the mature MCP-1 segment, there is 82% and 83% aa identity, canine to human and porcine MCP-1, respectively. When mature canine MCP-1 is compared to (125 aa) extended rodent MCP-1, there is 55% and 56% aa identity, canine to mouse and rat MCP-1, respectively. MCP-1 has three possible receptors. The first two are CCR2 (1) and CCR11 (4). The third receptor has only been identified in mice and is called L-CCR (5). Its function is unknown. MCP-1 is best known as a chemotactic agent for mononuclear cells. It also, however, induces enzyme and cytokine release in monocytes, NK cells, and lymphocytes and histamine release by basophils (1). Additionally, it is believed to reduce IL‑12 production by dendritic cells and promote a Th2 phenotype in CD4+ T cells (6).
Canine CCL2/JE/MCP‑1 Biotinylated Antibody
R&D Systems | Catalog # BAF1774

Key Product Details
Species Reactivity
Canine
Applications
Western Blot, ELISA Detection (Matched Antibody Pair), Immunocytochemistry
Label
Biotin
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant canine CCL2/JE/MCP‑1
Gln24-Pro101
Accession # P52203
Gln24-Pro101
Accession # P52203
Specificity
Detects canine CCL2/JE/MCP‑1 in ELISAs and Western blots. In sandwich immunoassay, less than 0.05% cross‑reactivity with recombinant mouse MCP-1 and recombinant human MCP-1 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Canine CCL2/JE/MCP‑1 Biotinylated Antibody
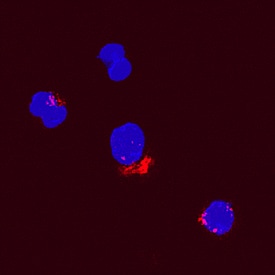
CCL2/JE/MCP‑1 in Canine PBMCs.
CCL2/JE/MCP-1 was detected in immersion fixed canine peripheral blood mononuclear cells (PBMCs) using Goat Anti-Canine CCL2/JE/MCP-1 Biotinylated Antigen Affinity-purified Polyclonal Antibody (Catalog # BAF1774) at 15 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Streptavidin (red; Catalog # NL999) and counterstained with DAPI (blue). Specific staining was localized to cytoplasmic. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Applications for Canine CCL2/JE/MCP‑1 Biotinylated Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed canine peripheral blood mononuclear cells (PBMCs)
Sample: Immersion fixed canine peripheral blood mononuclear cells (PBMCs)
Western Blot
0.1 µg/mL
Sample: Recombinant Canine CCL2/JE/MCP‑1 (Catalog # 1774-MC)
Sample: Recombinant Canine CCL2/JE/MCP‑1 (Catalog # 1774-MC)
Canine CCL2/JE/MCP-1 Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein.
Shipping
The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CCL2/JE/MCP-1
References
- Coillie, E.V. et al. (1999) Cytokine Growth Factor Rev. 10:61.
- Yoshie, O. et al. (2001) Adv. Immunol. 78:57.
- Kumar, A.G. et al. (1997) Circulation 95:693.
- Biber, K. et al. (2003) J. Leukoc. Biol. 74:243.
- Luther, S.A. and J.G. Cyster (2001) Nat. Immunol. 2:102.
Alternate Names
GDCF-2, HC11, HSMCR30, MCAF, MCP-1, SMC-CF
Gene Symbol
CCL2
UniProt
Additional CCL2/JE/MCP-1 Products
Product Documents for Canine CCL2/JE/MCP‑1 Biotinylated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Canine CCL2/JE/MCP‑1 Biotinylated Antibody
For research use only
Customer Reviews for Canine CCL2/JE/MCP‑1 Biotinylated Antibody
There are currently no reviews for this product. Be the first to review Canine CCL2/JE/MCP‑1 Biotinylated Antibody and earn rewards!
Have you used Canine CCL2/JE/MCP‑1 Biotinylated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars



