Interleukin 2 was initially identified as a T cell growth factor that is produced by T cells following activation by mitogens or antigens. Since then, it has been shown that IL-2 can also stimulate the growth and differentiation of B cells, natural killer (NK) cells, lymphocyte activated killer (LAK) cells, monocytes/macrophages, and oligodendrocytes. The biological activity of IL-2 is mediated by the binding to cell surface receptor complexes composed of three subunits designated as alpha, beta, and gamma subunits. IL-2 binds the alpha subunit with low affinity. The functional high affinity IL-2 receptor is a heterotrimeric complex of the alpha, beta, and gamma subunits. IL-2 binds with intermediate affinity to the complex containing the beta and gamma subunits, which is also capable of transducing IL-2 signals. In T cells, the beta and gamma subunits are shared with the IL-15 receptor complex. The gamma subunit of the IL-2 receptor complex has also been shown to be a subunit of the receptor complexes of IL-4, IL-7, and IL-9. At the amino acid sequence level, canine IL-2 shares 90%, 86%, 85%, 76%, and 75% sequence similarities to feline, human, equine, mouse, and bovine IL-2, respectively.
Canine IL‑2 Biotinylated Antibody
R&D Systems | Catalog # BAF1815

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Canine
Cited:
Mouse
Applications
Validated:
Western Blot, ELISA Detection (Matched Antibody Pair), Immunocytochemistry
Cited:
Immunohistochemistry
Label
Biotin
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant canine IL‑2
Ala21-Thr155 (Cys147Ser)
Accession # Q29416
Ala21-Thr155 (Cys147Ser)
Accession # Q29416
Specificity
Detects canine IL-2 in ELISAs and Western blots. In sandwich immunoassays, less than 0.2% cross-reactivity with recombinant human IL-2, recombinant mouse IL-2, recombinant rat IL-2, recombinant feline IL-2, recombinant bovine IL-2, recombinant equine IL-2, recombinant cotton rat IL-2, and recombinant porcine IL-2 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Canine IL‑2 Biotinylated Antibody
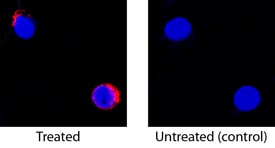
IL‑2 in Canine PBMCs.
IL-2 was detected in immersion fixed canine peripheral blood mononuclear cells (PBMCs) untreated or treated with calcium ionomycin and PMA using Goat Anti-Canine IL-2 Biotinylated Antigen Affinity-purified Polyclonal Antibody (Catalog # BAF1815) at 15 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Streptavidin (red; Catalog # NL999) and counterstained with DAPI (blue). Specific staining was localized to cell secretion. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Applications for Canine IL‑2 Biotinylated Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed canine peripheral blood mononuclear cells treated with calcium ionomycin and PMA
Sample: Immersion fixed canine peripheral blood mononuclear cells treated with calcium ionomycin and PMA
Western Blot
0.1 µg/mL
Sample: Recombinant Canine IL‑2 (Cys147Ser) (Catalog # 1815-CL)
Sample: Recombinant Canine IL‑2 (Cys147Ser) (Catalog # 1815-CL)
Canine IL-2 Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein.
Shipping
The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-2
Long Name
Interleukin 2
Alternate Names
Aldesleukin, IL2, Proleukin, TCGF
Entrez Gene IDs
Gene Symbol
IL2
UniProt
Additional IL-2 Products
Product Documents for Canine IL‑2 Biotinylated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Canine IL‑2 Biotinylated Antibody
For research use only
Related Research Areas
Citations for Canine IL‑2 Biotinylated Antibody
Customer Reviews for Canine IL‑2 Biotinylated Antibody
There are currently no reviews for this product. Be the first to review Canine IL‑2 Biotinylated Antibody and earn rewards!
Have you used Canine IL‑2 Biotinylated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
 Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
 IL-2 Signaling Pathways
IL-2 Signaling Pathways
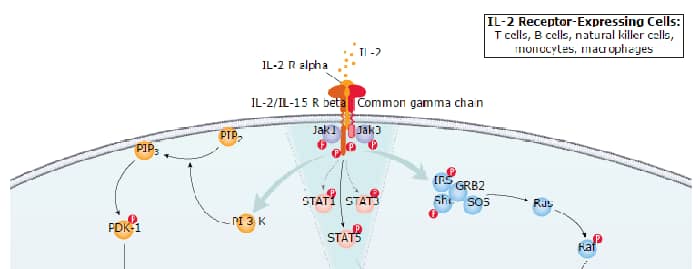 IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types

Innate Lymphoid Cell Differentiation Pathways
 Jak/STAT Signaling Pathway
Jak/STAT Signaling Pathway
 Th1 Differentiation Pathway
Th1 Differentiation Pathway
 Th2 Differentiation Pathway
Th2 Differentiation Pathway

 Jak/STAT Signaling Pathway
Jak/STAT Signaling Pathway
 Th1 Differentiation Pathway
Th1 Differentiation Pathway
 Th2 Differentiation Pathway
Th2 Differentiation Pathway

