CD11b/c Antibody - BSA Free
Novus Biologicals | Catalog # NB110-40766

![Western Blot: CD11b/c AntibodyBSA Free [NB110-40766] Western Blot: CD11b/c AntibodyBSA Free [NB110-40766]](https://resources.rndsystems.com/images/products/CD11b-c-Antibody---BSA-Free-Western-Blot-NB110-40766-img0002.jpg)
Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Localization
Marker
Clonality
Host
Isotype
Theoretical MW
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for CD11b/c Antibody - BSA Free
Western Blot: CD11b/c AntibodyBSA Free [NB110-40766]
Western Blot: CD11b/c Antibody - BSA Free [NB110-40766] - Analysis of CD11b/c in Raw 264.7 whole cell lysate.Immunocytochemistry/ Immunofluorescence: CD11b/c Antibody - BSA Free [NB110-40766]
Immunocytochemistry/Immunofluorescence: CD11b/c Antibody - BSA Free [NB110-40766] - CD11 antibody was tested in Raw264.7 cells with FITC (green). Nuclei were counterstained with DAPI (blue).Immunohistochemistry-Frozen: CD11b/c Antibody - BSA Free [NB110-40766]
Immunohistochemistry-Frozen: CD11b/c Antibody - BSA Free [NB110-40766] - 10 um Cryosection of murine acute brain slices stained with CD11b/c antibody (1:300 = 3.3 ug/ml) and Alexa Fluor 555 donkey anti rabbit IgG. Antibody shows specific staining, but the background is quite heavy. Image from verified customer review.Flow Cytometry: CD11b/c Antibody - BSA Free [NB110-40766]
Flow Cytometry: CD11b/c Antibody - BSA Free [NB110-40766] - A surface stain was performed on RAW 264.7 with NB110-40766 and a matched isotype control. Cells were incubated in an antibody dilution of 2.5 ug/mL for 30 minutes at room temperature, followed by Rabbit IgG (H+L) Cross-Adsorbed Secondary Antibody, DyLight 550.Immunocytochemistry/ Immunofluorescence: CD11b/c Antibody - BSA Free [NB110-40766]
CD11b-c-Antibody---BSA-Free-Immunocytochemistry-Immunofluorescence-NB110-40766-img0010.jpgImmunohistochemistry: CD11b/c Antibody - BSA Free [NB110-40766]
Immunohistochemistry: CD11b/c Antibody - BSA Free [NB110-40766] - Staining of human bone marrow, myeloid precursors.Western Blot: CD11b/c Antibody - BSA Free [NB110-40766] -
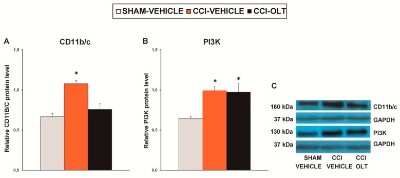
Western Blot: CD11b/c Antibody - BSA Free [NB110-40766] - Effects of oltipraz on the expression of CD11b/c, phosphoinositide 3-kinase (PI3K), phosphorylated protein kinase B (p-Akt), & phosphorylated inhibitor of kappa B alpha (p-IkB alpha ) in the spinal cord of the CCI-injured mice. The relative protein levels of (A) CD11b/c, (B) PI3K, (D) p-Akt, & (E) p-IKB alpha on the ipsilateral side of the spinal cord in the CCI-injured mice treated with oltipraz (OLT) or vehicle are represented. The sham-operated mice (SHAM) treated with vehicle were used as controls. (C) Representative examples of blots for CD11b/c (160 kDa), PI3K (130 kDa), & GAPDH (37 kDa), & (F) for p-Akt (60 kDa), Akt (60 kDa), p-IKB alpha (40 kDa) & IKB alpha (40 kDa). CD11b/c & PI3K are expressed relative to GAPDH levels whereas phosphorylated proteins are expressed relative to their corresponding total proteins. In all panels, * denotes significant differences vs. sham-operated mice treated with vehicle (p < 0.05; one-way ANOVA followed by the SNK test). Results are presented as the mean ± SEM; n = 5 samples per experimental group. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31234342), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: CD11b/c Antibody - BSA Free [NB110-40766] -
Western Blot: CD11b/c Antibody - BSA Free [NB110-40766] - Effects of oltipraz on the expression of CD11b/c, PI3K, p-Akt, & p-IkB alpha in the prefrontal cortex of the CCI-injured mice. The relative protein levels of (A) CD11b/c, (B) PI3K, (D) p-Akt, & (E) p-IKB alpha in the prefrontal cortex of the CCI-injured mice treated with oltipraz (OLT) or vehicle. The sham-operated mice (SHAM) treated with vehicle were used as controls. Representative examples of blots for (C) CD11b/c (160 kDa), PI3K (130 kDa) & GAPDH (37 kDa), & for (F) p-Akt (60 kDa), Akt (60 kDa), p-IKB alpha (40 kDa) & IKB alpha (40 kDa). CD11b/c & PI3K are expressed relative to GAPDH levels whereas phosphorylated proteins are expressed relative to their corresponding total proteins. In all panels, * denotes significant differences vs. sham-operated mice treated with vehicle (p < 0.05; one-way ANOVA followed by the SNK test). Results are presented as the mean ± SEM; n = 4-–5 samples per experimental group. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31234342), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: CD11b/c Antibody - BSA Free [NB110-40766] -
Western Blot: CD11b/c Antibody - BSA Free [NB110-40766] - Effects of oltipraz on the expression of CD11b/c, PI3K, p-Akt, & p-IkB alpha in the hippocampus of the CCI-injured mice. The relative protein levels of (A) CD11b/c, (B) PI3K, (D) p-Akt, & (E) p-IKB alpha in the hippocampus of the CCI-injured mice treated with oltipraz (OLT) or vehicle. The sham-operated mice (SHAM) treated with vehicle were used as controls. Representative examples of blots for (C) CD11b/c (160 kDa), PI3K (130 kDa), & GAPDH (37 kDa); & (F) for p-Akt (60 kDa), Akt (60 kDa), p-IKB alpha (40 kDa), & IKB alpha (40 kDa). CD11b/c & PI3K are expressed relative to GAPDH levels whereas phosphorylated proteins are expressed relative to their corresponding total proteins. In all panels, * denotes significant differences vs. sham-operated mice treated with vehicle (p < 0.05; one-way ANOVA followed by the SNK test). Results are presented as the mean ± SEM; n = 5 samples per experimental group. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31234342), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: CD11b/c Antibody - BSA Free [NB110-40766] -
Immunocytochemistry/ Immunofluorescence: CD11b/c Antibody - BSA Free [NB110-40766] - Phase contrast microscopy images of HFA & brain tumor cells & immunofluorescence images of cultured primary brain tumor cells, AA & HFA.HFA were derived from three different foetal brains. (A) 18-week foetus (10× magnification); (B) 19-week foetus, first week in culture (10× magnification); (C) 19-week foetus, day 5 in culture (20× magnification); (D) Primary GBM after 2 trypsinisations (20x); (E) Recurrent GBM (20x); (F) Secondary GBM after 3 trypsinisations (10x); (G) OII 14 days in culture (10x); (H) AII 13 days in culture (10x); (I) Negative control for NGS merged with DAPI; (J) Negative control for secondary antibodies merged with DAPI; (K) GBM cells derived from one patient – GFAP (DAKO) merged with DAPI & CD68 (zoomed: 2.4× magnification); (L) primary brain tumor cells from one patient (male, 40 years old) (passaged three times) - GFAP (Novocastra) merged with DAPI; (K & L) GFAP (green) was used as a GBM marker & CD68 (red) was used as a marker for microglia. (M) AA from one Female, 61 years old; (passaged twice) - GFAP (Novocastra) merged with DAPI; (N) AA from one male 40 years old; (passaged once) - GFAP (Sigma) merged with DAPI & CD68 (Abcam); (M & N) GFAP (green) was used as an astrocyte marker & CD68 (red) & CD11b (green) were used as markers for microglia; (O) HFA from one 17-week- old foetus (passaged once) - GFAP (Novocastra) merged with DAPI & CD11b (Novus); (P) HFA from an 18-week- old foetus (passaged once) - GFAP (DAKO) merged with DAPI & CD68 (zoomed: 2.1× magnification); (O & P) GFAP (green & red) was used as an astrocyte marker & CD68 (red) & CD11b (green) were used as markers for microglia. Nuclei indicated by DAPI (blue) in all images. Image collected & cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0112945), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for CD11b/c Antibody - BSA Free
Flow Cytometry
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Frozen
Immunohistochemistry-Paraffin
Western Blot
Reviewed Applications
Read 3 reviews rated 3.7 using NB110-40766 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: CD11b/c
Alternate Names
Gene Symbol
Additional CD11b/c Products
Product Documents for CD11b/c Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for CD11b/c Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for CD11b/c Antibody - BSA Free
Customer Reviews for CD11b/c Antibody - BSA Free (3)
Have you used CD11b/c Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Immunohistochemistry-FrozenSample Tested: Mouse brainSpecies: MouseVerified Customer | Posted 03/11/201910 µm Cryosection of murina acute brain slices stained with CD11b/c antibody (1:300 = 3.3 µg/ml) and Alexa Fluor 555 donkey anti rabbit IgG. Antibody shows specific staining, but the background is quite heavy.
-
Application: ImmunohistochemistrySample Tested: Tumor sectionsSpecies: HumanVerified Customer | Posted 08/04/2010
-
Application: Immunohistochemistry-ParaffinSample Tested: Human. Mucous membrane of the small intestineSpecies: HumanVerified Customer | Posted 01/30/2009
There are no reviews that match your criteria.
Protocols
View specific protocols for CD11b/c Antibody - BSA Free (NB110-40766):
Immunocytochemistry Protocol
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and add 10% formalin to the dish. Fix at room temperature for 30 minutes.
2. Remove the formalin and add ice cold methanol. Incubate for 5-10 minutes.
3. Remove methanol and add washing solution (i.e. PBS). Be sure to not let the specimen dry out. Wash three times for 10 minutes.
4. To block nonspecific antibody binding incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
5. Add primary antibody at appropriate dilution and incubate at room temperature from 2 hours to overnight at room temperature.
6. Remove primary antibody and replace with washing solution. Wash three times for 10 minutes.
7. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
8. Remove antibody and replace with wash solution, then wash for 10 minutes. Add Hoechst 33258 to wash solution at 1:25,0000 and incubate for 10 minutes. Wash a third time for 10 minutes.
9. Cells can be viewed directly after washing. The plates can also be stored in PBS containing Azide covered in Parafilm (TM). Cells can also be cover-slipped using Fluoromount, with appropriate sealing.
*The above information is only intended as a guide. The researcher should determine what protocol best meets their needs. Please follow safe laboratory procedures.
Immunohistochemistry
1. Prepare tissue with formalin fixation and by embedding it in paraffin wax.
2. Make 4-mm sections and place on pre-cleaned and charged microscope slides.
3. Heat in a tissue-drying oven for 45 minutes @ 60 degrees Celcius.
4. Deparaffinize the tissues by wash drying the slides in 3 changes of xylene for 5 minutes each @ RT.
5. Rehydrate the tissues by washing the slides in 3 changes of 100% alcohol for 3 minutes each @ RT.
6. Wash the slides in 2 changes of 95% alcohol for 3 minutes each @ RT.
7. Wash the slides in 1 change of 80% alcohol for 3 minutes @ RT.
8. Rinse the slides in gentle running distilled water for 5 minutes @ RT.
9. Perform antigen retrieval by steaming the slides in 0.01M sodium citrate buffer (pH 6.0) @ 99-100 degrees Celcius for 20 minutes.
10. Remove the slides from the heat and let stand in buffer @ RT for 20 minutes.
11. Rinse the slides in 1X TBS-T for 1 minute @ RT.
**Do not allow the tissues to dry at any time during the staining procedure**
12. Begin the immunostaining by applying a universal protein block for 20 minutes @ RT.
13. Drain protein block from the slides and apply the diluted primary antibody for 45 minutes @ RT.
14. Rinse the slide in 1X TBS-T for 1 minute @ RT.
15. Apply a biotinylated anti-rabbit IgG (H+L) secondary for 30 minutes @ RT.
16. Rinse the slide in 1X TBS-T for 1 minute @ RT.
17. Apply an alkaline phosphatase steptavidin for 30 minutes @ RT.
18. Rinse the slide in 1X TBS-T for 1 minute @ RT.
19. Apply an alkaline phosphatase chromagen substrate for 30 minutes @ RT.
20. Rinse the slide in distilled water for 1 minute @ RT.
**This method should only be used if the chromagen substrate is alcohol insoluble (ie: Vector Red, DAB)**
21. Dehydrate the tissue by washing the slides in 2 changes of 80% alcohol for 1 minute each @ RT.
22. Wash the slides in 2 changes of 95% alcohol for 1 minute each @ RT.
23. Wash the slides in 3 changes of 100% alcohol for 1 minute each @ RT.
24. Wash the slides in 3 changes of xylene for 1 minute each @ RT.
25. Apply cover slip.
Western Blot Protocol
1. Perform SDS-PAGE on samples to be analyzed, loading 40 ug of total protein per lane.
2. Transfer proteins to membrane according to the instructions provided by the manufacturer of the membrane and transfer apparatus.
3. Stain according to standard Ponceau S procedure (or similar product) to assess transfer success, and mark molecular weight standards where appropriate.
4. Rinse the blot.
5. Block the membrane using standard blocking buffer for at least 1 hour.
6. Wash the membrane in wash buffer three times for 10 minutes each.
7. Dilute primary antibody in blocking buffer and incubate 1 hour at room temperature.
8. Wash the membrane in wash buffer three times for 10 minutes each.
9. Apply the diluted HRP conjugated secondary antibody in blocking buffer (as per manufacturers instructions) and incubate 1 hour at room temperature.
10. Wash the blot in wash buffer three times for 10 minutes each (this step can be repeated as required to reduce background).
11. Apply the detection reagent of choice in accordance with the manufacturers instructions.
**Note: Tween-20 can be added to the blocking or antibody dilution buffer at a final concentration of 0.05-0.2%.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for CD11b/c Antibody - BSA Free
-
Q: I am trying to stain microglia in paraformaldehyde fixed (overnight), frozen sectioned (25 micron) free floating sections of rat brain with NB110-40766. I have yet to produce any significant staining. I have tried both 5 and 10 ug/ml concentrations of primary antibody and have tried the protocol with and without antigen retrieval using heated sodium citrate. Considering this is a membrane bound protein, could the triton be inhibiting antigen/antibody interaction, and would trying something less harsh such as Tween 20 be worthwhile?
A: Since our product will detect an antigen in the extracellular domain of the CD11b protein, I think elimination of the Triton X may help substantially. You can try replacing with 0.1% Tween-20, or just eliminating the detergent all together. We have only been able to validate this antibody in paraffin-embedded tissue sections, so we do not have a recommended protocol for frozen or free-floating sections at this time. However, with a fixation in PFA overnight, I think performing some type of antigen retrieval would be highly recommended. From other protocols for antigen retrieval on frozen tissue sections, heat induced with sodium citrate (pH 6.0) is the most commonly used buffer. I think with the modification to the detergent, you should be able to see a signal in your tissue.
-
Q: What type of antigen retrieval would you recommend for CD11b/c Antibody (NB110-40766)? I am working with paraformaldehyde-fixed free-floating mouse brain sections (40um).
A:
In regards to your inquiry I have spoken with the lab and while we have tested paraffin embedded sections and not free floating, we would recommend citrate buffer pH 6.0. Since they are free floating the lab indicates you can probably incubate for less time with the buffer. We would also recommend using a no antigen retrieval control. Especially if you wanted to try out a few to see which was the optimal one to use. Please see our product specific IHC-P protocol.
-
Q: I am trying to stain microglia in paraformaldehyde fixed (overnight), frozen sectioned (25 micron) free floating sections of rat brain with NB110-40766. I have yet to produce any significant staining. I have tried both 5 and 10 ug/ml concentrations of primary antibody and have tried the protocol with and without antigen retrieval using heated sodium citrate. Considering this is a membrane bound protein, could the triton be inhibiting antigen/antibody interaction, and would trying something less harsh such as Tween 20 be worthwhile?
A: Since our product will detect an antigen in the extracellular domain of the CD11b protein, I think elimination of the Triton X may help substantially. You can try replacing with 0.1% Tween-20, or just eliminating the detergent all together. We have only been able to validate this antibody in paraffin-embedded tissue sections, so we do not have a recommended protocol for frozen or free-floating sections at this time. However, with a fixation in PFA overnight, I think performing some type of antigen retrieval would be highly recommended. From other protocols for antigen retrieval on frozen tissue sections, heat induced with sodium citrate (pH 6.0) is the most commonly used buffer. I think with the modification to the detergent, you should be able to see a signal in your tissue.
-
Q: What type of antigen retrieval would you recommend for CD11b/c Antibody (NB110-40766)? I am working with paraformaldehyde-fixed free-floating mouse brain sections (40um).
A:
In regards to your inquiry I have spoken with the lab and while we have tested paraffin embedded sections and not free floating, we would recommend citrate buffer pH 6.0. Since they are free floating the lab indicates you can probably incubate for less time with the buffer. We would also recommend using a no antigen retrieval control. Especially if you wanted to try out a few to see which was the optimal one to use. Please see our product specific IHC-P protocol.

![Immunocytochemistry/ Immunofluorescence: CD11b/c Antibody - BSA Free [NB110-40766] Immunocytochemistry/ Immunofluorescence: CD11b/c Antibody - BSA Free [NB110-40766]](https://resources.rndsystems.com/images/products/CD11b-c-Antibody---BSA-Free-Immunocytochemistry-Immunofluorescence-NB110-40766-img0006.jpg)
![Immunohistochemistry-Frozen: CD11b/c Antibody - BSA Free [NB110-40766] Immunohistochemistry-Frozen: CD11b/c Antibody - BSA Free [NB110-40766]](https://resources.rndsystems.com/images/products/CD11b-c-Antibody---BSA-Free-Immunohistochemistry-Frozen-NB110-40766-img0009.jpg)
![Flow Cytometry: CD11b/c Antibody - BSA Free [NB110-40766] Flow Cytometry: CD11b/c Antibody - BSA Free [NB110-40766]](https://resources.rndsystems.com/images/products/CD11b-c-Antibody---BSA-Free-Flow-Cytometry-NB110-40766-img0008.jpg)
![Immunocytochemistry/ Immunofluorescence: CD11b/c Antibody - BSA Free [NB110-40766] Immunocytochemistry/ Immunofluorescence: CD11b/c Antibody - BSA Free [NB110-40766]](https://resources.rndsystems.com/images/products/CD11b-c-Antibody---BSA-Free-Immunocytochemistry-Immunofluorescence-NB110-40766-img0010.jpg)
![Immunohistochemistry: CD11b/c Antibody - BSA Free [NB110-40766] Immunohistochemistry: CD11b/c Antibody - BSA Free [NB110-40766]](https://resources.rndsystems.com/images/products/CD11b-c-Antibody---BSA-Free-Immunohistochemistry-NB110-40766-img0007.jpg)
![Western Blot: CD11b/c Antibody - BSA Free [NB110-40766] - CD11b/c Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb110-40766_rabbit-polyclonal-cd11b-c-antibody-310202416235533.jpg)
![Western Blot: CD11b/c Antibody - BSA Free [NB110-40766] - CD11b/c Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb110-40766_rabbit-polyclonal-cd11b-c-antibody-310202416235518.jpg)
![Western Blot: CD11b/c Antibody - BSA Free [NB110-40766] - CD11b/c Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb110-40766_rabbit-polyclonal-cd11b-c-antibody-310202416235538.jpg)
![Immunocytochemistry/ Immunofluorescence: CD11b/c Antibody - BSA Free [NB110-40766] - CD11b/c Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb110-40766_rabbit-polyclonal-cd11b-c-antibody-310202416235543.jpg)