Ferroportin/SLC40A1 Antibody - BSA Free
Novus Biologicals | Catalog # NBP1-21502

Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Localization
Clonality
Host
Isotype
Theoretical MW
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for Ferroportin/SLC40A1 Antibody - BSA Free
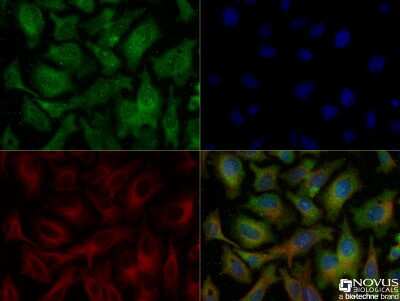
Immunocytochemistry/Immunofluorescence Analysis of Ferroportin/SLC40A1 in HepG2 Cells
HepG2 cells were fixed and permeabilized for 10 minutes using -20C MeOH. The cells were incubated with Ferroportin/SLC40A1 Antibody at 5.0 ug/ml overnight at 4C and detected with an anti-rabbit DyLight 488 (green) at a 1:500 dilution. Alpha tubulin (DM1A) [NB100-690] was used as a co-stain at a 1:1000 dilution and detected with an anti-mouse DyLight 550 (red) at a 1:500 dilution. Nuclei were counterstained with DAPI (blue). Cells were imaged using a 40X objective.Immunohistochemical Staining of Ferroportin/SLC40A1 in Paraffin Embedded Mouse Small Intestine
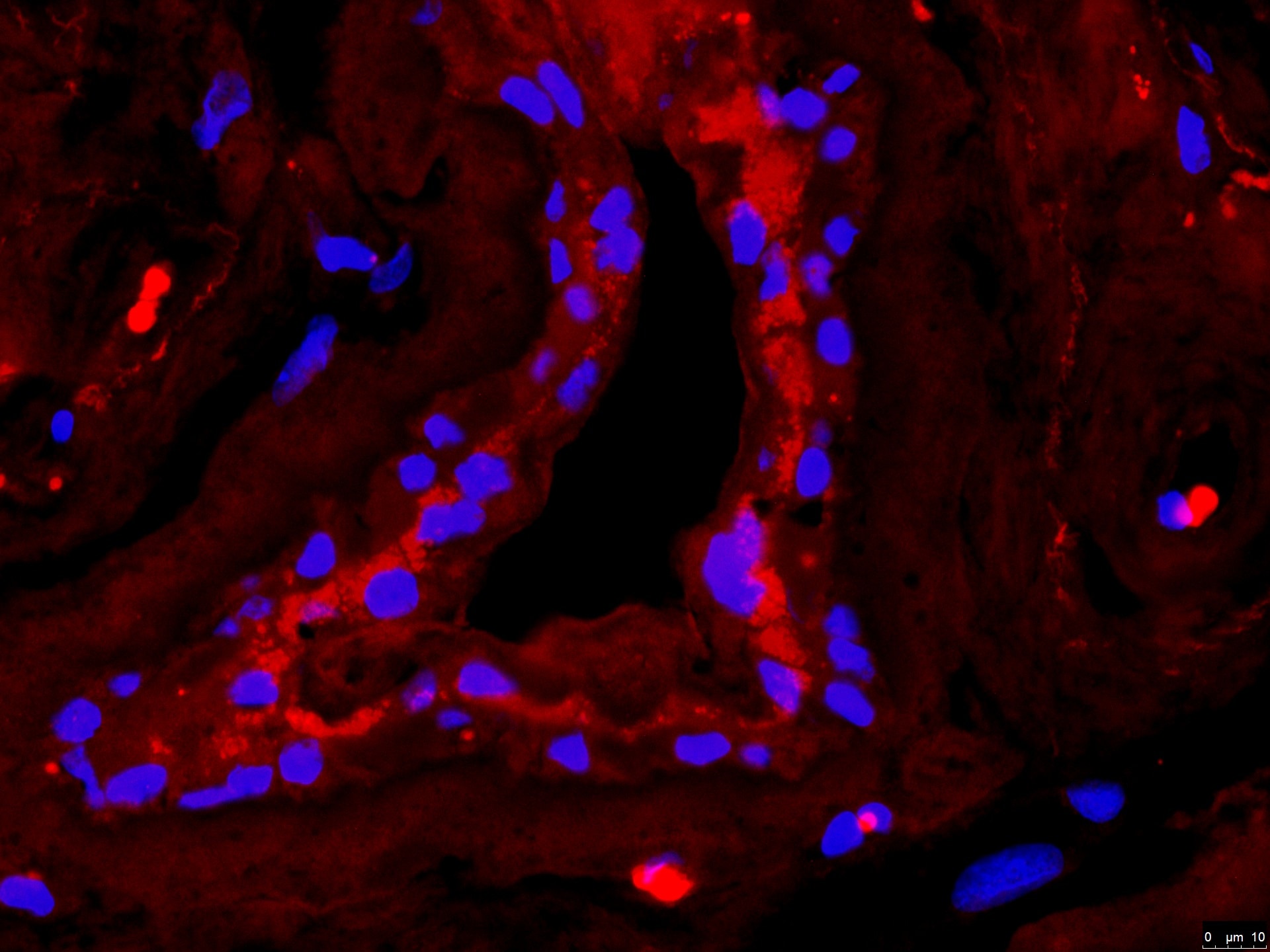
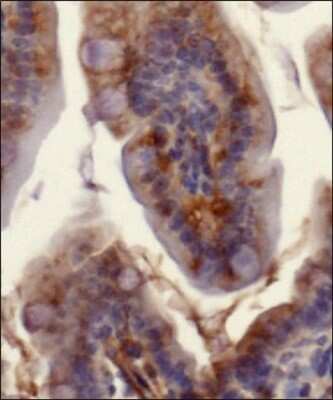
Detection of Ferroportin/SLC40A1 protein in murine small intestinal section using Ferroportin/SLC40A1 Antibody at a dilution of 1:200. The antibody primarily developed a membranous staining pattern in the intestinal epithelial cells.Flow Cytometry of HepG2 Cells Stained with Ferroportin/SLC40A1 Antibody
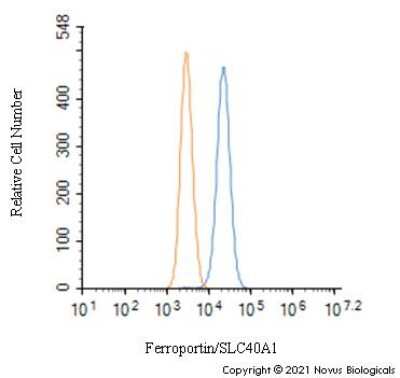
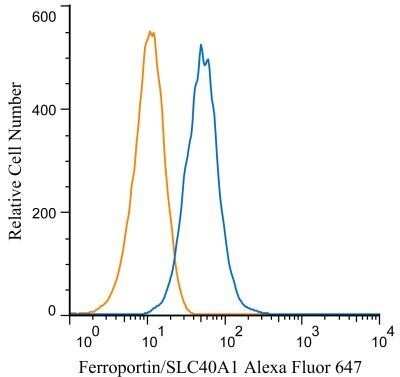
An intracellular stain was performed on HepG2 cells with Ferroportin/SLC40A1 Antibody NBP1-21502 (blue) and a matched isotype control NBP2-24891 (orange). Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 1.0 ug/mL for 30 minutes at room temperature, followed by Rabbit IgG (H+L) Cross-Adsorbed Secondary Antibody, Dylight 550 (SA5-10033, Thermo Fisher).Western Blot Detection of Ferroportin/SLC40A1 in Human Intestine Lysate
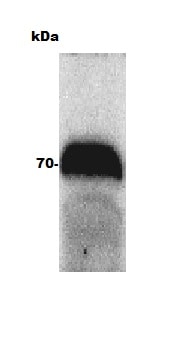
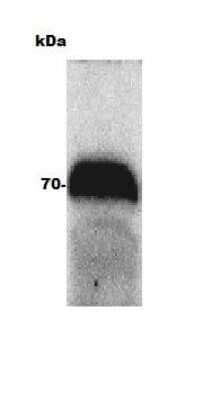
Western blot of human intestine lysate (Molecular weight: 62.5 KDa) using Ferroportin/SLC40A1 Antibody.Western Blot Detection of Ferroportin/SLC40A1 in Bovine Adult Eye
Western blot of bovine adult eye using Ferroportin/SLC40A1 Antibody. Image from verified customer review.Western Blot Detection of Ferroportin/SLC40A1 in Porcine Duodenal Mucosa
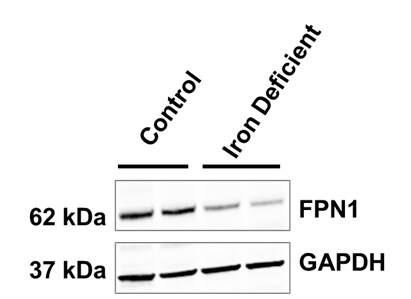
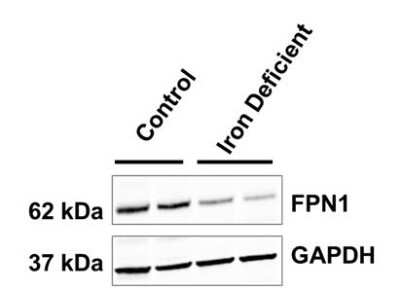
Duodenal mucosa samples from weanling pigs with iron replete or iron deficient status were probed using (NBP1-21502; 1:1000 dilution) and GAPDH (loading control). Image from verified customer review.Western Blotting of Ferroportin/SLC40A1 in Control and Transfected HepG2 Cell Lines
Ferroportin-SLC40A1-Antibody---BSA-Free-Western-Blot-NBP1-21502-img0019.jpgImmunohistochemical Staining of Ferroportin/SLC40A1 in Human Eye Segment
Ferroportin-SLC40A1 Antibody - BSA Free-Immunocytochemistry-Immunofluorescence-NBP1-21502-img0022.jpgImmunohistochemical Detection of Ferroportin/SLC40A1 in Murine Liver
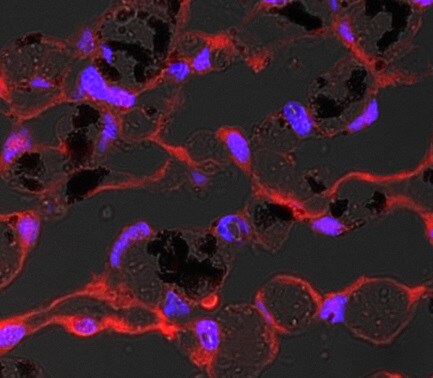
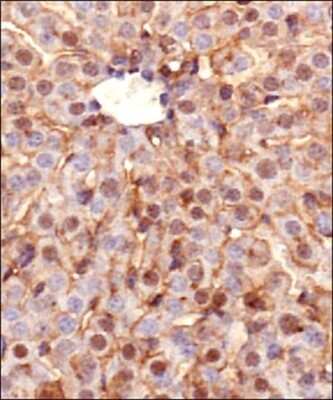
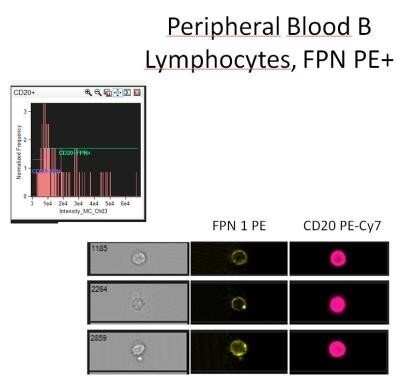
Detection of Ferroportin/SLC40A1 protein in murine liver section using Ferroportin/SLC40A1 Antibody at a dilution of 1:200. The representative image shows intense staining in the cellular membranes, whereas, a relatively milder postivity was observed in the cytoplasm of hepatocytes.Flow Cytometry Analysis of B Lymphocytes Stained with Phycoerythrin Conjugated Ferroportin/SLC40A1 Antibody
Analysis using the PE conjugate of Ferroportin/SLC40A1 Antibody. Staining of Ferroportin in human B lymphocytes in peripheral blood using PE conjugated anti-Ferroportin/SLC40A1 antibody. Image from verified customer review.Flow Cytometry of HepG2 Cells Stained with Alexa Fluor 647 Conjugated Ferroportin/SLC40A1 Antibody
An intracellular stain was performed on HepG2 cells with NBP1-21502AF647 (blue) and a matched isotype control (orange, NBP2-24893AF647). Cells were fixed with 4% PFA and then permeablized with 0.1% saponin. Cells were incubated in an antibody dilution of 5 ug/mL for 30 minutes at room temperature. Both antibodies were conjugated to Alexa Fluor 647.Flow Cytometry of Hek293 Cells Stained with Ferroportin/SLC40A1 Antibody
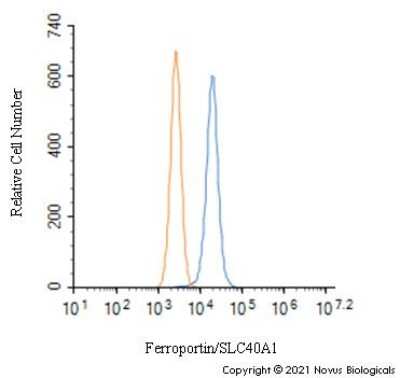
An intracellular stain was performed on Hek293 cells with NBP1-21502 (blue) and a matched isotype control NBP2-24891 (orange). Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 1.0 ug/mL for 30 minutes at room temperature, followed by Rabbit IgG (H+L) Cross-Adsorbed Secondary Antibody, Dylight 550 (SA5-10033, Thermo Fisher).Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
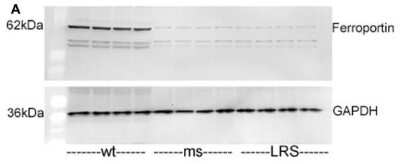
Mouse ferroportin, 5-aminolevulinic acid synthase (ALAS), and ferritin light chain (FLC) proteins are increased in sickle mice overexpressing human wt-FHC. Proteins of subcellular fractions isolated from livers of wt-, ms-, and LRS-treated mice (n = 4) were run on a western blot (30 ug protein/lane) and immunostained for (A) microsomal ferroportin.Immunohistochemistry-Paraffin: Rabbit Polyclonal Ferroportin/SLC40A1 Antibody [NBP1-21502] -
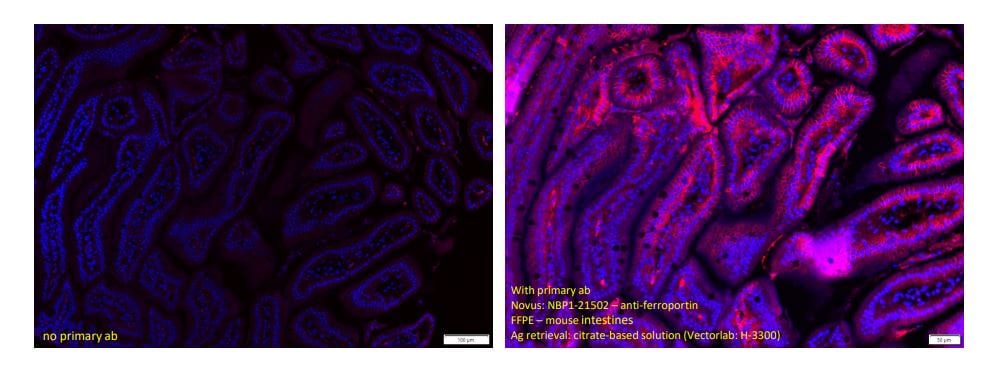
Immunohistochemistry-Paraffin: Rabbit Polyclonal Ferroportin/SLC40A1 Antibody [NBP1-21502] - Staining of mouse small intestines using Ferroportin/SLC40A1 Antibody. Tissue was fixed in formalin overnight and embedded in paraffin. ~5 micro sections on slides were dehydrated. Antigen retrieval was done using citrate buffer in rice steamer for 20mins. 1 hour blocking without permeabilization. Overnight primary ab incubation was done 1:200 (NBP1-21502) at 4C. 2-hr secondary antibody incubation was at room temp; mounting media with DAPI. Image from a verified customer review.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Analysis of Cu & Fe content & metal-binding protein expression in wild-type & PrPC-null mouse total brain at different ages. (A) The graph shows the ratio of Cu & Fe levels in Prnp0/0 & Prnp+/+ brain samples (P1, P365 N = 4; P7, P30, P180 N = 6; P90 N = 5). (B) Representative Western blot images showing metal-binding protein levels in Prnp0/0 & Prnp+/+ brain samples (N = 4). The constant level of the housekeeping proteins ( beta -III Tubulin & beta -Actin) are also reported. (C) The graph shows the up- or down-regulation of protein expression in Prnp0/0 samples compared to Prnp+/+, i.e., (Prnp0/0 protein OD/housekeeping OD)/(Prnp+/+ protein OD/housekeeping OD). All error bars indicate SD; N = 4 minimum; *p < 0.05; **p < 0.01; ***p < 0.001. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27729845), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Fisetin alleviates DOX-induced ferroptosis via the SIRT1/Nrf2-regulated signaling pathway. (A & B). Western blot results of SIRT1 protein levels after SIRT1 siRNA transfection. (C). Western blot of nuclear & cytosol Nrf2 levels in vitro after treatment with DOX and/or fisetin, SIRT1 siRNA. (D–E) nucleus or cytosol Nrf2 protein from Western blot results. (F). Protein expression levels of SIRT1, GPX4, Keap1, HO-1, FTH1, FTL, FPN, TfR1, & nuclear Nrf2 in H9c2 cells administrated with DOX and/or fisetin, SIRT1 siRNA. (G). Quantification of SIRT1, GPX4, Keap1, HO-1, FTH1, FTL, FPN, & TfR1 protein from Western blot results (n = 4). The values are presented as mean ± SD. *p < 0.05; **p < 0.01; ***p < 0.001; NS, no significance. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/35273493), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
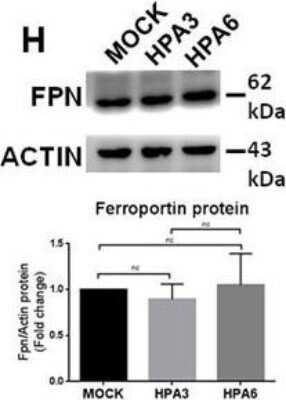
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Transgenic mice overexpressing heparanase have increased ferritin-iron & ferritin protein content in the liver.(A & B) Western blot of liver extracts from WT & TG-HPA mice (A) for ferritin L-chain (FTL) subunits in SDS-PAGE with GAPDH as calibrator & (B) for assembled ferritin in non-denaturing PAGE. (C) Prussian blue stain of non-denaturing PAGE loaded with 50 ug protein, before (upper) & after enhancing with DAB & H2O2 (lower). rFTL is control purified recombinant mouse FTL. (D) Western blot of Ferroportin (FPN) & (E) of Transferrin Receptor1 (TfR1) & their respective GAPDH as calibrator. Densitometry data were obtained from 3 independent experiments. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27711215), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Sodium ascorbate (150 μM) was added to the apical and/or basal medium of Caco-2 cells cultured on permeable Transwell® inserts. Ferroportin protein expression was measured with ELISA. Data are means ± SD, n = 12. Significant differences from the baseline (0 μM) are labeled with an asterisk (*). A western blot of ferroportin at the corresponding treatments is shown above the graph. Image collected & cropped by CiteAb from the following publication (http://www.mdpi.com/2072-6643/6/1/249), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - PPAR delta knockdown abrogated the effects of GW501516 on 6-OHDA-triggered protein levels of DMT1, FPN1, & Ferritin, & mRNA expression of DMT1. Cells stably expressing shRNA targeting scrambled sequences or PPAR delta were pretreated with DMSO or GW501516 for 8 h, & subsequently incubated with or without 6-OHDA for 16 h. Total RNA & protein were extracted, & levels of mRNA & protein were analyzed by real-time PCR (A–E) & Western blot (F). RPS18 & alpha -tubulin were used as internal controls for real-time PCR & Western blot, respectively. Results are expressed as means of triplicate ± SE (A–D). * p < 0.05, ** p < 0.01 relative to untreated group; #p < 0.05 relative to 6-OHDA-treated group. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/35624674), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
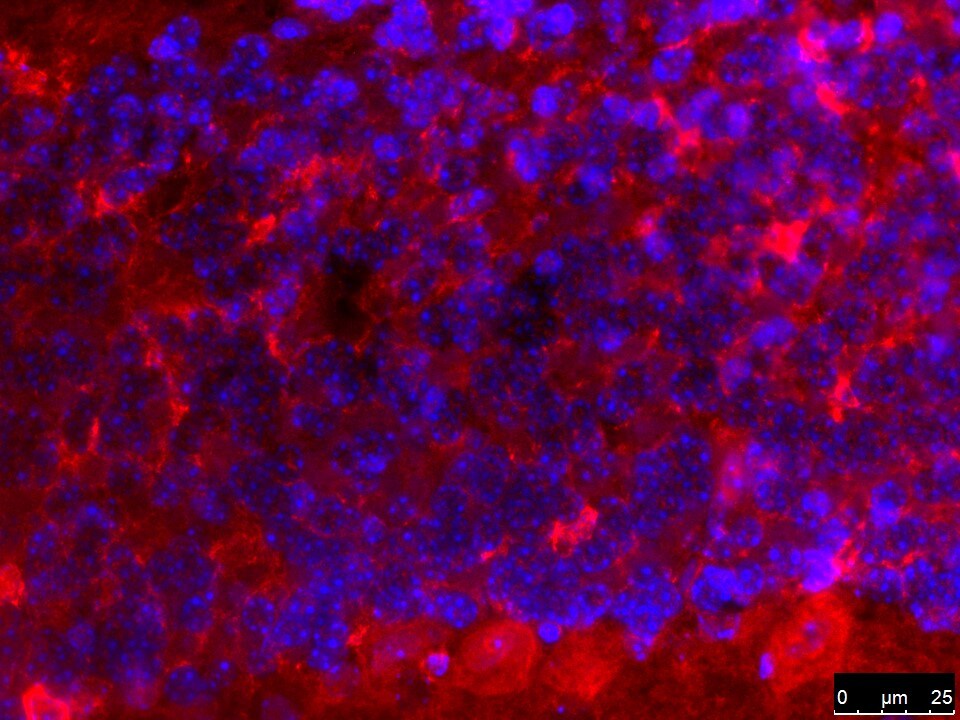
Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - The cellular localization of SIRT2 & FPN1 in the spinal cord. (A) SIRT2 levels decreased in the microglia of SNI rats. (B,C) FPN1 levels decreased in the microglia & neurons of SNI rats. The image in the first column on the left shows the overall outline of the spinal cord. Representative confocal images show the results of double immunofluorescence staining of SIRT2 or FPN1 (red) in the spinal dorsal horn; CD11b, a microglia marker (green) or NeuN, a neuronal marker (green) in the sham group & SNI group. Scale bars, 100 μm. SNI, spared nerve injury; SIRT2, sirtuin 2; FPN1, ferroportin 1. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/35401208), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Inhibition of GLRX5 upregulates an iron-starvation response. (A-D) Protein & mRNA expression of (TfR1, TFRC), iron regulatory protein (IRP2, IREB2), ferritin (FTH1) & ferroportin (Fpn, SLC40A1) genes in HN4R cells stably transduced with a vector or shGLRX5 & GLRX5res, which were subjected to 1 mM SAS treatment for 24 h. (E-H) Aconitase & alpha -ketoglutarate dehydrogenase ( alpha KGDH) activities, immunoblots, & cell death of HN4R cells stably transduced with a vector control or shGLRX5 & resistant GLRX5 cDNA (GLRX5res) wild type (WT) or catalytically inactive mutant cDNA (K101Q). The error bars represent standard errors from three replicates. ** P < 0.01 between the different groups treated with SAS. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/32685019), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - The cellular localization of SIRT2 & FPN1 in the spinal cord. (A) SIRT2 levels decreased in the microglia of SNI rats. (B,C) FPN1 levels decreased in the microglia & neurons of SNI rats. The image in the first column on the left shows the overall outline of the spinal cord. Representative confocal images show the results of double immunofluorescence staining of SIRT2 or FPN1 (red) in the spinal dorsal horn; CD11b, a microglia marker (green) or NeuN, a neuronal marker (green) in the sham group & SNI group. Scale bars, 100 μm. SNI, spared nerve injury; SIRT2, sirtuin 2; FPN1, ferroportin 1. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/35401208), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Effects of HDAC3 inhibition in vivo on hepcidin expression in iron-deficient mice. Four-week-old C57Bl/6 mice were fed a low-iron diet (2 ppm) for 3 weeks, followed by two doses of RGFP966 2 h apart, & killed 2 h following the second dose. Effect on hepatic aCdkn1a, bHamp1, cId1, dAtoh8, eSmad7, & fBmp6 mRNA expression, g liver iron. h Western blot for hepatic ferroportin expression. i Effect of RGFP966 on spleen iron. N = 6 per group. Data are means ± s.e.m. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001; ****P ≤ 0.0001; NS, P > 0.05 Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28864822), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - SHP abrogates the BMP6 effect on iron metabolism through inhibition of hepcidin gene expression in mice.(a–d) C57/BL6 mice were injected with Ad-GFP (n = 4 per group, 5.9 × 109 pfu) or Ad-Flag-SHP (n = 5 per group, 5.9 × 109 pfu) via the tail-vein, & treated with Vehicle or BMP6 (500 μg/kg, i.p.) for 6 h at day 5 after the infection. (a) Serum iron level. (b) Q-PCR analysis showing hepcidin & SHP mRNA levels in liver. (c) Serum hepcidin levels. (d) Western blot analysis showing SMAD1/5/8 phosphorylation & SHP expression in liver. (e) Western blot analysis showing FPN expression in spleen (top). Graphical representation showing FPN expression (bottom). Data are presented as means ± SD. Arrows show locations of molecular weight markers. The western blot images were cropped with a grey cropping line. All gels for western blot analysis were run under the same experimental conditions. **P < 0.01, ***P < 0.001 by two-tailed Student t-test. Image collected & cropped by CiteAb from the following publication (https://www.nature.com/articles/srep34630), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Upregulation of H3K4me3 increases iron exporter Fpn1 & exhibits neuroprotection.a, b CPI-455 treatment (24 h) increased the protein level of H3K4me3 & Fpn1 in SH-SY5Y cells. c MTT assay showed CPI-455 showed neuroprotection against the toxicity of 6-OHDA in SH-SY5Y cells. d–g SH-SY5Y cells were infected by AAV-KDM5C-silencing virus, & KDM5C was significantly decreased at the mRNA level as reflected by RT-PCR d & at the protein level by Western blot e, f. In addition, Fpn1 & H3K4me3 were increased in AAV-KDM5C-silencing virus infected cells e, f. Moreover, g silencing KDM5C showed a slight neuroprotection against 6-OHDA-induced PD model in SH-SY5Y cells. *P < 0.05; **P < 0.01; ***P < 0.001. Data are presented as mean ± SEM. n = 3–4 for each group in Western blot & n = 5 for each group in MTT assay. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/33116116), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Upregulation of H3K4me3 increases iron exporter Fpn1 & exhibits neuroprotection.a, b CPI-455 treatment (24 h) increased the protein level of H3K4me3 & Fpn1 in SH-SY5Y cells. c MTT assay showed CPI-455 showed neuroprotection against the toxicity of 6-OHDA in SH-SY5Y cells. d–g SH-SY5Y cells were infected by AAV-KDM5C-silencing virus, & KDM5C was significantly decreased at the mRNA level as reflected by RT-PCR d & at the protein level by Western blot e, f. In addition, Fpn1 & H3K4me3 were increased in AAV-KDM5C-silencing virus infected cells e, f. Moreover, g silencing KDM5C showed a slight neuroprotection against 6-OHDA-induced PD model in SH-SY5Y cells. *P < 0.05; **P < 0.01; ***P < 0.001. Data are presented as mean ± SEM. n = 3–4 for each group in Western blot & n = 5 for each group in MTT assay. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/33116116), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - SHP deficiency alters hepcidin gene expression in liver of HID mice.(a–i) WT & SHP KO mice (n = 5 per group) were fed with high-iron diet (HID, 8 g/kg) for 3 weeks. (a) Serum iron level. (b) Hepcidin mRNA level in liver. (c) Hepcidin expression in mouse liver. IHC was performed using an antibody against hepcidin. Scale bar shows 50 μm. (d) Serum hepcidin level. (e) Western blot analysis (left panel) showing hepatic SHP & splenic FPN expression & graphical representation (right panel) showing splenic FPN expression. (f) Splenic iron level. (g) Perls’ prussian blue staining in spleen. Scale bar shows 200 μm. (h) BMP6 & BMP9 mRNA levels in liver. (i) Western blot analysis (top) & graphical representation (bottom) showing SMAD1/5/8 phosphorylation in liver. The grouping of the images is from different parts of the same gel. Data are presented as means ± SD. Arrows show locations of molecular weight markers. The experiment was repeated on a minimum of three separate occasions. The western blot images were cropped with a grey cropping line. All gels for western blot analysis were run under the same experimental conditions. *P < 0.05, **P < 0.01, ***P < 0.001 by two-tailed Student t-test. Image collected & cropped by CiteAb from the following publication (https://www.nature.com/articles/srep34630), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Differential expression of proteins of iron metabolism in CCA cells. The CCA4, CCLP1 & HUCCT1 human CCA-derived cell lines were cultured as adherent monolayers (MON) or in 3D sphere conditions (SPH). Panel a. Top left, representative immunoblot analysis. Cell extracts were reacted with antibodies against transferrin receptor (TfR1), ferroportin (FPN), ferritin H subunit (FtH) & vinculin. Cropped blots are displayed. The original full blot images can be found in Supplementary Information. The graphs show densitometric quantification of immunoblot analyses. The values were normalized to vinculin & expressed as a fraction of respective MON cells normalized to 1. Mean values ± SEM (n = 6), *p ≤ 0.05, **p ≤ 0.01 vs control MON for each cell line. Panel b. RNA bandshift analysis of IRP activity. Cytoplasmic extracts were incubated with a 32P-labeled iron-responsive element (IRE) probe & RNA-protein complexes separated on non-denaturing polyacrylamide gels. On the left a representative autoradiogram is shown. A cropped gel is displayed. The original full gel image can be found in Supplementary Information. The graph on the right shows the densitometric quantification of IRPs bands by direct nuclear counting, as described in Materials & Methods; mean percentages ± SEM of control values (n = 6),, **p ≤ 0.01, ***p< 0.001 vs control MON for each cell line. Panel c. TfR1 & FPN mRNA levels were measured by quantitative RT-PCR. Samples were analyzed in triplicate, normalized to the housekeeping gene 18 S & expressed as percentage of respective MON cells normalized to 1. Mean values ± SEM (n = 6), **p ≤ 0.01 vs control MON for each cell line. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29247214), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Iron (55Fe) transport across the apical & basal borders. (a) Apical transport (uptake) of 55Fe in response to intracellular/basal ascorbate (150 μM) was increased compared to no ascorbate. The cells were incubated with ascorbate in the basal chamber for 24 h. Values are means of 10 samples ±SD. The difference between treatments was significant (p = 0.04). (b) Basolateral transport of iron (as 55Fe) in response to intracellular/basal ascorbate (150 μM) was increased compared to no ascorbate. The cells were incubated with ascorbate in the basal chamber for 24 h. Values are means of 10 samples ±SD. The difference between treatments was significant (p = 0.03). (c) Western Blot of cells treated with ascorbate (150 μM). Lane 1: Control cells (no treatment). Lane 2: Basal ascorbate at 150 μM; these were the ferroportin levels before iron addition in (a). Lane 3: Ferroportin levels 22 h after the iron addition (20 μM for 2 h) in (a). Image collected & cropped by CiteAb from the following publication (http://www.mdpi.com/2072-6643/6/1/249), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Metformin rescues BMP6-mediated alteration of iron metabolism in mice.(a–e) C57/BL6 mice (n = 4 per group) were treated with BMP6 (500 μg/kg, i.p.) & metformin (200 mg/kg, p.o.). (a) Serum iron level. (b) Q-PCR analysis showing hepcidin & SHP mRNA levels in liver. (c) Serum hepcidin levels. (d) Western blot analysis showing SMAD1/5/8 phosphorylation & FPN in liver. (e) Western blot analysis (top) & graphical representation (bottom) showing FPN expression in spleen. (f) Schematic diagram of SHP-mediated inhibition of BMP6-SMADs pathway. Data are presented as means ± SD. Arrows show locations of molecular weight markers. The western blot images were cropped with a grey cropping line. All gels for western blot analysis were run under the same experimental conditions. *P < 0.05, **P < 0.01, ***P < 0.001 by two-tailed Student t-test. Image collected & cropped by CiteAb from the following publication (https://www.nature.com/articles/srep34630), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Comparison of Cu, Fe, & metal-binding protein expression levels in wild-type & PrPC-null mouse spleen at different ages. (A) The graph shows the ratio of Cu & Fe levels in Prnp0/0 & Prnp+/+ spleen samples (P15 N = 3; P30 N = 4; P90, P180 N = 6; P365 N = 5). (B) The graph shows the weight of spleen extracted from Prnp0/0 & Prnp+/+ mice; N = 4. (C) Representative Western blot images showing metal-binding protein levels in Prnp0/0 & Prnp+/+ spleen samples. The constant level of the housekeeping protein ( beta -Actin) are also reported. (D) The graph shows the up- or down-regulation of protein expression in Prnp0/0 samples compared to Prnp+/+, i.e., (Prnp0/0 protein OD/housekeeping OD)/ (Prnp+/+ protein OD/housekeeping OD); N = 4. All error bars indicate SD; *p < 0.05; **p < 0.01; ***p < 0.001. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27729845), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Htt elimination disrupts brain iron homeostasis.(A) Representative photographs of immunohistochemical staining for ferritin light chain (Ft). Left panels: cerebella of 21mo CTL noTM & 18mo cKO TM@3mo; middle panels: thalami of 12mo CTL TM@9mo & 12mo cKO TM@9mo; right panels: striata of 21mo CTL noTM & 18mo cKO TM@3mo. Note that Ft levels are already extremely reduced in the thalamus 3 months after Htt elimination (middle panels), & that Ft expression is practically abolished in the cerebellum in the absence of Htt (left panels). (B) Representative western blots of Tfr & Fpn protein expression in 10 month-old brains from CTL & cKO mice TM-treated at 6 months of age. Antibody against actin was used as internal control for loading. Note that Tfr & Fpn levels are increased in TM-treated cKO compared to controls. (C) Quantification of Tfr & Fpn expression levels. Western blots of total protein extracts from 10 month-old brains CTL (n = 5) & cKO (n = 5) TM-treated at 6 months of age were probed with anti-Tfr or anti-Fpn, stripped & re-probed with anti-actin antibody. Bands intensities were quantitated using Image J. Tfr & Fpn levels were normalized over actin levels. Values represent mean relative to controls ± SD (***P<0.001, Student’s t-test). Image collected & cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pgen.1006846), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - GW501516 activation of PPAR delta regulates 6-OHDA-triggered expression of level of DMT1 & FPN1 protein, & DMT1 mRNA, but not FPN1 mRNA. (A) Cells were treated with 6-OHDA for the indicated durations. (B,C) Cells pretreated with DMSO or GW501516 for 8 h were incubated with or without 6-ODHA for 16 h. (D) Cells were treated with GW501516 for the indicated durations. (E,F) Cells pretreated with DMSO or GW501516 for 8 h were incubated with or without 6-ODHA for 16 h. Total RNA & protein were extracted, & mRNA & protein levels were analyzed by real-time PCR (A,B,D,E) & Western blot (C,F), respectively. RPS18 & alpha -tubulin were used as internal controls for real-time PCR & Western blot, respectively. Results are expressed as means ± SE (n = 3). An image analyzer was used to quantify band intensity of Western blot, & the ratio of protein to alpha -tubulin is indicated above each lane. * p < 0.05, ** p < 0.01 relative to the untreated group; #p < 0.05, ##p < 0.01 relative to the 6-OHDA-treated group. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/35624674), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Fisetin protects against DOX-induced ferroptosis via regulating Nrf2 in rats. (A). Western blot results of Nrf2 & Keap1, HO-1, FTH1, FTL, FPN, & TfR1 protein in control & different drug-treated rat heart tissues. (B). Quantification of (A). (C). Expression of Nrf2 was detected by immunohistochemistry (IHC) (Representative images, 200X & 400X, Scale bar = 100 & 50 μm, n = 6 rats per groups) in cardiac tissue of each group. The values are presented as mean ± SD. *p < 0.05; **p < 0.01; ***p < 0.001; NS, no significance; DOX, doxorubicin. Nrf2, nuclear factor erythroid 2-related factor 2; Keap1, Kelch-like ECH-associated protein 1; HO-1, heme oxygenase-1; FTH1, ferritin heavy chain 1; FTL, ferritin light chain; FPN, ferroportin; TfR1, transferrin receptor 1. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/35273493), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
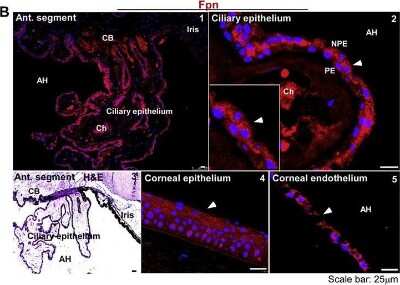
Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Distribution of TfR & Fpn in the anterior segment of bovine eye:(A) The reaction for TfR is evident on the ciliary epithelium facing the AH (panel 1). TfR is localized on the apical membrane & intracellular vesicles of NPE cells as in the human sample (panel 2). The corneal epithelium does not react for TfR (panel 3), while the corneal endothelium shows a distinct reaction on the plasma membrane (panel 4). H&E staining shows multiple layers of corneal epithelium, the stroma, & a single layer of corneal endothelium (panel 5). Scale bar: 25 μm. (B) Fpn is expressed on the ciliary epithelium & the iris (panel 1). The expression of Fpn is limited to NPE cells as in the human sample (panel 2). H&E staining shows the orientation of ciliary epithelium with respect to the iris & AH (panel 3). Unlike human samples, the bovine corneal epithelium did not react for Fpn (panel 4). However, corneal endothelium showed strong reactivity for Fpn (panel 5). Scale bar: 25 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29859760), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - GW501516 PPAR delta activation suppresses 6-OHDA-induced expression of IRP1 in SH-SY5Y cells. (A,B) Cells were treated with 6-OHDA (A) or GW501516 (B) for the indicated durations. (C) Cells stably expressing shRNA targeting scrambled sequences or PPAR delta were pretreated with DMSO or GW501516 for 8 h & subsequently incubated in the presence or absence of 6-ODHA for 16 h. (D,E) Cells pretreated with DMSO or GW501516 for 8 h were incubated with or without 6-ODHA for 16 h. Total RNA & protein were extracted, & levels of mRNA & protein were analyzed by real-time PCR (A–D) & Western blot (E). The immunoblots are separate from those shown in Figure 4C,F. RPS18 & alpha -tubulin were used as internal controls for real-time PCR & Western blot, respectively. Results are expressed as means of triplicate ± SE (A–D). * p < 0.05, ** p < 0.01 relative to untreated group; #p < 0.05 relative to 6-OHDA-treated group. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/35624674), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] -
Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Expression of iron modulating proteins in the cornea & ciliary body.(A) Probing of lysates from bovine cornea (Cor) & CB for Cp, TfR, Tf, & ferritin shows the expression of all of these proteins in both samples (lanes 1–4). Deglycosylation results in faster migration of Cp, TfR, & Tf on SDS-PAGE, indicating the presence of glycans (lanes 2 & 4). AH & the vitreous show abundant presence of Cp & Tf, both of which migrate faster upon deglycosylation (lanes 7 & 9). No reactivity for TfR or ferritin is detected in these samples (lanes 6–9). Human brain lysate was processed in parallel as a positive control (lane 5). Gapdh served as a loading control. (Cor: cornea; Ft: ferritin). (B) Relative distribution of iron modulating proteins within each tissue shows higher expression of TfR relative to Cp & ferritin in the cornea, & higher levels of Cp relative to the TfR & ferritin in the CB. (C) Quantitative comparison of protein expression by densitometry shows significantly higher levels of ferritin & Cp, & lower levels of TfR in the CB relative to the cornea. All values were normalized to Gapdh that provided the loading control. Values represent fold change ± SEM of the indicated n. (D) Probing of Western blots of bovine cornea & CB for Fpn revealed increased expression of Fpn (3.2 fold) in CB relative to the cornea (lanes 1–4). Lysates from human brain & bovine retina were analyzed in parallel as controls (lanes 5 & 6). Gapdh served as a loading control. (E) Quantification by densitometry shows 3.2 fold higher levels of Fpn in the CB relative to the cornea. Values are mean + SEM of the indicated n. **p < 0.01. The full images of the cropped blots have been provided in the Supplementary Data. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29859760), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for Ferroportin/SLC40A1 Antibody - BSA Free
Flow Cytometry
Immunohistochemistry
Immunohistochemistry-Paraffin
Western Blot
Reviewed Applications
Read 8 reviews rated 4.6 using NBP1-21502 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: Ferroportin/SLC40A1
FPN1 regulation is dependent on the cell type and involves transcriptional, posttranscriptional, and posttranslational mechanisms including hepcidin-mediated endocytosis and proteolysis. Hepcidin controls the concentration of FPN1 in the membrane, with hepcidin deficiency resulting in iron overload (high iron) and hepcidin excess leading to iron restriction and anemia (2). Ferroportin disease or hemochromatosis type 4 (HFE4) is associated with distinct FPN1 variants with either reduced FPN1 cell surface expression/iron export capacity or hepcidin resistance and iron overload (3, 4).
References
1. De Domenico I, Ward DM, Kaplan J. (2011) Hepcidin and ferroportin: the new players in iron metabolism. Semin Liver Dis. 31(3):272-9. PMID: 21901657
2. Drakesmith H, Nemeth E, Ganz T. (2015) Ironing out Ferroportin. Cell Metab. 22(5):777-87. PMID: 26437604
3. Pietrangelo A. (2017) Ferroportin disease: pathogenesis, diagnosis and treatment. Haematologica. 102(12):1972-1984. PMID: 29101207
4. Vlasveld LT, Janssen R, Bardou-Jacquet E, Venselaar H, Hamdi-Roze H, Drakesmith H, Swinkels DW. (2019) Twenty Years of Ferroportin Disease: A Review or An Update of Published Clinical, Biochemical, Molecular, and Functional Features. Pharmaceuticals (Basel). 12(3). pii: E132. PMID: 31505869
Long Name
Alternate Names
Gene Symbol
UniProt
Additional Ferroportin/SLC40A1 Products
Product Documents for Ferroportin/SLC40A1 Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Ferroportin/SLC40A1 Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for Ferroportin/SLC40A1 Antibody - BSA Free
Customer Reviews for Ferroportin/SLC40A1 Antibody - BSA Free (8)
Have you used Ferroportin/SLC40A1 Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Immunohistochemistry-ParaffinSample Tested: mouse small intestine and small intestinesSpecies: MouseVerified Customer | Posted 05/09/2024IF on FFPE mouse small intestines - worked well.Tissue fixed in formalin overnight; embedded in paraffin; ~5micro sections on slides; dehydrated; antigen retrieval using citrate buffer in rice steamer for 20mins; 1 hour blocking without permeabilization; overnight primary ab incubation @ 1:200 (NBP1-21502) at 4c; 2-hr secondary ab incubation at room temp; mounting media with DAPI.
-
Application: Western BlotSample Tested: Adult pancreasSpecies: MouseVerified Customer | Posted 08/08/2018
-
Application: Western BlotSample Tested: duodenal mucosaSpecies: PigVerified Customer | Posted 07/12/2018Duodenal mucosa samples collected from weanling pigs with iron replete or iron deficient status were probed for FPN1 (NBP1-21502; 1:1000 dilution) and GAPDH (loading control) following routine western blot procedures.
-
Application: Immunohistochemistry-ParaffinSample Tested: Adult eye and muscle tissue (labeled E and M, respectively)Species: HumanVerified Customer | Posted 04/04/20181:100
-
Application: Western BlotSample Tested: Adult eyeSpecies: BovineVerified Customer | Posted 04/04/20181:1000
-
Application: Immunohistochemistry-ParaffinSample Tested: eyeSpecies: BovineVerified Customer | Posted 10/24/2017excellent !!!use at 1:100
-
Application: ImmunohistochemistrySample Tested: Mouse brainSpecies: MouseVerified Customer | Posted 06/27/2017
-
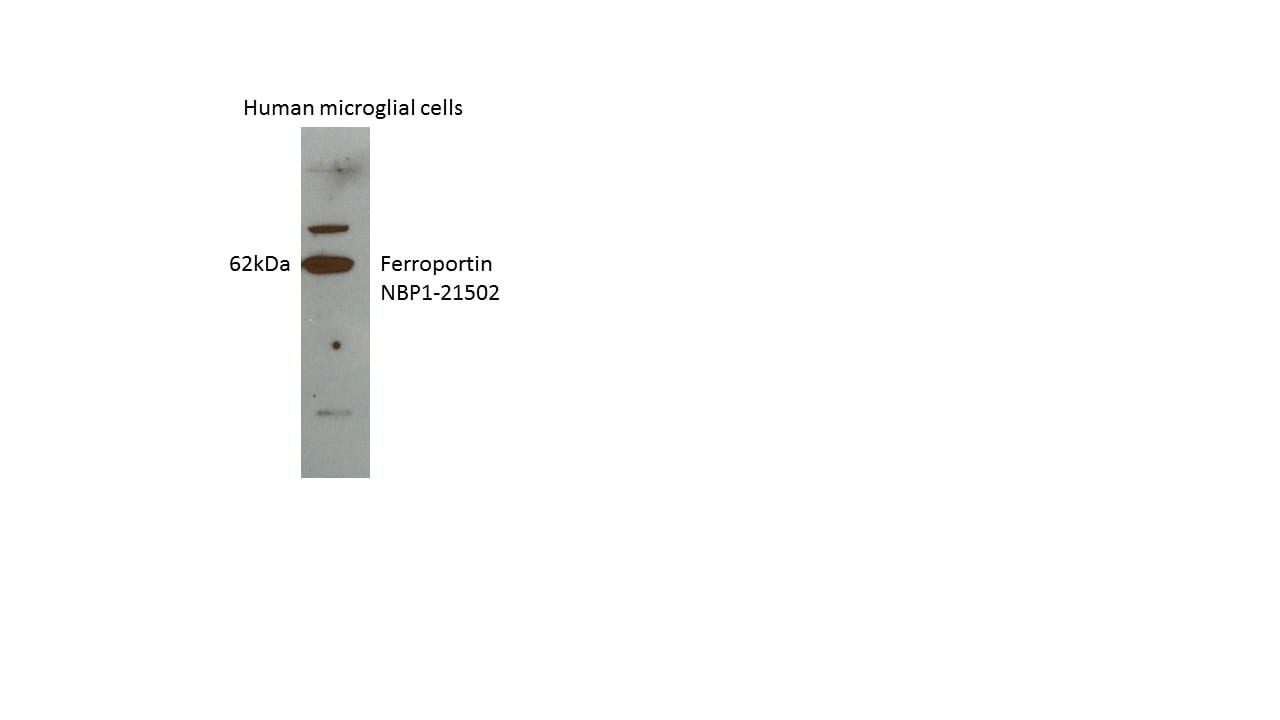
Application: Western BlotSample Tested: Human microglia cell lysateSpecies: HumanVerified Customer | Posted 06/13/2017
There are no reviews that match your criteria.
Protocols
View specific protocols for Ferroportin/SLC40A1 Antibody - BSA Free (NBP1-21502):
Sample Preparation.
1. Grow cells to 60-85% confluency. Flow cytometry requires between 2 x 105 and 1 x 106 cells for optimal performance.
2. If cells are adherent, harvest gently by washing once with staining buffer and then scraping. Avoid using trypsin as this can disrupt certain epitopes of interest. If enzymatic harvest is required, use Accutase, Collagenase, or TrypLE Express for a less damaging option.
3. Reserve 100 uL for counting, then transfer cell volume into a 50 mL conical tube and centrifuge for 8 minutes at 400 RCF.
a. Count cells using a hemocytometer and a 1:1 trypan blue exclusion stain to determine cell viability before starting the flow protocol. If cells appear blue, do not proceed.
4. Re-suspend cells to a concentration of 1 x 106 cells/mL in staining buffer (NBP2-26247).
5. Aliquot out 100 uL samples in accordance with your experimental samples.
Tip: When cell surface and intracellular staining are required in the same sample, it is advisable that the cell surface staining be performed first since the fixation and permeabilization steps might reduce the availability of surface antigens.
Intracellular Staining.
Tip: When performing intracellular staining, it is important to use appropriate fixation and permeabilization reagents based upon the target and its subcellular location. Generally, our Intracellular Flow Assay Kit (NBP2-29450) is a good place to start as it contains an optimized combination of reagents for intracellular staining as well as an inhibitor of intracellular protein transport (necessary if staining secreted proteins). Certain targets may require more gentle or transient permeabilization protocols such as the commonly employed methanol or saponin-based methods.
Protocol for Cytoplasmic Targets:
1. Fix the cells by adding 100 uL fixation solution (such as 4% PFA) to each sample for 10-15 minutes.
2. Permeabilize cells by adding 100 uL of a permeabilization buffer to every 1 x 106 cells present in the sample. Mix well and incubate at room temperature for 15 minutes.
a. For cytoplasmic targets, use a gentle permeabilization solution such as 1X PBS + 0.5% Saponin or 1X PBS + 0.5% Tween-20.
b. To maintain the permeabilized state throughout your experiment, use staining buffer + 0.1% of the permeabilization reagent (i.e. 0.1% Tween-20 or 0.1% Saponin).
3. Following the 15 minute incubation, add 2 mL of the staining buffer + 0.1% permeabilizer to each sample.
4. Centrifuge for 1 minute at 400 RCF.
5. Discard supernatant and re-suspend in 100 uL of staining buffer + 0.1% permeabilizer.
6. Add appropriate amount of each antibody (eg. 1 test or 1 ug per sample, as experimentally determined).
7. Mix well and incubate at room temperature for 30 minutes- 1 hour. Gently mix samples every 10-15 minutes.
8. Following the primary/conjugate incubation, add 1-2 mL/sample of staining buffer +0.1% permeabilizer and centrifuge for 1 minute at 400 RCF.
9. Wash twice by re-suspending cells in staining buffer (2 mL for tubes or 200 uL for wells) and centrifuging at 400 RCF for 5 minutes. Discard supernatant.
10. Add appropriate amount of secondary antibody (as experimentally determined) to each sample.
11. Incubate at room temperature in dark for 20 minutes.
12. Add 1-2 mL of staining buffer and centrifuge at 400 RCF for 1 minute and discard supernatant.
13. Wash twice by re-suspending cells in staining buffer (2 mL for tubes or 200 uL for wells) and centrifuging at 400 RCF for 5 minutes. Discard supernatant.
14. Resuspend in an appropriate volume of staining buffer (usually 500 uL per sample) and proceed with analysis on your flow cytometer.
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and wash the cells briefly in PBS. Add 10% formalin to the dish and fix at room temperature for 10 minutes.
2. Remove the formalin and wash the cells in PBS.
3. Permeablize the cells with 0.1% Triton X100 or other suitable detergent for 10 min.
4. Remove the permeablization buffer and wash three times for 10 minutes each in PBS. Be sure to not let the specimen dry out.
5. To block nonspecific antibody binding, incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
6. Add primary antibody at appropriate dilution and incubate overnight at 4C.
7. Remove primary antibody and replace with PBS. Wash three times for 10 minutes each.
8. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
9. Remove secondary antibody and replace with PBS. Wash three times for 10 minutes each.
10. Counter stain DNA with DAPi if required.
Antigen Unmasking:
Bring slides to a boil in 10 mM sodium citrate buffer (pH 6.0) then maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench-top for 30 minutes (keep slides in the sodium citrate buffer at all times).
Staining:
1. Wash sections in deionized water three times for 5 minutes each.
2. Wash sections in PBS for 5 minutes.
3. Block each section with 100-400 ul blocking solution (1% BSA in PBS) for 1 hour at room temperature.
4. Remove blocking solution and add 100-400 ul diluted primary antibody. Incubate overnight at 4 C.
5. Remove antibody solution and wash sections in wash buffer three times for 5 minutes each.
6. Add 100-400 ul HRP polymer conjugated secondary antibody. Incubate 30 minutes at room temperature.
7. Wash sections three times in wash buffer for 5 minutes each.
8. Add 100-400 ul DAB substrate to each section and monitor staining closely.
9. As soon as the sections develop, immerse slides in deionized water.
10. Counterstain sections in hematoxylin.
11. Wash sections in deionized water two times for 5 minutes each.
12. Dehydrate sections.
13. Mount coverslips.
1. Perform SDS-PAGE on samples to be analyzed, loading 10-25 ug of total protein per lane.
2. Transfer proteins to PVDF membrane according to the instructions provided by the manufacturer of the membrane and transfer apparatus.
3. Stain the membrane with Ponceau S (or similar product) to assess transfer success, and mark molecular weight standards where appropriate.
4. Rinse the blot TBS -0.05% Tween 20 (TBST).
5. Block the membrane in 5% Non-fat milk in TBST (blocking buffer) for at least 1 hour.
6. Wash the membrane in TBST three times for 10 minutes each.
7. Dilute primary antibody in blocking buffer and incubate overnight at 4C with gentle rocking.
8. Wash the membrane in TBST three times for 10 minutes each.
9. Incubate the membrane in diluted HRP conjugated secondary antibody in blocking buffer (as per manufacturer's instructions) for 1 hour at room temperature.
10. Wash the blot in TBST three times for 10 minutes each (this step can be repeated as required to reduce background).
11. Apply the detection reagent of choice in accordance with the manufacturer's instructions.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for Ferroportin/SLC40A1 Antibody - BSA Free
-
Q: I can’t get positive a result in IHC-P using NBP1-21502 (tissue: atherosclerosis hemorrhage plaque). PV-6001 and DAB is working. Can you make optimization suggestions on how to get this antibody to work?
A:
Optimization could really make a difference as we have seen some staining with the same antibody on frozen section. The main thing would be to focus on antigen retrieval step and the buffer used. For the antigen retrieval method, we would suggest an increase to at least 10-20 minutes to the pressure cooker step of the HIER method; the duration of the retrieval step can greatly determine if the antigen will be exposed or not for the antigen binding site of the antibody to recognize the protein. Also, I the incubation time for peroxidase blocking time could be reduced from 30min to 15 min, as we have mentioned. Since this is a TMP, permeabilization reagents should be used for the experiment. You can review what we have available on our site in terms of our general protocols:
HIER and IHC-P
-
Q: I have some leftovers of this antibody from two years ago and tried it for immunofluorescence on mouse, which did work well. I may need more of this antibody. Some things surprise me. In the old datasheet it says that there is cross reactivity with mouse. It does not say so in the new data sheet here on the web site. However, it does say so for all the conjugated versions of the Ab. What is correct? Also, in the old data sheet it says that it is for Western blotting. Now immunofluorescence is also mentioned. Is this still the same antibody? I don't have a lot number, but we got it in September 2010.
A: NBP1-21502 is still the same antibody it was two years ago. Mouse was mistakenly removed during a website update and has since been re-added. This antibody still cross-reacts with mouse. In the past two years we have begun to validate more of our products for use in ICC and this is one that has had its uses updated since you last purchased.
-
Q: Is the epitope for NBP1-21502 extracellular or will it require permeabilization? Is it possible to get a sample size of one of the conjugated versions?
A: The immunogen used for this Ferroportin/SLC40A1 Antibody NBP1-21502 falls within a helical domain of the protein and some permeabilization is recommended for staining. Our lab uses 0.2% Tween-20 included in the blocking and diluent buffers to perform this permeabilization. As our directly conjugated antibodies are prepared per order, we unfortunately do not have sample sizes available.
-
Q: We are having difficulty producing a clean blot, i.e. when we probe our western blots with the recommended concentration of 1.0 ug/ml, we are seeing multiple bands. The order of the molecular weight ladder we used is as followed: 250, 130, 100, 70, 55, 35, 25, 15, and 10 kDa. Are you able to indicate what band may be ferroportin.?
A:
There is a western blot protocol specific to this antibody that can be found on the datasheet; it differs from your protocol quite a lot. I would give this protocol a try and see if you get better results.
Although I am uncertain of your blocking method, we find that BSA and PVDF can give high non-specific background so the nitrocellulose and NFDM are recommend for this antibody. In addition, there seems to be no protein expression of the SLC40A1 in liver or spleen based on Human Protein Atlas.
https://www.proteinatlas.org/ENSG00000138449-SLC40A1/tissue
A good positive control might be bone marrow.
-
Q: Would it be possible to conjugate this antibody to PE or PerCP?
A:
With regards to conjugating NBP1-21502SS (0.025 ml) to PE or PerCP, there are two possibilities: The first is that you could conjugate the antibody yourself, using easy-to-use kits. You would choose from Lightning-Link R-PE 3 x 0.01 mg, or Lightning-Link PerCP 3 x 0.01 mg. Because the buffer of the NBP1-21502 antibody is Tris-glycine, which is not compatible with the Lightning-Link conjugation chemistry as that targets amine groups, you would perform a buffer exchange first using the Antibody Concentration and Clean Up Kit. The lab has informed me that yes, they could conjugate your choice of PE or PerCP to this antibody for you. However, it would have to be the full-size antibody vial (0.1 ml) rather than the sample size (0.025 ml).
-
Q: I can’t get positive a result in IHC-P using NBP1-21502 (tissue: atherosclerosis hemorrhage plaque). PV-6001 and DAB is working. Can you make optimization suggestions on how to get this antibody to work?
A:
Optimization could really make a difference as we have seen some staining with the same antibody on frozen section. The main thing would be to focus on antigen retrieval step and the buffer used. For the antigen retrieval method, we would suggest an increase to at least 10-20 minutes to the pressure cooker step of the HIER method; the duration of the retrieval step can greatly determine if the antigen will be exposed or not for the antigen binding site of the antibody to recognize the protein. Also, I the incubation time for peroxidase blocking time could be reduced from 30min to 15 min, as we have mentioned. Since this is a TMP, permeabilization reagents should be used for the experiment. You can review what we have available on our site in terms of our general protocols:
HIER and IHC-P
-
Q: I have some leftovers of this antibody from two years ago and tried it for immunofluorescence on mouse, which did work well. I may need more of this antibody. Some things surprise me. In the old datasheet it says that there is cross reactivity with mouse. It does not say so in the new data sheet here on the web site. However, it does say so for all the conjugated versions of the Ab. What is correct? Also, in the old data sheet it says that it is for Western blotting. Now immunofluorescence is also mentioned. Is this still the same antibody? I don't have a lot number, but we got it in September 2010.
A: NBP1-21502 is still the same antibody it was two years ago. Mouse was mistakenly removed during a website update and has since been re-added. This antibody still cross-reacts with mouse. In the past two years we have begun to validate more of our products for use in ICC and this is one that has had its uses updated since you last purchased.
-
Q: Is the epitope for NBP1-21502 extracellular or will it require permeabilization? Is it possible to get a sample size of one of the conjugated versions?
A: The immunogen used for this Ferroportin/SLC40A1 Antibody NBP1-21502 falls within a helical domain of the protein and some permeabilization is recommended for staining. Our lab uses 0.2% Tween-20 included in the blocking and diluent buffers to perform this permeabilization. As our directly conjugated antibodies are prepared per order, we unfortunately do not have sample sizes available.
-
Q: We are having difficulty producing a clean blot, i.e. when we probe our western blots with the recommended concentration of 1.0 ug/ml, we are seeing multiple bands. The order of the molecular weight ladder we used is as followed: 250, 130, 100, 70, 55, 35, 25, 15, and 10 kDa. Are you able to indicate what band may be ferroportin.?
A:
There is a western blot protocol specific to this antibody that can be found on the datasheet; it differs from your protocol quite a lot. I would give this protocol a try and see if you get better results.
Although I am uncertain of your blocking method, we find that BSA and PVDF can give high non-specific background so the nitrocellulose and NFDM are recommend for this antibody. In addition, there seems to be no protein expression of the SLC40A1 in liver or spleen based on Human Protein Atlas.
https://www.proteinatlas.org/ENSG00000138449-SLC40A1/tissue
A good positive control might be bone marrow.
-
Q: Would it be possible to conjugate this antibody to PE or PerCP?
A:
With regards to conjugating NBP1-21502SS (0.025 ml) to PE or PerCP, there are two possibilities: The first is that you could conjugate the antibody yourself, using easy-to-use kits. You would choose from Lightning-Link R-PE 3 x 0.01 mg, or Lightning-Link PerCP 3 x 0.01 mg. Because the buffer of the NBP1-21502 antibody is Tris-glycine, which is not compatible with the Lightning-Link conjugation chemistry as that targets amine groups, you would perform a buffer exchange first using the Antibody Concentration and Clean Up Kit. The lab has informed me that yes, they could conjugate your choice of PE or PerCP to this antibody for you. However, it would have to be the full-size antibody vial (0.1 ml) rather than the sample size (0.025 ml).
-
Q: I can’t get positive a result in IHC-P using NBP1-21502 (tissue: atherosclerosis hemorrhage plaque). PV-6001 and DAB is working. Can you make optimization suggestions on how to get this antibody to work?
A:
Optimization could really make a difference as we have seen some staining with the same antibody on frozen section. The main thing would be to focus on antigen retrieval step and the buffer used. For the antigen retrieval method, we would suggest an increase to at least 10-20 minutes to the pressure cooker step of the HIER method; the duration of the retrieval step can greatly determine if the antigen will be exposed or not for the antigen binding site of the antibody to recognize the protein. Also, I the incubation time for peroxidase blocking time could be reduced from 30min to 15 min, as we have mentioned. Since this is a TMP, permeabilization reagents should be used for the experiment. You can review what we have available on our site in terms of our general protocols:
HIER and IHC-P
-
Q: I have some leftovers of this antibody from two years ago and tried it for immunofluorescence on mouse, which did work well. I may need more of this antibody. Some things surprise me. In the old datasheet it says that there is cross reactivity with mouse. It does not say so in the new data sheet here on the web site. However, it does say so for all the conjugated versions of the Ab. What is correct? Also, in the old data sheet it says that it is for Western blotting. Now immunofluorescence is also mentioned. Is this still the same antibody? I don't have a lot number, but we got it in September 2010.
A: NBP1-21502 is still the same antibody it was two years ago. Mouse was mistakenly removed during a website update and has since been re-added. This antibody still cross-reacts with mouse. In the past two years we have begun to validate more of our products for use in ICC and this is one that has had its uses updated since you last purchased.
-
Q: Is the epitope for NBP1-21502 extracellular or will it require permeabilization? Is it possible to get a sample size of one of the conjugated versions?
A: The immunogen used for this Ferroportin/SLC40A1 Antibody NBP1-21502 falls within a helical domain of the protein and some permeabilization is recommended for staining. Our lab uses 0.2% Tween-20 included in the blocking and diluent buffers to perform this permeabilization. As our directly conjugated antibodies are prepared per order, we unfortunately do not have sample sizes available.
-
Q: We are having difficulty producing a clean blot, i.e. when we probe our western blots with the recommended concentration of 1.0 ug/ml, we are seeing multiple bands. The order of the molecular weight ladder we used is as followed: 250, 130, 100, 70, 55, 35, 25, 15, and 10 kDa. Are you able to indicate what band may be ferroportin.?
A:
There is a western blot protocol specific to this antibody that can be found on the datasheet; it differs from your protocol quite a lot. I would give this protocol a try and see if you get better results.
Although I am uncertain of your blocking method, we find that BSA and PVDF can give high non-specific background so the nitrocellulose and NFDM are recommend for this antibody. In addition, there seems to be no protein expression of the SLC40A1 in liver or spleen based on Human Protein Atlas.
https://www.proteinatlas.org/ENSG00000138449-SLC40A1/tissue
A good positive control might be bone marrow.
-
Q: Would it be possible to conjugate this antibody to PE or PerCP?
A:
With regards to conjugating NBP1-21502SS (0.025 ml) to PE or PerCP, there are two possibilities: The first is that you could conjugate the antibody yourself, using easy-to-use kits. You would choose from Lightning-Link R-PE 3 x 0.01 mg, or Lightning-Link PerCP 3 x 0.01 mg. Because the buffer of the NBP1-21502 antibody is Tris-glycine, which is not compatible with the Lightning-Link conjugation chemistry as that targets amine groups, you would perform a buffer exchange first using the Antibody Concentration and Clean Up Kit. The lab has informed me that yes, they could conjugate your choice of PE or PerCP to this antibody for you. However, it would have to be the full-size antibody vial (0.1 ml) rather than the sample size (0.025 ml).
-
Q: I can’t get positive a result in IHC-P using NBP1-21502 (tissue: atherosclerosis hemorrhage plaque). PV-6001 and DAB is working. Can you make optimization suggestions on how to get this antibody to work?
A:
Optimization could really make a difference as we have seen some staining with the same antibody on frozen section. The main thing would be to focus on antigen retrieval step and the buffer used. For the antigen retrieval method, we would suggest an increase to at least 10-20 minutes to the pressure cooker step of the HIER method; the duration of the retrieval step can greatly determine if the antigen will be exposed or not for the antigen binding site of the antibody to recognize the protein. Also, I the incubation time for peroxidase blocking time could be reduced from 30min to 15 min, as we have mentioned. Since this is a TMP, permeabilization reagents should be used for the experiment. You can review what we have available on our site in terms of our general protocols:
HIER and IHC-P
-
Q: I have some leftovers of this antibody from two years ago and tried it for immunofluorescence on mouse, which did work well. I may need more of this antibody. Some things surprise me. In the old datasheet it says that there is cross reactivity with mouse. It does not say so in the new data sheet here on the web site. However, it does say so for all the conjugated versions of the Ab. What is correct? Also, in the old data sheet it says that it is for Western blotting. Now immunofluorescence is also mentioned. Is this still the same antibody? I don't have a lot number, but we got it in September 2010.
A: NBP1-21502 is still the same antibody it was two years ago. Mouse was mistakenly removed during a website update and has since been re-added. This antibody still cross-reacts with mouse. In the past two years we have begun to validate more of our products for use in ICC and this is one that has had its uses updated since you last purchased.
-
Q: Is the epitope for NBP1-21502 extracellular or will it require permeabilization? Is it possible to get a sample size of one of the conjugated versions?
A: The immunogen used for this Ferroportin/SLC40A1 Antibody NBP1-21502 falls within a helical domain of the protein and some permeabilization is recommended for staining. Our lab uses 0.2% Tween-20 included in the blocking and diluent buffers to perform this permeabilization. As our directly conjugated antibodies are prepared per order, we unfortunately do not have sample sizes available.
-
Q: We are having difficulty producing a clean blot, i.e. when we probe our western blots with the recommended concentration of 1.0 ug/ml, we are seeing multiple bands. The order of the molecular weight ladder we used is as followed: 250, 130, 100, 70, 55, 35, 25, 15, and 10 kDa. Are you able to indicate what band may be ferroportin.?
A:
There is a western blot protocol specific to this antibody that can be found on the datasheet; it differs from your protocol quite a lot. I would give this protocol a try and see if you get better results.
Although I am uncertain of your blocking method, we find that BSA and PVDF can give high non-specific background so the nitrocellulose and NFDM are recommend for this antibody. In addition, there seems to be no protein expression of the SLC40A1 in liver or spleen based on Human Protein Atlas.
https://www.proteinatlas.org/ENSG00000138449-SLC40A1/tissue
A good positive control might be bone marrow.
-
Q: Would it be possible to conjugate this antibody to PE or PerCP?
A:
With regards to conjugating NBP1-21502SS (0.025 ml) to PE or PerCP, there are two possibilities: The first is that you could conjugate the antibody yourself, using easy-to-use kits. You would choose from Lightning-Link R-PE 3 x 0.01 mg, or Lightning-Link PerCP 3 x 0.01 mg. Because the buffer of the NBP1-21502 antibody is Tris-glycine, which is not compatible with the Lightning-Link conjugation chemistry as that targets amine groups, you would perform a buffer exchange first using the Antibody Concentration and Clean Up Kit. The lab has informed me that yes, they could conjugate your choice of PE or PerCP to this antibody for you. However, it would have to be the full-size antibody vial (0.1 ml) rather than the sample size (0.025 ml).
-
Q: I can’t get positive a result in IHC-P using NBP1-21502 (tissue: atherosclerosis hemorrhage plaque). PV-6001 and DAB is working. Can you make optimization suggestions on how to get this antibody to work?
A:
Optimization could really make a difference as we have seen some staining with the same antibody on frozen section. The main thing would be to focus on antigen retrieval step and the buffer used. For the antigen retrieval method, we would suggest an increase to at least 10-20 minutes to the pressure cooker step of the HIER method; the duration of the retrieval step can greatly determine if the antigen will be exposed or not for the antigen binding site of the antibody to recognize the protein. Also, I the incubation time for peroxidase blocking time could be reduced from 30min to 15 min, as we have mentioned. Since this is a TMP, permeabilization reagents should be used for the experiment. You can review what we have available on our site in terms of our general protocols:
HIER and IHC-P
-
Q: I have some leftovers of this antibody from two years ago and tried it for immunofluorescence on mouse, which did work well. I may need more of this antibody. Some things surprise me. In the old datasheet it says that there is cross reactivity with mouse. It does not say so in the new data sheet here on the web site. However, it does say so for all the conjugated versions of the Ab. What is correct? Also, in the old data sheet it says that it is for Western blotting. Now immunofluorescence is also mentioned. Is this still the same antibody? I don't have a lot number, but we got it in September 2010.
A: NBP1-21502 is still the same antibody it was two years ago. Mouse was mistakenly removed during a website update and has since been re-added. This antibody still cross-reacts with mouse. In the past two years we have begun to validate more of our products for use in ICC and this is one that has had its uses updated since you last purchased.
-
Q: Is the epitope for NBP1-21502 extracellular or will it require permeabilization? Is it possible to get a sample size of one of the conjugated versions?
A: The immunogen used for this Ferroportin/SLC40A1 Antibody NBP1-21502 falls within a helical domain of the protein and some permeabilization is recommended for staining. Our lab uses 0.2% Tween-20 included in the blocking and diluent buffers to perform this permeabilization. As our directly conjugated antibodies are prepared per order, we unfortunately do not have sample sizes available.
-
Q: We are having difficulty producing a clean blot, i.e. when we probe our western blots with the recommended concentration of 1.0 ug/ml, we are seeing multiple bands. The order of the molecular weight ladder we used is as followed: 250, 130, 100, 70, 55, 35, 25, 15, and 10 kDa. Are you able to indicate what band may be ferroportin.?
A:
There is a western blot protocol specific to this antibody that can be found on the datasheet; it differs from your protocol quite a lot. I would give this protocol a try and see if you get better results.
Although I am uncertain of your blocking method, we find that BSA and PVDF can give high non-specific background so the nitrocellulose and NFDM are recommend for this antibody. In addition, there seems to be no protein expression of the SLC40A1 in liver or spleen based on Human Protein Atlas.
https://www.proteinatlas.org/ENSG00000138449-SLC40A1/tissue
A good positive control might be bone marrow.
-
Q: Would it be possible to conjugate this antibody to PE or PerCP?
A:
With regards to conjugating NBP1-21502SS (0.025 ml) to PE or PerCP, there are two possibilities: The first is that you could conjugate the antibody yourself, using easy-to-use kits. You would choose from Lightning-Link R-PE 3 x 0.01 mg, or Lightning-Link PerCP 3 x 0.01 mg. Because the buffer of the NBP1-21502 antibody is Tris-glycine, which is not compatible with the Lightning-Link conjugation chemistry as that targets amine groups, you would perform a buffer exchange first using the Antibody Concentration and Clean Up Kit. The lab has informed me that yes, they could conjugate your choice of PE or PerCP to this antibody for you. However, it would have to be the full-size antibody vial (0.1 ml) rather than the sample size (0.025 ml).


![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-271220231285619.jpg)
![Immunohistochemistry-Paraffin: Rabbit Polyclonal Ferroportin/SLC40A1 Antibody [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/antibody/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-immunohistochemistry-paraffin-165202493537..jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202415394535.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202415382437.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202415363729.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202415371986.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-31020241534539.jpg)
![Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202415392561.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-31020241535654.jpg)
![Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202415371999.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202415382421.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202416165613.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-31020241616071.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-31020241616370.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202416173542.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-31020241616561.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202416171416.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-31020241616098.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202416173510.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202416171422.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202416171453.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-31020241616065.jpg)
![Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-31020241618269.jpg)
![Western Blot: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-3102024161602.jpg)
![Immunocytochemistry/ Immunofluorescence: Ferroportin/SLC40A1 Antibody - BSA Free [NBP1-21502] - Ferroportin/SLC40A1 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp1-21502_rabbit-polyclonal-ferroportin-slc40a1-antibody-310202416171438.jpg)