HIF-1 alpha Antibody (ESEE122) - BSA Free
Novus Biologicals | Catalog # NB100-131

Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Localization
Clonality
Host
Isotype
Theoretical MW
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for HIF-1 alpha Antibody (ESEE122) - BSA Free
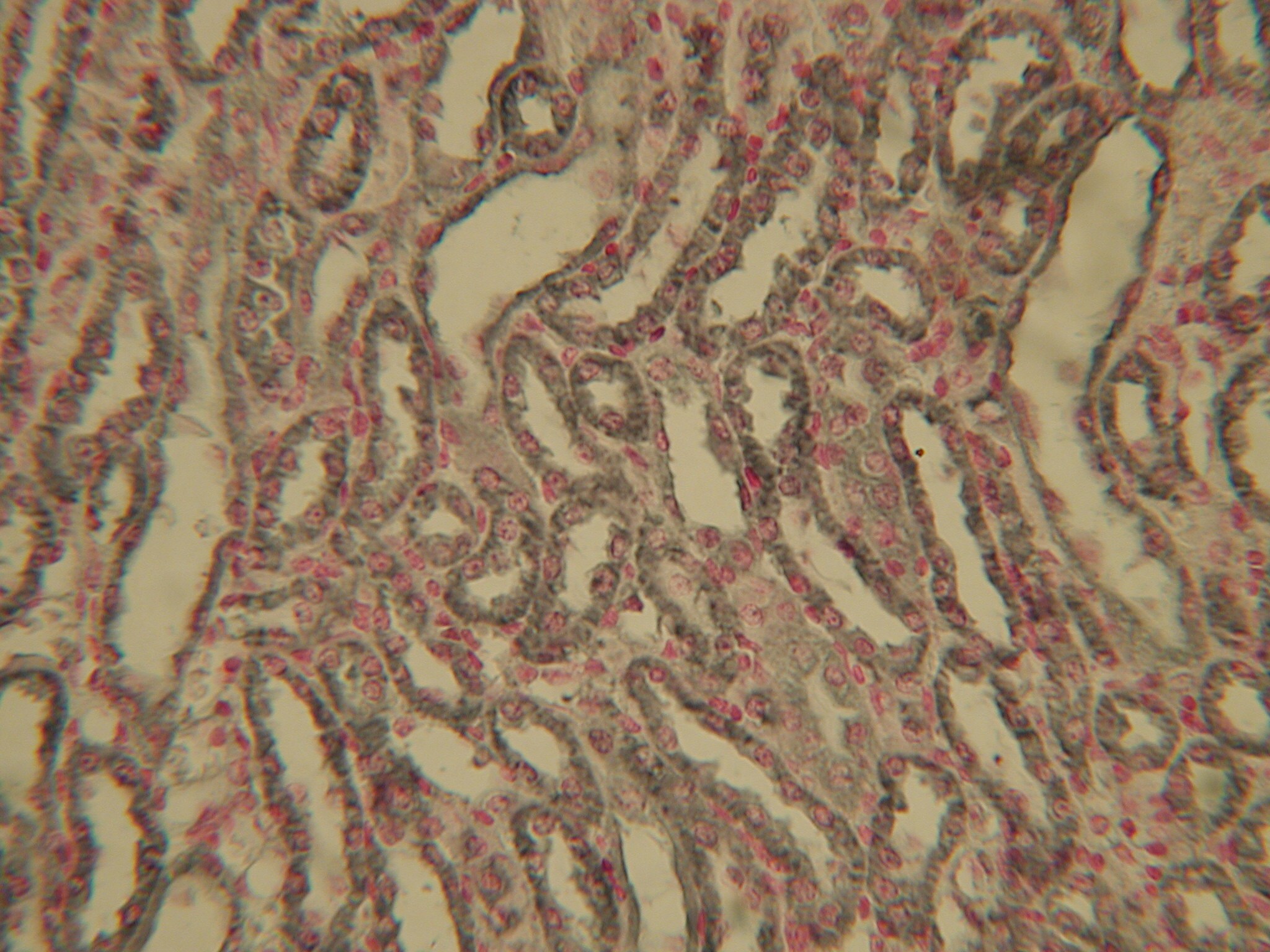
Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131]
HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-NB100-131-img0030.jpgImmunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131]
HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-NB100-131-img0029.jpgSimple Western: HIF-1 alpha Antibody (ESEE122) [NB100-131]
Simple Western: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Image shows a specific band for HIF-1 alpha in 0.5 mg/mL of Hypoxic HeLa lysate. This experiment was performed under reducing conditions using the 12-230 kDa separation system.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131]
Immunocytochemistry/Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Detection of HIF-1 alpha (red dye 568) in a cultured raw mouse macrophage cell line, using NB100-131. Photos courtesy of Susan Alexander and Hattie Gresham, PhD.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131]
Immunocytochemistry/Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with S. aureus. 100X magnification. Blue: DAPI nuclear staining.Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131]
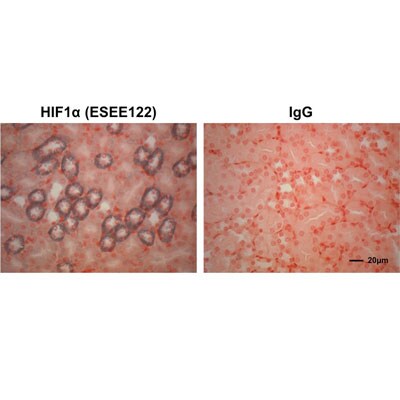
Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Analysis of a FFPE mouse kidney tissue section using HIF-1 alpha antibody clone ESEE122 at 1ug/mL concentration. The detection was performed using X-cell plus universal HRP polymer detection system with Vector SG chromagen substrate. Image courtesy of a product review by Steven Grover.Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131]
HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-NB100-131-img0031.jpgImmunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131]
Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Analysis of HIF-1 alpha in paraffin-embedded mouse kidney tissue section using anti-HIF-1 alpha antibody. Image from verified customer review.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131]
Immunocytochemistry/Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with S. aureus, using NB100-131. Blue: DAPI nuclear staining. Image courtesy of Susan Alexander and Hattie Gresham, PhD.Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131]
Immunocytochemistry/Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Detection of HIF-1 alpha (red dye 568) in a cultured raw mouse macrophage cell line. 100X magnification. Image courtesy of Susan Alexander and Hattie Gresham, PhD.Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131]
Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Negative control stain of human placenta (from sea level) using mouse IgG at 1:100. 4uM paraffin-embedded section.Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131]
Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Analysis of a FFPE tissue section of human renal cancer xenograft using HIF-1 alpha antibody (NB100-131 Lot 83115) at 1:200 dilution. The antibody generated a strong cytoplasmic staining mainly in the cancer cells. Only a fraction of cells depicted nuclear staining, while weak to negligible positivity was seen in the tumor stromal cells.Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131]
Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - HIF-1 alpha staining in hypoxia-induced human placenta.Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131]
HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-Paraffin-NB100-131-img0032.jpgImmunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] -
Positive immunohistochemical staining for (A) VEGF, (B) HIF-1 alpha, (C) Dll4 (tumor cells), (D) Dll4 (endothelial cells), and (E) CD31 (for microvessel counting, ×200 magnification).Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] -
Immunohistochemical analysis in non-GIST STS representing negative, and score 1–3 of CAIX, GLUT-1, HIF-1 alpha, and HIF-2 alpha. non-GIST STS: non-gastrointestinal stromal tumor soft-tissue sarcomas, CAIX: carbonic anhydrase IX, GLUT-1: glucose transporter-1, and HIF-1/2 alpha : hypoxia induced factor 1/2 alpha.Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] -
Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Histologically distinct cell types in hemangioblastomas do not arise from a common ancestral clone. “Representative images of sample SH-0622 acquired at 400x of (a) H + E & IHC for (b) HIF1-alpha, (c) VEGF, & (d) PDGFR-beta reveal heterogenous cell types in this tumor characterized by a rich vascular network. Arrowheads indicate that the stromal cells demonstrate increased cytoplasmic staining for HIF1-alpha & VEGF, whereas the double arrowheads highlight PDGFR-beta protein restricted to vascular endothelium. Scale bar is 25 μm. Image collected & cropped by CiteAb from the following publication (https://actaneurocomms.biomedcentral.com/articles/10.1186/s40478-014-01…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] -
Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Immunohistochemical analysis in non-GIST STS representing negative, & score 1–3 of CAIX, GLUT-1, HIF-1 alpha, & HIF-2 alpha. non-GIST STS: non-gastrointestinal stromal tumor soft-tissue sarcomas, CAIX: carbonic anhydrase IX, GLUT-1: glucose transporter-1, & HIF-1/2 alpha : hypoxia induced factor 1/2 alpha. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/22454562), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] -
Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Digital image analysis of cytoplasmic & membranous staining. Cytoplasmic HIF-1 alpha staining is shown (A) & automated image analysis utilizing TissueIA recognizes cytoplasmic HIF-1 alpha staining highlighted in green color (B). CA9 is shown in membranous staining (C) & automated image analysis determines membranous CA9 staining highlighted in green color (D). The output from the algorithm returns a number of quantitative measurements for intensity & percentage of positive staining present. Scale bar: 100 μm. Image collected & cropped by CiteAb from the following publication (http://translational-medicine.biomedcentral.com/articles/10.1186/1479-5…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] -
Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Immunohistochemical staining of HIF-1 alpha, VEGF-A & VEGF-C in normal renal tissue (A-C) & clear cell renal cell carcinoma (CCRCC) (D-F). A homogeneous cytoplasmic staining of tubular cells & weak staining in glomerules was observed with HIF-1 alpha (A), while VEGF-A & VEGF-C were positive in tubular cells, glomerular mesangium & interstitial macrophages (B & C). In CCRCC, HIF-1 alpha immmunoreactivity was nuclear and/or cytoplasmic (D), while it was perimembranous and/or diffuse cytoplasmic for VEGF-A & VEFG-C (E & F). (magnification ×200). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/19302703), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] -
Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Positive immunohistochemical staining for (A) VEGF, (B) HIF-1 alpha, (C) Dll4 (tumor cells), (D) Dll4 (endothelial cells), & (E) CD31 (for microvessel counting, ×200 magnification). Image collected & cropped by CiteAb from the following publication (https://www.spandidos-publications.com/10.3892/or.2012.2075), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] -
Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Immunohistochemical staining of HIF-1 alpha, VEGF-A & VEGF-C in normal renal tissue (A-C) & clear cell renal cell carcinoma (CCRCC) (D-F). A homogeneous cytoplasmic staining of tubular cells & weak staining in glomerules was observed with HIF-1 alpha (A), while VEGF-A & VEGF-C were positive in tubular cells, glomerular mesangium & interstitial macrophages (B & C). In CCRCC, HIF-1 alpha immmunoreactivity was nuclear and/or cytoplasmic (D), while it was perimembranous and/or diffuse cytoplasmic for VEGF-A & VEFG-C (E & F). (magnification ×200). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/19302703), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] -
Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Representative immunohistochemical expression for HIF-1 alpha, c-Met, CA9 & GLUT1. HIF-1 alpha is stained in cytoplasm shown with no staining in normal cervix (A), weak staining intensity in high grade CIN (B), & strong staining intensity in squamous cell carcinoma (C). c-Met (D-F), CA9 (G, H) & GLUT1 (I) shows cell membranous staining. Representative c-Met expression in cervical samples shown with no staining in normal cervix (D), weak membranous staining intensity in squamous cell carcinoma (E) & strong intensity in squamous cell carcinoma (F). CA9 expression showing moderate intensity staining in carcinoma in situ (CIS) (G) & strong staining in adenocarcinoma (H). GLUT1 expression showing strong intensity in squamous cell carcinoma (I). Scale bar: 50 μm. Image collected & cropped by CiteAb from the following publication (http://translational-medicine.biomedcentral.com/articles/10.1186/1479-5…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] -
Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - Representative immunohistochemical expression for HIF-1 alpha, c-Met, CA9 & GLUT1. HIF-1 alpha is stained in cytoplasm shown with no staining in normal cervix (A), weak staining intensity in high grade CIN (B), & strong staining intensity in squamous cell carcinoma (C). c-Met (D-F), CA9 (G, H) & GLUT1 (I) shows cell membranous staining. Representative c-Met expression in cervical samples shown with no staining in normal cervix (D), weak membranous staining intensity in squamous cell carcinoma (E) & strong intensity in squamous cell carcinoma (F). CA9 expression showing moderate intensity staining in carcinoma in situ (CIS) (G) & strong staining in adenocarcinoma (H). GLUT1 expression showing strong intensity in squamous cell carcinoma (I). Scale bar: 50 μm. Image collected & cropped by CiteAb from the following publication (http://translational-medicine.biomedcentral.com/articles/10.1186/1479-5…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody (ESEE122) - BSA Free [NB100-131] -
CASQ2 affects the TME and invasiveness through HIF1 alpha. (A) Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis using the mouse genome (Differentially expressed genes, DEGs; n = 530). (B) The heatmap for average of expression (FPKM) value of most significant differentially expressed genes of angiogenesis and the HIF1 alpha signaling pathway. (C) The mRNA level of Hif1a, Pgf, Flt1, Kdr, Flt4, and Nos3 in Hs578T‐CTL and CASQ2 o/e tumors (means +/- SEM, n = 3; ***P < 0.001 by the multiple t‐test). (D, E) Expression level of alpha ‐SMA, FSP, and HIF1 alpha in WI‐38 cells after indirect coculture with Hs578T‐CTL or Hs578T‐CASQ2 o/e cells (means +/- SEM, n = 3; *P < 0.05 by the multiple t‐test). (F, G) Expression level of CASQ2 and vimentin in Hs578T‐CTL or Hs578T‐CASQ2 o/e cells indirectly cocultured with WI‐38 (means +/- SEM, n = 3; *P < 0.05 by the multiple t‐test). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/34743414), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody (ESEE122) - BSA Free [NB100-131] -
CASQ2 affects the TME and invasiveness through HIF1 alpha. (A) Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis using the mouse genome (Differentially expressed genes, DEGs; n = 530). (B) The heatmap for average of expression (FPKM) value of most significant differentially expressed genes of angiogenesis and the HIF1 alpha signaling pathway. (C) The mRNA level of Hif1a, Pgf, Flt1, Kdr, Flt4, and Nos3 in Hs578T‐CTL and CASQ2 o/e tumors (means +/- SEM, n = 3; ***P < 0.001 by the multiple t‐test). (D, E) Expression level of alpha ‐SMA, FSP, and HIF1 alpha in WI‐38 cells after indirect coculture with Hs578T‐CTL or Hs578T‐CASQ2 o/e cells (means +/- SEM, n = 3; *P < 0.05 by the multiple t‐test). (F, G) Expression level of CASQ2 and vimentin in Hs578T‐CTL or Hs578T‐CASQ2 o/e cells indirectly cocultured with WI‐38 (means +/- SEM, n = 3; *P < 0.05 by the multiple t‐test). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/34743414), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody (ESEE122) - BSA Free [NB100-131] -
CASQ2 affects the tumor invasiveness through HIF1 alpha. (A, B) Expression level of HIF1 alpha in caffeine‐treated Hs578T cells with or without overexpression of CASQ2 (means +/- SEM, n = 3; *P < 0.05, **P < 0.01 by the Mann–Whitney U‐test). (C) Confocal microscopy images showing F‐actin in Hs578T cells overexpressing CASQ2 under hypoxic conditions (**P < 0.01 by the Mann–Whitney U‐test). Scale bar = 10 μm. (D) Proliferation rate of control (CTL) and CASQ2‐overexpressing (CASQ2 o/e) breast cancer cells under normoxic and hypoxic conditions (mean +/- SEM, n = 3; *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001 by ANOVA with Tukey’s post hoc test). (E, F) Hematoxylin and eosin staining of lung tissue sections from Hs578T‐CTL‐ or CASQ2‐derived tumors (mean +/- SEM, n = 5; *P < 0.05 and **P < 0.01 by two‐tailed Student’s t‐test). Scale bar = 100 μm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/34743414), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: HIF-1 alpha Antibody (ESEE122) - BSA Free [NB100-131] -
CASQ2 induces phenotypic changes in breast cancer cells. (A) Relationship between the proliferation rate and overexpression of CASQ2 in breast cancer cell lines (mean +/- SEM, n = 3; *P < 0.05, **P < 0.01, and ***P < 0.001 using the multiple t‐test). (B) Migration and invasion rates of Hs578T cells (mean +/- SEM, n = 3; **P < 0.01 using two‐tailed Student’s t‐test). Scale bar = 50 μm. (C) Three‐dimensional culture of Hs578T cells (mean +/- SEM, n = 3; **P < 0.01 using two‐tailed Student’s t‐test). Scale bar = 100 μm. (D) Tumorsphere culture of Hs578T cells (mean +/- SEM, n = 3; **P < 0.01 using two‐tailed Student’s t‐test). Scale bar = 100 μm. (E) Expression of CD44, CD24, and ALDH1 cancer stem cell markers in adherent and tumorsphere cultures of Hs578T cells. (F) Measurement of intracellular Ca2+ in Hs578T cells using a calcium crimson reagent. Cells were loaded with the calcium indicator calcium crimson (5 μm) for 30 min, and then, BAPTA‐AM (10 μm) was added. After washing, the cells were stimulated by treatment with 5 μm caffeine to measure the intracellular calcium concentration at 360 s (mean +/- SEM, n = 3; *P < 0.05 and **P < 0.01 after ANOVA with Tukey’s post hoc test). (G) Effect of lacidipine on the expression of cancer stem cell markers in tumorspheres of breast cancer cells. The figure shows one of three independent experiments. Statistical test result is shown in Fig. S6. (H) Epithelial–mesenchymal transition (EMT)‐related protein expression in breast cancer cells. All results are representatives of three independent experiments. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/34743414), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for HIF-1 alpha Antibody (ESEE122) - BSA Free
Flow Cytometry
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Frozen
Immunohistochemistry-Paraffin
Immunoprecipitation
Simple Western
Western Blot
In Simple Western only 10 - 15 uL of the recommended dilution is used per data point.
See Simple Western Antibody Database for Simple Western validation: Tested in Hypoxic HeLa lysate 0.5 mg/mL, separated by Size, antibody dilution of 1:2000. Separated by Size-Wes, Sally Sue/Peggy Sue.
Reviewed Applications
Read 3 reviews rated 4.3 using NB100-131 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: HIF-1 alpha/HIF1A
HIF-1 or hypoxia inducible factor 1 (predicted molecular weight 93kDa), is a transcription factor commonly referred to as a "master regulator of the hypoxic response" for its central role in the regulation of cellular adaptations to hypoxia. In its active form under hypoxic conditions, HIF-1 is stabilized by the formation of a heterodimer of HIF-1 alpha and ARNT/HIF-1 beta subunits. Nuclear HIF-1 engages p300/CBP for binding to hypoxic response elements (HREs). This process induces transcription and regulation of genes including EPO, VEGF, iNOS2, ANGPT1 and OCT4 (4,5).
Under normoxic conditions, the HIF-1 alpha subunit is rapidly targeted and degraded by the ubiquitin proteasome system. This process is mediated by prolyl hydroxylase domain enzymes (PHDs), which catalyze the hydroxylation of key proline residues (Pro-402 and Pro-564) within the oxygen-dependent degradation domain of HIF-1 alpha. Once hydroxylated, HIF-1 alpha binds the von Hippel-Lindau tumor suppressor protein (pVHL) for subsequent ubiquitination and proteasomal degradation (4). pVHL dependent regulation of HIF-1 alpha plays a role in normal physiology and disease states. Regulation of HIF-1 alpha by pVHL is critical for the suppressive function of FoxP3+ regulatory Tcells (6). Repression of pVHL expression in chronic lymphocytic leukemia (CLL) B cells leads to HIF-1 alpha stabilization and increased VEGF secretion (7).
References
1. Semenza, G. L., Agani, F., Feldser, D., Iyer, N., Kotch, L., Laughner, E., & Yu, A. (2000). Hypoxia, HIF-1, and the pathophysiology of common human diseases. Advances in Experimental Medicine and Biology.
2. Muz, B., de la Puente, P., Azab, F., & Azab, A. K. (2015). The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia. https://doi.org/10.2147/hp.s93413
3. Huang, Y., Lin, D., & Taniguchi, C. M. (2017). Hypoxia inducible factor (HIF) in the tumor microenvironment: friend or foe? Science China Life Sciences. https://doi.org/10.1007/s11427-017-9178-y
4. Koyasu, S., Kobayashi, M., Goto, Y., Hiraoka, M., & Harada, H. (2018). Regulatory mechanisms of hypoxia-inducible factor 1 activity: Two decades of knowledge. Cancer Science. https://doi.org/10.1111/cas.13483
5. Dengler, V. L., Galbraith, M. D., & Espinosa, J. M. (2014). Transcriptional regulation by hypoxia inducible factors. Critical Reviews in Biochemistry and Molecular Biology. https://doi.org/10.3109/10409238.2013.838205
6. Lee, J. H., Elly, C., Park, Y., & Liu, Y. C. (2015). E3Ubiquitin Ligase VHL Regulates Hypoxia-Inducible Factor-1 alpha to Maintain Regulatory T Cell Stability and Suppressive Capacity. Immunity. https://doi.org/10.1016/j.immuni.2015.05.016
7. Ghosh, A. K., Shanafelt, T. D., Cimmino, A., Taccioli, C., Volinia, S., Liu, C. G.,... Kay, N. E. (2009). Aberrant regulation of pVHL levels by microRNA promotes the HIF/VEGF axis in CLL B cells. Blood. https://doi.org/10.1182/blood-2008-10-185686
Long Name
Alternate Names
Gene Symbol
Additional HIF-1 alpha/HIF1A Products
Product Documents for HIF-1 alpha Antibody (ESEE122) - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for HIF-1 alpha Antibody (ESEE122) - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for HIF-1 alpha Antibody (ESEE122) - BSA Free
Customer Reviews for HIF-1 alpha Antibody (ESEE122) - BSA Free (3)
Have you used HIF-1 alpha Antibody (ESEE122) - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: ImmunocytochemistrySample Tested:Species: MouseVerified Customer | Posted 02/04/2015HIF1alpha staining on paraffin-embedded kidney sections
-
Application: Immunohistochemistry-ParaffinSample Tested: Murine KidneySpecies: MouseVerified Customer | Posted 01/31/2013
-
Application: Immunohistochemistry-ParaffinSample Tested: human melanomaSpecies: HumanVerified Customer | Posted 02/02/2009
There are no reviews that match your criteria.
Protocols
View specific protocols for HIF-1 alpha Antibody (ESEE122) - BSA Free (NB100-131):
Immunohistochemistry Procedures
Paraffin Sections
1.Prior to performing the IPOX (immunoperoxidase) experiment, dewax the paraffin sections by baking them at 60 degrees C for 30 minutes and then putting them through citroclear [Citroclear is a mounting agent (chemical name Limonene, also known as Histoclear, Bioclear)].
2.Hydrate the sections through the following series:
A.3 X 5 minutes xylenes
B.3 X 5 minutes 100% Etoh
C.2 minutes 95% Etoh
D.2 minutes 70% Etoh
E.1 minute 50% Etoh
F.1 minute ddH2O
G.1 minute TBS
1.Block endogenous peroxidase with 0.5% hydrogen peroxide in water, for 30 minutes.
2. Antigen unmasking is performed by incubating at 60 degrees C for 16 hours, in 50mmol/L Tris and 0.2 mmol/L EDTA (pH 9.0), using a covered water bath.
1.Rinse slides with PBS and then incubate with PBS containing 0.2% Triton X-100 for 10 minutes.
2.Rinse slides with PBS.
3. Incubate sections with 1:8000 dilution of anti-HIF-1 alpha (NB100-131) for 90 minutes at room temperature (RT).
4. Incubate sections in secondary HRP-conjugated goat anti-mouse serum for 30 minutes at RT.
5. Incubate sections in tertiary HRP-conjugated rabbit anti-goat serum for 30 minutes at RT.
6. Develop the peroxidase reaction using diaminobenzidine.
7. Wash slide and mount in aqueous mountant.
Substitution of the primary antibody with PBS can be used as a negative control.
1. Deparaffinize to water:Xylene #1-10 dipsXylene #2-10 dips 100%EtOH #1-10 dips 100%EtOH #2-10 dips 95% EtOH-10 dips 70%EtOH-10 dips diH2O-2 changes
2. Rinse in PBS for two minutes.
3. Quench slides is MeOH/H2O2 for 5-10 minutes (1 part 30% H2O2/36 parts 70% MeOH; 8 mls H2O2/288 mls 70% MeOH).
4. Unmask antigens by boiling for 3 minutes in 0.01 M Citrate Buffer, pH 5.5. 47.2gr Sodium Citrate 8.3gr Citric Acid pH to 5.5 qs to 0.5 L dH2O
5. Rinse in PBS.
6. Apply 2 drops blocking solution (10% non-immune normal goat serum, Zymed Labs, Cat # 50-197). Incubate for 10 minutes in humidity chamber
7. Incubate for 10 minutes in humidity chamber.
8. Do not rinse.
9. Incubate in mAb HIF-1 alpha (cat# NB 100-131), diluted 1:250 in PBS (10ul /2.5mls) overnight at 4 degrees C, in humidity chamber.
10. Rinse in PBS.
11. Incubate in 2 drops Biotinylated Secondary Antibody for 10 minutes in humidity chamber.
12. Rinse in PBS.
13. Incubate in 2 drops Enzyme Conjugate solution (HRP-Streptavidin) for 10 minutes in humidity chamber.
14. Rinse in PBS.
15. Incubate in 2 drops Substrate-Chromatogen solution AEC solution, (AEC Single Solution, Zymed Labs, Cat# 00-1111) for 5-10 minutes in humidity chamber.
16. Rinse well in dH20.
17. Counterstain with hematoxylin for 1 minute.
18. Rinse well in tap water until it runs clear.
19. Mount coverslip with water soluble mounting media. Do not dehydrate. (Alcohols will remove the AEC color).
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for HIF-1 alpha Antibody (ESEE122) - BSA Free
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
-
Q: Can HIF-1 alpha Antibody (Dylight 488), product NB100-479G, react with goat species? Does this product have preservatives in it?
A:
NB100-479G has not been tested in goat species. Only the listed species on the product page and datasheet will be guaranteed.
Reactivity: Hu, Mu, Rt, Ca, Fi, Ha, Pm, Rb
The immunogen for this antibody corresponds to amino acids 530-825 of mouse Hifa. Running a sequence alignment of this sequence with the goat sequence found on UniProt yields around 81% homology.Mouse Hif1a: https://www.uniprot.org/uniprot/Q61221#sequences
Goat Hif1a: https://www.uniprot.org/uniprot/A0A023R978#sequencesLastly, there is 0.05% Sodium Azide present in the formulation of the product. This is also listed on the product page and datasheet.
Buffer: 50mM Sodium Borate
Preservative: 0.05% Sodium Azide -
Q: Could you clarify if Yeast is a reactive species for HIF1a product #NB100-105?
A: This HIF-1 alpha antibody has not been tested in yeast. The homology is not significantly homologous so we do not believe there will be cross reactivity to the yeast protein.
-
Q: Do you have any more protocol information regarding the cytospin staining images on your datasheet, credited to Susan Alexander and Hattie Gresham?
A: We do have the following notes:
1) cultured raw cells (mouse macrophage cell line) were used
2) cell cytospin from a lavage of a skin pouch (mouse) infected with staph aureus. 10 100X pic
3) Also a cell cytospin from a lavage of a skin pouch of a mouse infected with staph aureus. 14. WT pic.
4) cultured raw cells. 19. pic The blue staining is DAPI (nuclear stain). The stain was conjugated with a kit from molecular probes (A-20184) following their protocol. The final antibody concentration is uncertain but possibly around 25 ug/mL, used at 1:2 or 1:3 (about 8-12ug/mL). -
Q: I am curious to know the biochemical reactions of CoCl2 that mimic hypoxia. Is it that CoCl2 can bind any ubiquitin enzyme which regulates their degradation?
A:
CoCl2 inhibits PHD enzymes (the body’s “oxygen sensors”) by replacing the Fe ion with Co, preventing these enzymes from marking HIF-1 alpha for degradation. CoCl2-based hypoxia mimetic samples are often used as positive control in HIF analysis. For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs.
-
Q: I am doing HIF1 westerns in HIF-overexpressing mouse liver and adipose tissue using Novus antirabbit HIF1a antibody with overnight incubation. I am getting strong bands around 90kDa. I am aware that HIF theoretical molecular weight is 93kDa, but in westerns, the HIF band is usually around 120kDa according to my internet research. Can someone let me know if I’m getting the right HIF band or just some non-specific bands? Thanks.
A:
(1) HIF-1 alpha’s theoretical molecular weight is 93kDa. The post translationally modified/ubiquitinated form of HIF-1 alpha protein (fails to undergo proteasomal degradation) shows up as a band in the 110-130 kDa range on a Western blot.
(2) The dimeric protein may appear at a position above 200 kDa on non-reducing gels.
(3) Importantly, HIFs are among the most rapidly degradable proteins; therefore, sample preparation is highly important when analyzing HIF1 alpha or HIF2 alpha. When degraded, HIF-1 alpha may show up between 40-80 kDa position on Western blot. Degradation may be avoided by preparing the samples as soon as possible after collection of cells/tissues in hypoxic chamber. Notably, the tissues/cells should be kept on ice during lysate preparation and the lysates should be analyzed as soon as possible.
(4) For troubleshooting suggestions/feedback on more than 25 similar frequently asked questions, I would recommend visiting Novus page: FAQs - Hypoxia and HIFs
(5) Last but not the least, Novus technical support team may be contacted via email -
Q: I have Hif1a nuclear protein extract at -80C. I am wondering if anyone knows how long it would be good for at that temperature since HIf1a is known to be degraded easily.
Thank you!A: You could try a few things to further inhibit the degradation.
1) Use the protease inhibitors (if you are not already using them).
2) Lyse cells into a buffer that contains SDS or LDS (eg: Laemmli's buffer), since SDS and LDS denature and inhibit proteases. Lysis may even be performed with reducing agents in the buffer (eg. DTT), but this will make your lysates unsuitable for BCA assay.
3) Lysing samples rapidly ensures that the samples are instantly homogenized (it also shears DNA released by the SDS).
5) Flash-freezing samples in liquid nitrogen rather than freezing at -80*C reduces the window of time for protease activity.
6) Freeze samples in individual aliquots, instead of thawing the same vial multiple times. -
Q: I performed several Western Blots of HIF-1 alpha with different lysis buffers, whole lysates, and cytoplasm/nuclei extractions. I can’t seem to get a good western blot (poor signal, band much lower than expected, etc.). Can someone suggest some technical considerations/tricks I should consider using?
A:
A major issue that researchers working with HIF-1 alpha is degradation due to exposure to oxygen. In western blot, this results in a weaker band and/or the appearance of multiple low molecular weight bands (40-80 kDa). We recommend preparing the lysates after collection of cells/tissues as quickly as possible (on ice), preferably in a hypoxic chamber. We also recommend including a true hypoxia mimetic control (eg: cells treated with CoCl2, DMOG… etc.). The controls help distinguish your band of interest from potential degradation/dimer bands.
For more troubleshooting tips and frequently asked questions regarding hypoxia/HIFs, you can refer to our hypoxia-related FAQs. -
Q: I would like to know, does a path exist for detection of HIF 1 in venous blood before and after revascularization of the leg?
A: We are not entirely sure if HIF-1 alpha will be present in the leg after revascularization. It may be present, but you may want to search the literature to see if this has been looked at before. If not, then this would certainly be an experiment worth doing.
-
Q: I've done colorations with an HIF-1alpha antibody and am now wondering if it is HIF-1alpha we see or it may be cross-reactivity with any other in the cytoplasm in mast cells. The image is reminiscent of the Immunofluorescence staining available on your site for HIF-1 alpha Antibody (ESEE122) [NB100-131] (Detection of HIF-1 alpha (red dye) in a cell cytospin from a lavage of a murine skin pouch infected with Staph Aureus, using NB100-131.) Gresham and Alexander have done that picture, but I cannot find it published. How to explain the appearance of HIF-1 staining. Our staining is localized to the nucleus, but is bead-like.
A:
HIF-1 alpha can be found at very low levels in the cytoplasm under normoxic conditions. During hypoxia, HIF1a is stabilized and translocates to the nucleus to act as a transcription factor. You should expect to see nuclear staining in your hypoxic samples, but may also observe faint cytoplasmic staining corresponding to degraded HIF. We do not have a publication from Gresham and Alexander on our website, and I believe that their images were submitted as a customer review of the antibody. However we do have five other publications referring to immunofluorescent staining, which you may find helpful. You can see these at the bottom of the "Images, Reviews and Publications" tab by filtering on application and selecting "ICC/IF".
-
Q: Is cross-reactivity with HIF-2 alpha tested/predicted?
A: Although we don’t have cross-reactivity data with regards to HIF-2 alpha, we predict minimal cross-reactivity based on low sequence similarity observed from BLAST analysis between HIF-1 alpha and HIF-2 alpha.
-
Q: Is your HIF-1 alpha Antibody able to work in Western Blot application?
A: Yes, this antibody can be used in Western Blot.
-
Q: Our customer wants to buy the peptide for your antibody NB100-131. Which catalog number does the peptide have?
A: This antibody was made to a recombinant protein containing amino acids 329-530 of HIF-1 alpha. Unfortunately we do not carry the recombinant protein as a sellable item.
-
Q: Our lab recently ordered NB100-449, HIF-1 alpha antibody. Unfortunately an inexperienced technician stored it at -20C rather than 4C for approximately 2 days. Have you done any tests to determine antibody functionality if frozen?
A: The recommended storage condition of HIF-1 alpha antibody NB100-449 is 4C and we highly recommended not storing the product lower than the freezing point, as it may potentially disrupt the protein folding and destroy the antigen binding site of the antibody. Since we likely have not tested a storage condition of -20C for this antibody, we cannot really say if this antibody has been impaired by the storage condition. Our recommendation would be to test the antibody in a small portion of your treated cell line and see if the antibody is still reactive to the HIF-1 alpha protein.
-
Q: We got the Hif1a (NB100-105) antibody from you guys. I used the concentration that is mentioned on your website, but I am getting a band of a completely different size (~70kDa) and not the 120 kDa mentioned.
A:
HIF-1 alpha is a notoriously difficult protein to work with due to its rapid degradation. Therefore, the ~70kDa bands are most likely degradation products. It is very important to lyse the cells in hypoxic conditions. We strongly recommend lysing the cells directly into the Laemmli buffer and doing that quickly, so that the exposure to oxygen is minimized.
Please go through our hypoxia related FAQs, you should find them very informative.Also, running a positive control may help confirm the band specificity in your samples. You may prepare them yourself or choose some from our catalog, for example:
1) HeLa Hypoxic / Normoxic Cell Lysate (NBP2-36452)
2) HeLa Hypoxic (CoCl2) / Normoxic Cell Lysate (NBP2-36450) -
Q: We ordered and received the HIF-1 alpha antibody NB100-449 and on the packing slip it says that is prepared in TBS+0.1% BSA. I will be using it for western blots. DO you think my choice of blocking buffer (milk or Blotto) could interfere with the activity of this antibody or should it be necessarily BSA based?
A:
Choice of blocking buffer is entirely within your discretion; it will not affect the antibody binding activity. Please note, some blocking buffers may work better than others and sometimes optimization is needed.
When working with hypoxia there are other important factors to consider, as HIF-alpha is very easily degraded. The lysates should be freshly prepared. Also lysate preparation should be as quick as possible to avoid any exposure to oxygen - we recommend lysing cells directly into the SDS sample loading buffer (Laemmli buffer). We also highly recommend using positive control (you can prepare them yourself or choose some of those we have for sale).
I have attached some additional information that you may find quite useful. Also here are some hypoxia related FAQ addressing common concerns. -
Q: What is the molecular weight (kDa) of protein HIF 1 alpha in western blot?
A: The theoretical molecular weight of HIF 1-alpha is ~93kDa. However, you will likely see a band between 100-120kDa due to phosphorylation.
-
Q: What's the difference between NBP2-75977 vs NBP2-75978?
A: While the same immunogen was used to make both HIF-1 alpha antibodies, they are different clones, meaning they recognize a different epitope on the immunogen.
-
Q: Which antibody(ies) do you recommend for the detection of HIF-1a by immunohistochemistry in the sections of paraffin-embedded mouse liver samples? I would appreciate if you can give me several choices and rank them in the order of performance. My goal is to distinguish HIF upregulation by prolyl hydroxylase inhibitor in different liver cells.
A: All of our antibodies are of high quality and are well tested/validated in species/applications we list on the datasheet. However, we suggest the following four HIF-1 alpha antibodies based upon customer reviews, as well as the number of peer reviewed publications in which these products have been cited by researchers from reputed institutes. (1) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-105) (cited in at least 218 peer reviewed publications) (2) HIF-1 alpha Antibody (cat# NB100-479) (cited in at least 51 peer reviewed publications) (3) HIF-1 alpha Antibody (H1alpha67) (cat# NB100-123 ) (cited in at least 38 peer reviewed publications) (4) HIF-1 alpha Antibody (cat# NB100-449) (cited in at least 31 peer reviewed publications).
-
Q: Why is there a difference between the theoretical MW for HIF1A and the observed MW for HIF-1 alpha?
A: HIF1A, like many other proteins, has post-translational modifications. Depending on the size, amount and nature of the post-translational modifications, it can cause subtle to very large changes in molecular weight.
Associated Pathways
 HIF Repressor Pathways
HIF Repressor Pathways
 Notch Signaling Pathways
Notch Signaling Pathways


![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-NB100-131-img0029.jpg)
![Simple Western: HIF-1 alpha Antibody (ESEE122) [NB100-131] Simple Western: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Simple-Western-NB100-131-img0020.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunocytochemistry-Immunofluorescence-NB100-131-img0017.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-NB100-131-img0030.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunocytochemistry-Immunofluorescence-NB100-131-img0016.jpg)
![Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-Paraffin-NB100-131-img0019.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-NB100-131-img0031.jpg)
![Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-Paraffin-NB100-131-img0021.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-NB100-131-img0033.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunocytochemistry-Immunofluorescence-NB100-131-img0004.jpg)
![Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunocytochemistry/ Immunofluorescence: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunocytochemistry-Immunofluorescence-NB100-131-img0007.jpg)
![Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-Paraffin-NB100-131-img0018.jpg)
![Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-Paraffin-NB100-131-img0024.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-NB100-131-img0025.jpg)
![Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131] Immunohistochemistry-Paraffin: HIF-1 alpha Antibody (ESEE122) [NB100-131]](https://resources.rndsystems.com/images/products/HIF-1-alpha-Antibody-ESEE122-Immunohistochemistry-Paraffin-NB100-131-img0032.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - HIF-1 alpha Antibody (ESEE122)](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-271220231253347.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - HIF-1 alpha Antibody (ESEE122)](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-27122023125331.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - HIF-1 alpha Antibody (ESEE122)](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-31020241534345.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - HIF-1 alpha Antibody (ESEE122)](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-310202415371952.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - HIF-1 alpha Antibody (ESEE122)](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-31020241534362.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - HIF-1 alpha Antibody (ESEE122)](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-310202415371953.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - HIF-1 alpha Antibody (ESEE122)](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-310202415384140.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - HIF-1 alpha Antibody (ESEE122)](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-310202415363744.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - HIF-1 alpha Antibody (ESEE122)](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-310202415532087.jpg)
![Immunohistochemistry: HIF-1 alpha Antibody (ESEE122) [NB100-131] - HIF-1 alpha Antibody (ESEE122)](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-31020241553723.jpg)
![Western Blot: HIF-1 alpha Antibody (ESEE122) - BSA Free [NB100-131] - HIF-1 alpha Antibody (ESEE122) - BSA Free](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-western-blot-132202612125124.jpg)
![Western Blot: HIF-1 alpha Antibody (ESEE122) - BSA Free [NB100-131] - HIF-1 alpha Antibody (ESEE122) - BSA Free](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-western-blot-13220261213183.jpg)
![Western Blot: HIF-1 alpha Antibody (ESEE122) - BSA Free [NB100-131] - HIF-1 alpha Antibody (ESEE122) - BSA Free](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-western-blot-132202613302211.jpg)
![Western Blot: HIF-1 alpha Antibody (ESEE122) - BSA Free [NB100-131] - HIF-1 alpha Antibody (ESEE122) - BSA Free](https://resources.rndsystems.com/images/products/nb100-131_mouse-monoclonal-hif-1-alpha-antibody-esee122-western-blot-13220261219356.jpg)