HIF Enhancer Pathways
and Increases Transactivation
and Increases Transactivation
to Increase Transactivation
and Increases Transactivation
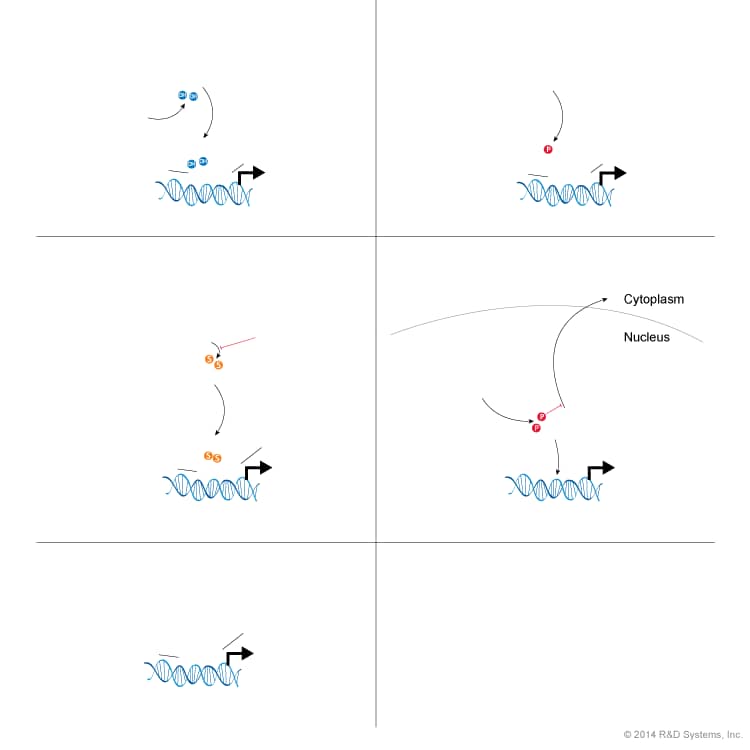
Overview of HIF Transcription Factor Stability and Activity
Hypoxia Inducible Factors (HIFs) are transcription factors that are stabilized mainly in response to decreased oxygen availability. Stabilized HIF induces the expression of target genes that act to maintain biological homeostasis. The functional HIF transcription factors consist of an unstable, constitutively expressed alpha subunit (HIF-1, HIF-2 or HIF-3) and a stable, constitutively expressed beta subunit (ARNT; also known as HIF-1 beta). Of the alpha subunits, HIF-1 alpha is the most extensively studied, followed by HIF-2 alpha and HIF-3 alpha, about which the least is known. The regulation of HIF-1 alpha and HIF-2 alpha stability during normoxia and hypoxia by the von Hippel-Lindau protein (pVHL) and other ubiquitin E3 ligases is displayed below. When HIF-1 alpha and HIF-2 alpha are stable, they are able to translocate to the nucleus where they individually dimerize with ARNT to form either the HIF-1 or HIF-2 transcription factor, respectively. HIF-1 and HIF-2 bind hypoxic response elements (HREs) within the promoters of hypoxic responsive genes and interact with co-activators, including p300/CBP, to activate transcription. Hypoxic responsive genes code for proteins that are involved in angiogenesis, epithelial-to-mesenchymal transition (EMT), survival, proliferation, metastasis, glycolysis, and pluripotency. HIF-1 and HIF-2 transcriptional activity can be modulated by both repressors and enhancers. HIF transcriptional activity can be repressed via post-translational modifications and protein-protein interactions that block the binding of HIF alpha subunits to ARNT and p300/CBP. There are also post-translational modifications and protein-protein interactions that enhance the transactivation of HIF, including SUMOylation of HIF-1 alpha by Cbx4 and the binding of SART1/HAF to HIF-2 alpha. HIF-1 alpha activity in tumors correlates with increased angiogenesis and tumor growth, which has led to the investigation of HIF-1 alpha as a pharmacological target.
To learn more, please visit our HIF Transcription Factors Research Area.

