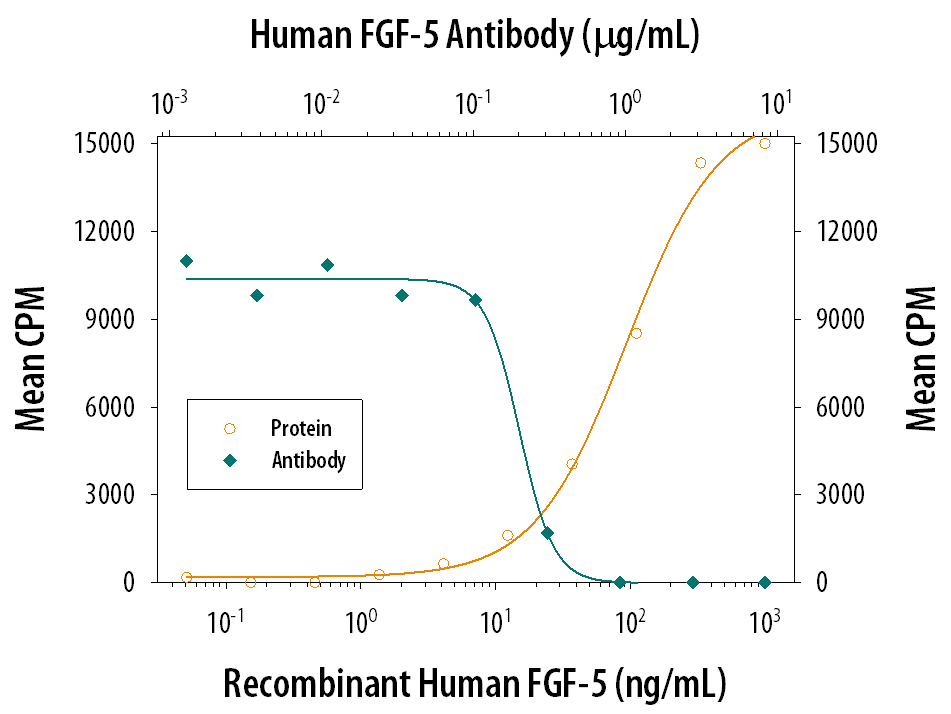
The FGF family is comprised of at least seven polypeptides that are potent regulators of cell proliferation, differentiation and function. All FGFs have two conserved cysteine residues and share 30-50% sequence homology at the amino acid level. FGF-5 was originally identified as a transforming gene by the NIH-3T3 focus formation assay using DNA derived from human tumors. FGF-5 cDNA encodes a 267 amino acid residue protein with a putative 22 amino acid residue signal peptide. The murine homologue of FGF-5 was cloned and found to be 84% homologous to the human protein at the amino acid sequence level. Human and murine FGF-5 exhibit cross species activity.
In vitro, recombinant human FGF-5 is a mitogen for Balb/3T3 fibroblasts and bovine heart endothelial cells. FGF-5 was also reported to be a major muscle-derived survival factor for cultured spinal motoneurons. In vivo, FGF-5 is suggested to play important roles in both embryology and neurobiology. Developmentally, FGF-5 mRNA is initially found in the embryoblast followed by the lateral somatic mesoderm, where it may play a role in angiogenesis, plus the myotomes cranial to the tail region, where it may delay terminal myoblast differentiation during cell migration. FGF-5 continues to impact muscle post-natally where it is believed to function as a target-derived neurotrophic factor of skeletal muscle. In the nervous system, FGF-5 has been most often identified in neurons associated with the limbic system, notably in neurons of the olfactory bulb and pyramidal cells of the hippocampus. Hippocampal FGF-5 is suggested to serve as a neurotrophic and differentiative factor for cholinergic and serotonergic neurons projecting to this region.