The Glypicans (glypiated proteoglycans) are a small multigene family of GPI-linked proteoglycans that play a key role in growth factor signaling (1, 2, 3, 4). There are six known mammalian Glypicans. They all share a common-sized protein core of 60‑70 kDa, an N-terminus which likely forms a compact globular domain, 14 conserved cysteines that form multiple intrachain disulfide bonds, and a number of C-terminal N- and O-linked carbohydrate attachment sites. Based on exon organization and the location of O-linked glycosylation sites, at least two subfamilies of Glypicans are known, with one subfamily containing Glypicans 1, 2, 4 and 6, and another subfamily containing Glypicans 3 and 5 (3, 5). Human Glypican 1 (GPC-1) is synthesized as a 558 amino acid (aa) preproprecursor that contains a 23 aa signal sequence, a 507 aa mature segment, and a 28 aa C-terminal prosegment (6, 7). There are two potential N-linked and four potential O-linked sites for glycosylation or glycanation. There are potentially two heparan sulfate (HS) modifications on GPC-1 that could contribute to a native molecular weight of approximately 200 kDa (7, 8, 9). Mature human GPC-1 shares 91% aa identity with mature mouse GPC-1. There are two potential splice variants of human GPC-1. Both show an alternate start site at Met73, while one has an additional 65 aa substitution for the C-terminal 264 amino acids (10, 11). Cells known to express GPC-1 include neurons, smooth and skeletal muscle cells, keratinocytes, osteoblasts, Schwann cells, immature dendritic cells, and tumor, plus tumor-associated vascular endothelial cells (8, 9, 12‑15). The function of GPC-1 is complex and varied. As a proteoglycan, it appears to make use of its HS adduct to impact select growth factor activity (16). This is accomplished by having juxtramembrane HS attachment sites, and a flexible, GPI-linkage (17). Data suggests GPC-1 and sulfation enzymes may collaborate to regulate FGF signaling. HS modules that are rich in 2-O- and 6-O- sulfate upregulate FGF-2 activation of FGFR1c (18). Similarly, FGF-1 requires both 2-O- and 6-O-sulfation to bind to FGFR2c and 3c. By contract, FGF-1 requires no sulfation to bind to FGFR2b, and FGF-8b needs only 6-O-sulfation to activate FGFR3c. Thus, many FGF receptor isoform specific effects may be attributed to an interaction between Glypican family members and the cell sulfation system (19).
Human Glypican 1 Antibody
R&D Systems | Catalog # AF4519


Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Asp24-Ser530
Accession # P35052
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Glypican 1 Antibody
Detection of Glypican 1 in MDA‑MB‑231 Human Cell Line by Flow Cytometry.
MDA-MB-231 human breast cancer cell line was stained with Goat Anti-Human Glypican 1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4519, filled histogram) or isotype control antibody (Catalog # AB-108-C, open histogram), followed by Phycoerythrin-conjugated Anti-Goat IgG Secondary Antibody (Catalog # F0107).Glypican 1 in MDA‑MB‑231 Human Cell Line.
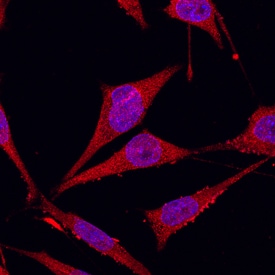
Glypican 1 was detected in immersion fixed MDA-MB-231 human breast cancer cell line using Goat Anti-Human Glypican 1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4519) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI(blue). Specific staining was localized to cytoplasmic. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Glypican 1 by Immunocytochemistry/ Immunofluorescence
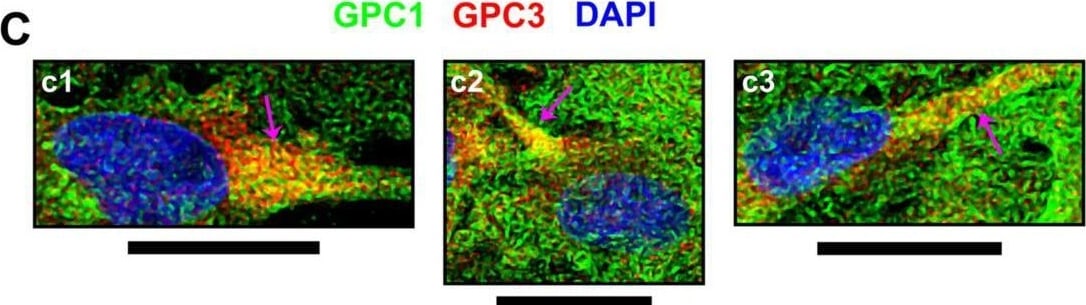
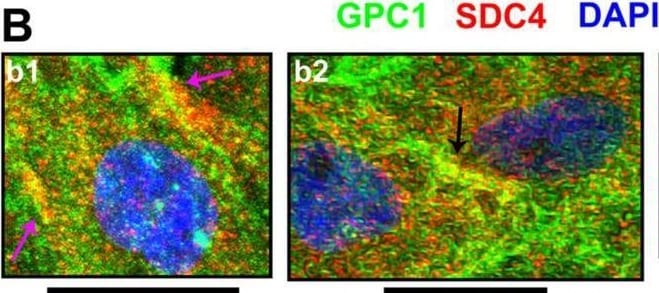
Cellular events indicating GPC1 and its association with SDC4 or GPC3.From immunofluorescence assays, through confocal microscopy, special events including (A) GPC1 and its co-localization with (B) SDC4 or (C) GPC3, both in red, were imaged. GPC1 is always represented in green, and DAPI staining for cells’ nuclei is in blue. Images were obtained from U-251 MG cells (b2, b3 and c1-c4) or C- (a1-a3 and b1) as there was evident GPC1 expression in these cell lines. Arrows have the following color code: detection in extracellular vesicles (white); extracellular vesicles’ interaction with the cellular membrane (cyan); co-localization in the cell membrane (magenta); co-localization in cell-cell junctions (black). The images were obtained with a Leica TCS SP8 CARS confocal microscope. All scale bars refer to 25 µm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32180897), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Glypican 1 by Immunocytochemistry/ Immunofluorescence
Cellular events indicating GPC1 and its association with SDC4 or GPC3.From immunofluorescence assays, through confocal microscopy, special events including (A) GPC1 and its co-localization with (B) SDC4 or (C) GPC3, both in red, were imaged. GPC1 is always represented in green, and DAPI staining for cells’ nuclei is in blue. Images were obtained from U-251 MG cells (b2, b3 and c1-c4) or C- (a1-a3 and b1) as there was evident GPC1 expression in these cell lines. Arrows have the following color code: detection in extracellular vesicles (white); extracellular vesicles’ interaction with the cellular membrane (cyan); co-localization in the cell membrane (magenta); co-localization in cell-cell junctions (black). The images were obtained with a Leica TCS SP8 CARS confocal microscope. All scale bars refer to 25 µm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/32180897), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Glypican 1 Antibody
CyTOF-ready
Flow Cytometry
Sample: MDA‑MB‑231 human breast cancer cell line
Immunocytochemistry
Sample: Immersion fixed MDA‑MB‑231 human breast cancer cell line
Western Blot
Sample: Recombinant Human Glypican 1 (Catalog # 4519-GP)
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Glypican 1
References
- Song, H.H. and J. Filmus (2002) Biochim. Biophys. Acta 1573:241.
- Fransson, L-A. et al. (2004) Cell. Mol. Life Sci. 61:1016.
- De Cat, B. and G. David (2001) Semin. Cell Dev. Biol. 12:117.
- Lamoureux, F. et al. (2007) BioEssays 29:758.
- Veugelers, M. et al. (1999) J. Biol. Chem. 274:26968.
- GenBank Accession # P35052.
- David, G. et al. (1990) J. Cell Biol. 111:3165.
- Lories, V. et al. (1992) J. Biol. Chem. 267:1116.
- Lories, V. et al. (1989) J. Biol. Chem. 264:7009.
- GenBank Accession # EAW71184.
- GenBank Accession # EAW71183.
- Chernousov, M.A. et al. (2006) J. Neurosci. 26:508.
- Wegrowski, Y. et al. (2006) Clin. Exp. Immunol. 144:485.
- Qiao, D. et al. (2003) J. Biol. Chem. 278:16045.
- Kayed, H. et al. (2006) Int. J. Oncol. 29:1139.
- Selleck, S.B. (2006) SciSTKE, April 4:pe17.
- Qiao, D. et al. (2003) J. Biol. Chem. 278:16045.
- Su, G. et al. (2006) Am. J. Pathol. 168:2014.
- Allen, B.L. and A.C. Rapraeger (2003) J. Cell Biol. 163:637.
Alternate Names
Gene Symbol
UniProt
Additional Glypican 1 Products
Product Documents for Human Glypican 1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Glypican 1 Antibody
For research use only
Related Research Areas
Citations for Human Glypican 1 Antibody
Customer Reviews for Human Glypican 1 Antibody
There are currently no reviews for this product. Be the first to review Human Glypican 1 Antibody and earn rewards!
Have you used Human Glypican 1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways
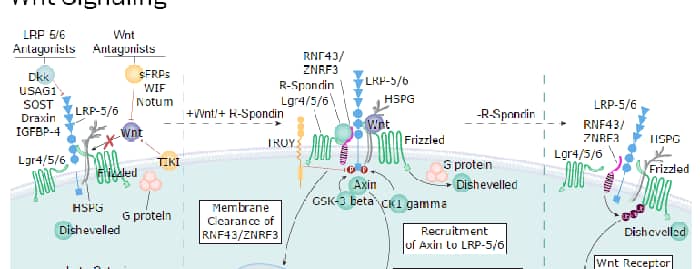
 Wnt Signaling Pathways: beta-Catenin-dependent Wnt Signaling
Wnt Signaling Pathways: beta-Catenin-dependent Wnt Signaling