The interleukin 17 (IL-17) family of cytokines, comprising six members (IL-17, IL-17B through IL-17F), are structurally related proteins with a conserved cysteine-knot structure. These proinflammatory cytokines can induce local cytokine productions and are involved in the regulation of the immune response. The cognate receptors activated by some of these cytokines have been identified (1). Interleukin-17 B Receptor (IL-17 RB), also known as IL-17Rh1, IL-17ER and EVI27, represents the second receptor of the IL-17 family of cytokines to be recognized (2-4). Human IL-17 RB cDNA encodes a 502 amino acid (aa) residue type I membrane protein with a putative 17 aa signal peptide, a 275 aa extracellular domain, a 21 aa transmembrane domain and a 189 aa cytoplasmic tail. By alternative splicing, a secreted variant of IL-17 RB has also been identified (4). Human and mouse IIL-17 RB share 76% aa sequence identity. The human IL-17 RB protein sequence is only 19.2% identical to the human IL-17 R sequence, but the two receptors share many conserved cysteine residues within their extracellular domains as well as additional conserved elements within their cytoplasmic domains. Three additional type I transmembrane receptors with homology to IL-17 R have been reported, increasing the number of the IL-17 R family members to five (5, 6). By Northern blot analysis, human IL-17 RB is highly expressed in kidneys and liver but is expressed at lower levels in testes, brain, small intestine and other endocrine tissues (2-4). The expression of IL-17 RB is significantly up-regulated under inflammatory conditions. IL-17 RB binds strongly to IL-17E and weakly to IL-17B. It does not bind IL-17, IL-17C, and IL-17F. Activation of IL-17 RB by its ligands results in the activation of nuclear factor kappa-B (2-4).
Human IL‑17 RB Antibody
R&D Systems | Catalog # MAB12072

Discontinued Product
MAB12072 has been discontinued.
View all IL-17RB products.
Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, Flow Cytometry
Cited:
Neutralization
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 938975
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived human IL-17 RB
Arg18-Gly289
Accession # Q9NRM6
Arg18-Gly289
Accession # Q9NRM6
Specificity
Detects human IL-17 RB in direct ELISAs.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Scientific Data Images for Human IL‑17 RB Antibody
Detection of Human IL‑17 RB by Western Blot.
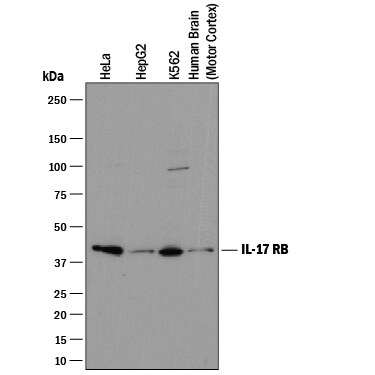
Western blot shows lysates of HeLa human cervical epithelial carcinoma cell line, HepG2 human hepatocellular carcinoma cell line, K562 human chronic myelogenous leukemia cell line, and human brain (motor cortex) tissue. PVDF membrane was probed with 2 µg/mL of Mouse Anti-Human IL-17 RB Monoclonal Antibody (Catalog # MAB12072) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF018). A specific band was detected for IL-17 RB at approximately 46 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of IL‑17 RB in HEK293 Human Cell Line Transfected with Human IL-17 RB by Flow Cytometry.
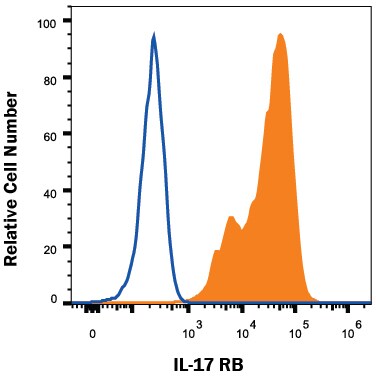
HEK293 human embryonic kidney cell line transfected with human IL-17 RB was stained with Mouse Anti-Human IL-17 RB Monoclonal Antibody (Catalog # MAB12072, filled histogram) or isotype control antibody (Catalog # MAB002, open histogram), followed by Phycoerythrin-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # F0102B). View our protocol for Staining Membrane-associated Proteins.Applications for Human IL‑17 RB Antibody
Application
Recommended Usage
Flow Cytometry
0.25 µg/106 cells
Sample: HEK293 human embryonic kidney cell line transfected with human IL-17 RB
Sample: HEK293 human embryonic kidney cell line transfected with human IL-17 RB
Western Blot
2 µg/mL
Sample: HeLa human cervical epithelial carcinoma cell line, HepG2 human hepatocellular carcinoma cell line, K562 human chronic myelogenous leukemia cell line, and human brain (motor cortex) tissue
Sample: HeLa human cervical epithelial carcinoma cell line, HepG2 human hepatocellular carcinoma cell line, K562 human chronic myelogenous leukemia cell line, and human brain (motor cortex) tissue
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-17RB
References
- Aggarwal, S. and A.L. Gurney (2002) J. Leukoc. Biol. 71:1.
- Shi Y, et al. (2000) J. Biol. Chem. 275:19167.
- Lee, J, et al. (2001) J. Biol. Chem. 276:1660.
- Tian E, et al. (2000) Oncogene 19:2098.
- Haudenschild, D. et al. (2002) J. Biol. Chem. 277:4309.
- Hurst, S.D. et al. (2002) J. Immunol. 169:443.
Long Name
Interleukin 17B Receptor
Alternate Names
Evi27, IL-17 RB, IL-17 RH1, IL-17ER, IL17RB
Gene Symbol
IL17RB
UniProt
Additional IL-17RB Products
Product Documents for Human IL‑17 RB Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IL‑17 RB Antibody
For research use only
Related Research Areas
Citations for Human IL‑17 RB Antibody
Customer Reviews for Human IL‑17 RB Antibody
There are currently no reviews for this product. Be the first to review Human IL‑17 RB Antibody and earn rewards!
Have you used Human IL‑17 RB Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Cellular Response to Hypoxia Protocols
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways
 Th2 Differentiation Pathway
Th2 Differentiation Pathway