IL-33, also known as NF-HEV and DVS 27, is a 30 kDa proinflammatory protein that may also regulate gene transcription (1‑3). DVS 27 was identifed as a gene that is up‑regulated in vasospastic cerebral arteries (1). NF-HEV was described as a nuclear factor that is preferentially expressed in the endothelial cells of high endothelial venules relative to endothelial cells from other tissues (2). IL-33 was identified based on sequence and structural homology with IL-1 family cytokines (3). DVS 27, NF-HEV, and IL-33 share 100% amino acid sequence identity. IL-33 is constitutively expressed in smooth muscle and airway epithelia. It is up‑regulated in arterial smooth muscle, dermal fibroblasts, and keratinocytes following IL-1 alpha or IL-1 beta stimulation (1, 3). Similar to IL-1, IL-33 can be cleaved in vitro by caspase-1, generating an N-terminal fragment that is slightly shorter than the C-terminal fragment (3, 4). The N-terminal portion of full length IL-33 contains a predicted bipartite nuclear localization sequence and a homeodomain-like helix-turn-helix DNA binding domain. By immunofluorescence, full length IL-33 localizes to the nucleus in HUVECs and transfectants (2). The C-terminal fragment, corresponding to mature IL-33, binds and triggers signaling through mast cell IL-1 R4/ST2L, a longtime orphan receptor involved in the augmentation of Th2 cell responses (3, 5-7). A ternary signaling complex is formed by the subsequent association of IL-33 and ST2L with IL-1R AcP (8). Stimulation of Th2 polarized lymphocytes with mature IL-33 in vitro induces IL-5 and IL-13 secretion (3). In vivo administration of mature IL-33 promotes increased production of IL-5, IL-13, IgE, and IgA, as well as splenomegaly and inflammatory infiltration of mucosal tissues (3). Full length and mature human IL-33 share 52‑58% aa sequence identity with mouse and rat IL-33. Human IL-33 shares less than 20% aa sequence identity with other IL-1 family proteins.


Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Ser112-Thr270
Accession # O95760
Specificity
Clonality
Host
Isotype
Endotoxin Level
Scientific Data Images for Human IL‑33 Antibody
Cell Proliferation Induced by IL‑33 and Neutralization by Human IL‑33 Antibody.
Recombinant Human IL-33 (3625-IL) stimulates proliferation in the D10.G4.1 mouse helper T cell line in a dose-dependent manner (orange line). Under these conditions, proliferation elicited by Recombinant Human IL-33 (1 ng/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Human IL-33 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3625). The ND50 is typically ≤ 3.0 µg/mL.IL‑33 in Human Tonsil.
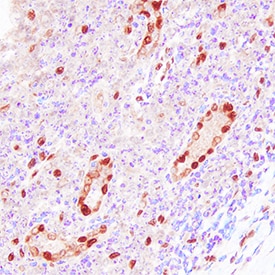
IL-33 was detected in immersion fixed paraffin-embedded sections of human tonsil using Goat Anti-Human IL-33 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3625) at 10 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to nuclei. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.IL-33 in Human Tonsil Using Dual RNAscope®ISH and IHC.
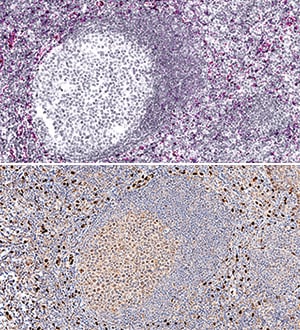
IL-33 mRNA was detected in formalin-fixed paraffin-embedded tissue sections of human tonsil probed with ACD RNAScope®Probe (Catalog # 400111) and stained using ACD RNAscope®2.5 HD Detection Reagents-Red (top image, Catalog # 32260). Adjacent tissue section was processed for immunohistochemistry using R&D Systems Goat Anti-Human IL-33 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3625) at 3 ug/mL with overnight incubation at 4 °C followed by incubation with the Anti-Goat IgG VisUCyte HRP Polymer Antibody (R&D Systems, Catalog # VC004) and DAB chromogen (lower image, yellow-brown). Tissues were counterstained with hematoxylin (blue).Detection of IL-33 by Western Blot
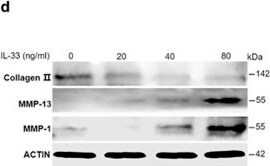
IL-33 increases MMP-1 and MMP-13 expression in chondrocytes. (a–c) Dose-dependent expression of Mmp-1 (a), Mmp-13 (b), and type II collagen (c) induced by IL-33 in human chondrocytes. (d) Western blot of MMP-1, MMP-13, and type II collagen expression in human chondrocytes induced by different doses of IL-33. (e and f) Quantification of Mmp-1 (e) and Mmp-13 (f) expression in mouse cartilage tissues 24 or 48 h after IL-33 intra-articular injection. (g) Immunohistochemical analysis of type II collagen expression in mouse articular tissues 48 h after IL-33 intra-articular injection. (h) Safranin O staining of mouse articular tissues 48 h after IL-33 intra-articular injection. Scale bar represents 100 μm. **P<0.01 and ***P<0.001. P-values were analyzed by one-way ANOVA Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/29095435), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Western Blot
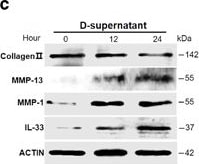
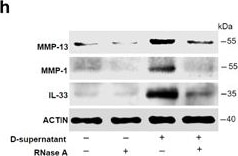
RNAs released from injured osteoarthritic cartilages induce IL-33 expression in normal human chondrocytes. (a) TUNEL immunofluorescent staining in normal and osteoarthritic cartilage tissue slices. Scale bar represents 50 μm. (b) Western blot analysis of cleaved caspase-3 in normal and osteoarthritic cartilages. Samples from four OA patients were pooled together. (c) Western blot for IL-33, MMP-1, MMP-13, and type II collagen expression in normal human chondrocytes treated with the supernatants of osteoarthritic cartilage tissue homogenates. D-supernatant represents supernatant from damaged cartilage lysate. (d–g) Quantification of Il-33 (d), Mmp-1 (e), and Mmp-13 (f) and collagen II (g) expression in normal human chondrocytes induced by the supernatant of damaged cartilage lysate at different time points. (h) Western blot analysis for IL-33, MMP-1, and MMP-13 induced by the supernatant of osteoarthritic cartilage tissue homogenates in human chondrocytes pretreated with or without 5 μg/ml RNase A. D-supernatant represents supernatant from damaged cartilage lysate. (i–k) Quantification of Il-33 (i), Mmp-1 (j), and Mmp-13 (k) expression in normal human chondrocytes induced by the supernatant of osteoarthritic cartilage tissue homogenates in human chondrocytes pretreated with or without 5 μg/ml RNase A. D-supernatant represents supernatant from damaged cartilage lysate. **P<0.01 and ***P<0.001. P-values were analyzed by one-way ANOVA Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/29095435), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Western Blot
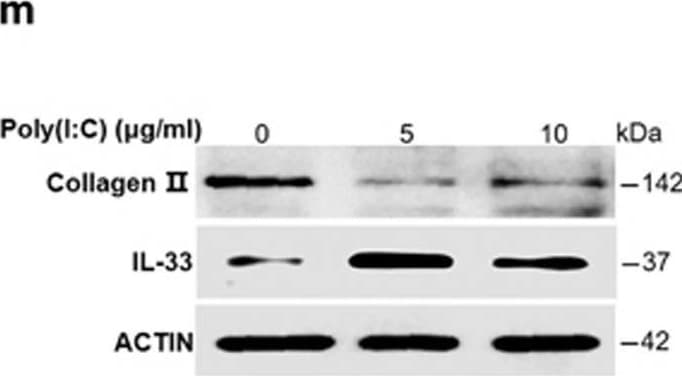
dsRNA released from damaged cartilages activates TLR3 to induce IL-33 expression. (a–d) Quantification of Il-33 (b), Mmp-1 (c), and Mmp-13 (d) expression in normal human chondrocytes induced by the supernatant of osteoarthritic cartilage tissue homogenates in human chondrocytes transfected with or without Tlr3-siRNA. D-supernatant represents supernatant from damaged cartilage lysate. (e–h) Quantification of Il-33 (f), Mmp-1 (g), and Mmp-13 (h) expression in normal human chondrocytes induced by the supernatant of osteoarthritic cartilage tissue homogenates in human chondrocytes transfected with or without Tlr7 siRNA. D-supernatant represents supernatant from damaged cartilage lysate. (i–l) Quantification of Il-33 (i), Mmp-1 (j), Mmp-13 (k) and collagen II (l) expression in normal human chondrocytes induced by different doses of poly(I:C) for 24 h. (m) Western blot analysis of IL-33 and type II collagen in human chondrocytes induced by different doses of poly(I:C) for 24 h. *P<0.05, **P<0.01, and ***P<0.001. P-values were analyzed by one-way ANOVA Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/29095435), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Western Blot
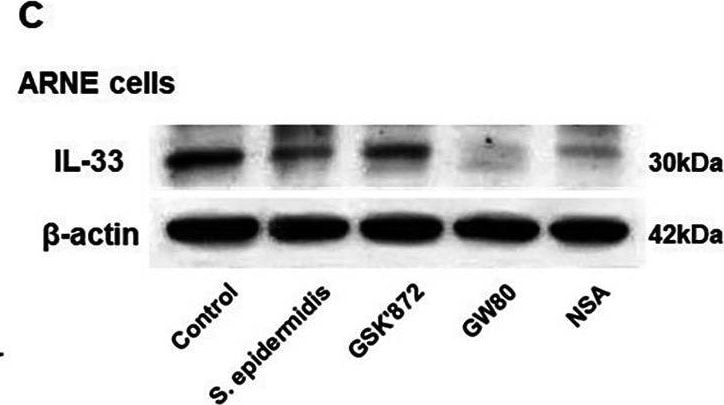
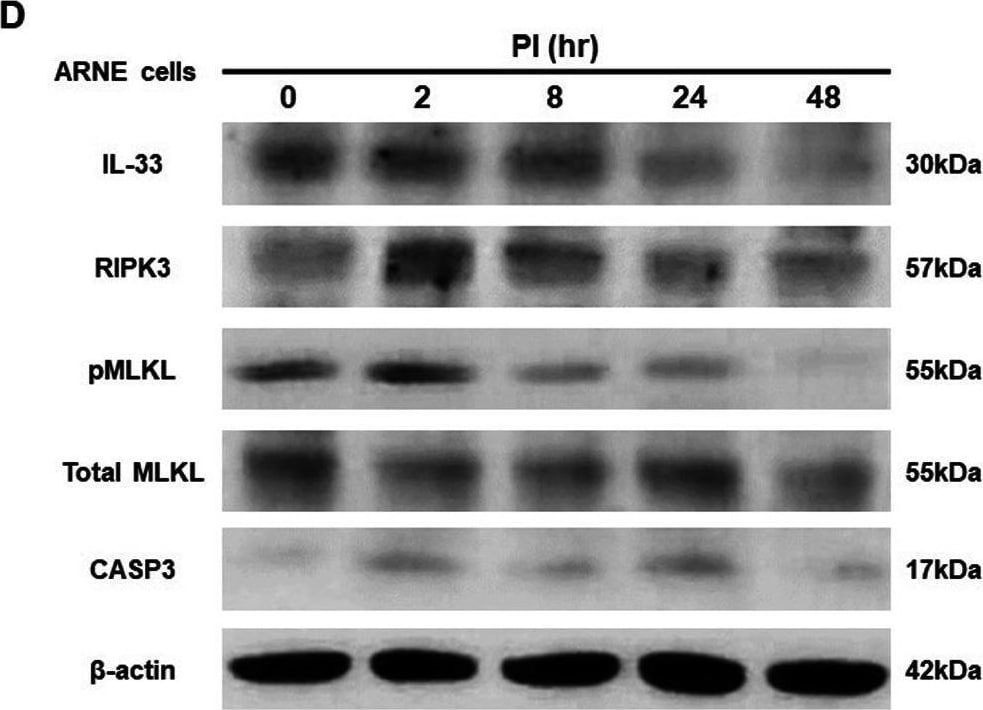
S. epidermidis reduces IL-33 production via suppression of RIPK3/MLKL-dependent necroptosis and is independent of apoptosis in allergic nasal epithelium. ARNE cells from four AR subjects were inoculated with human nasal commensal, S. epidermidis at a multiplicity of infection (MOI) of 0.25. The mRNA expression of IL-33 (A) was measured by real-time PCR, IL-33 protein concentration (B) secreted from ARNE cells was measured by ELISA using supernatants, and intracellular IL-33 protein expression (C) was also measured. after treatment of necroptosis inhibitors. Key molecules of necroptosis including RIPK3 (cropped blot) and MLKL and apoptosis including caspase3 were monitored by Western blot analysis (D). The mRNA expression of RIPK3 (E), and MLKL (F), and caspase3 (G) was measured by real-time PCR. Results are presented as mean ± SD values from four independent experiments. *p < 0.05 vs. mock-infected ARNE cells Image collected and cropped by CiteAb from the following open publication (https://bmcmicrobiol.biomedcentral.com/articles/10.1186/s12866-023-0289…), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Western Blot
RNAs released from injured osteoarthritic cartilages induce IL-33 expression in normal human chondrocytes. (a) TUNEL immunofluorescent staining in normal and osteoarthritic cartilage tissue slices. Scale bar represents 50 μm. (b) Western blot analysis of cleaved caspase-3 in normal and osteoarthritic cartilages. Samples from four OA patients were pooled together. (c) Western blot for IL-33, MMP-1, MMP-13, and type II collagen expression in normal human chondrocytes treated with the supernatants of osteoarthritic cartilage tissue homogenates. D-supernatant represents supernatant from damaged cartilage lysate. (d–g) Quantification of Il-33 (d), Mmp-1 (e), and Mmp-13 (f) and collagen II (g) expression in normal human chondrocytes induced by the supernatant of damaged cartilage lysate at different time points. (h) Western blot analysis for IL-33, MMP-1, and MMP-13 induced by the supernatant of osteoarthritic cartilage tissue homogenates in human chondrocytes pretreated with or without 5 μg/ml RNase A. D-supernatant represents supernatant from damaged cartilage lysate. (i–k) Quantification of Il-33 (i), Mmp-1 (j), and Mmp-13 (k) expression in normal human chondrocytes induced by the supernatant of osteoarthritic cartilage tissue homogenates in human chondrocytes pretreated with or without 5 μg/ml RNase A. D-supernatant represents supernatant from damaged cartilage lysate. **P<0.01 and ***P<0.001. P-values were analyzed by one-way ANOVA Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/29095435), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Western Blot
S. epidermidis reduces IL-33 production via suppression of RIPK3/MLKL-dependent necroptosis and is independent of apoptosis in allergic nasal epithelium. ARNE cells from four AR subjects were inoculated with human nasal commensal, S. epidermidis at a multiplicity of infection (MOI) of 0.25. The mRNA expression of IL-33 (A) was measured by real-time PCR, IL-33 protein concentration (B) secreted from ARNE cells was measured by ELISA using supernatants, and intracellular IL-33 protein expression (C) was also measured. after treatment of necroptosis inhibitors. Key molecules of necroptosis including RIPK3 (cropped blot) and MLKL and apoptosis including caspase3 were monitored by Western blot analysis (D). The mRNA expression of RIPK3 (E), and MLKL (F), and caspase3 (G) was measured by real-time PCR. Results are presented as mean ± SD values from four independent experiments. *p < 0.05 vs. mock-infected ARNE cells Image collected and cropped by CiteAb from the following open publication (https://bmcmicrobiol.biomedcentral.com/articles/10.1186/s12866-023-0289…), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Western Blot
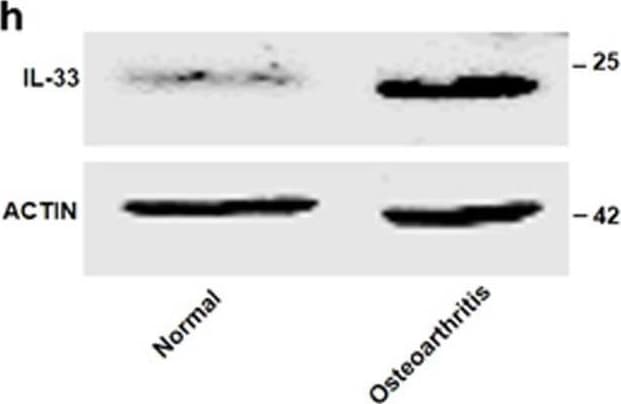
IL-33 expression is increased in the articular chondrocytes of osteoarthritic patients. (a–c) Quantification of Il-33 (a), Mmp-1 (b), and Mmp-13 (c) expression in articular tissues from normal and osteoarthritic patients. (d) Immunohistochemical analysis of IL-33 expression in articular tissue from normal and osteoarthritic patients. Scale bar represents 20 μm. (e–g) Quantification of IL-33 (e), MMP-1 (f), and MMP-13 (g) in normal and osteoarthritic cartilages detected by ELISA. (h) Western blot of IL-33 in normal and osteoarthritic cartilages. (i–k) Quantification of Il-33 (i), Mmp-1 (j), and Mmp-13 (k) expression in normal and osteoarthritic cartilages. (l–n) Quantification of IL-33 (l), MMP-1 (m), and MMP-13 (n) in synovial fluid of normal and osteoarthritic patients detected by ELISA. *P<0.05, **P<0.01, and ***P<0.001. P-values were analyzed by t-test Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/29095435), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Western Blot
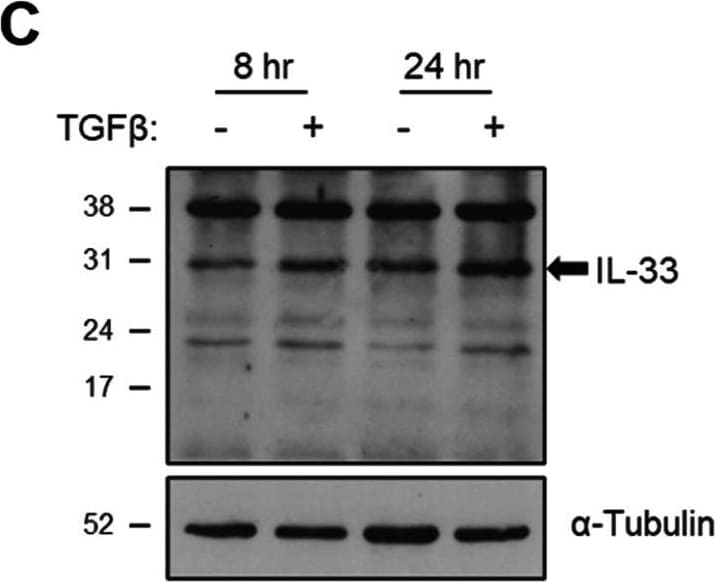
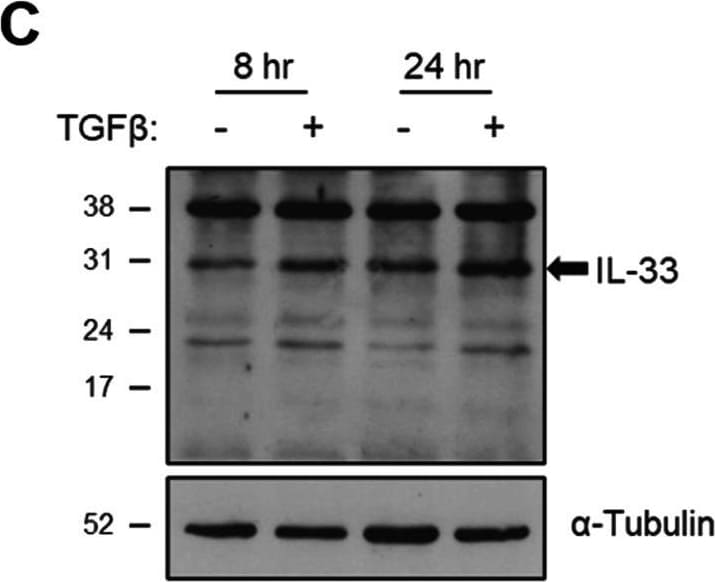
The effect of TGF beta on IL-33 expression by fibroblasts over time. Non-IPF (n = 5) and IPF (n = 5) HLFs were stimulated with 2 ng/ml of TGF beta for 4, 8 and 24 h and IL33 gene expression measured by qPCR. For each donor, all data was normalised to the unstimulated media alone control at 4 h (A). Non-IPF (n = 5) and IPF (n = 4) HLFs were stimulated with 2 ng/ml of TGF beta for 8 and 24 h, total protein isolated and 20 μg protein/lane separated by SDS-PAGE. IL-33 protein expression by non-IPF (B) and IPF (C) HLFs was assessed by western blot and quantified relative to alpha -Tubulin via densitometry (D). Representative cropped western blots from non-IPF and IPF donors are shown. Whole IL-33 blots for the representative non-IPF and IPF donors are viewable in Additional file 1: Figs. S2 and Fig. S3 respectively. IL-33 release from non-IPF (n = 3) and IPF (n = 3) HLFs treated with 2 ng /ml TGF beta for 4, 8 or 24 h was measured by ELISA. Recombinant IL-33 was titrated as a positive control (E) and analysed alongside neat HLF supernatants (F) Non-IPF and IPF donors shown in black and grey respectively. Bars indicate median values and each data point represents a single donor with statistical analysis performed by Wilcoxon signed-rank test Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36949463), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Western Blot
The effect of TGF beta on IL-33 expression by fibroblasts over time. Non-IPF (n = 5) and IPF (n = 5) HLFs were stimulated with 2 ng/ml of TGF beta for 4, 8 and 24 h and IL33 gene expression measured by qPCR. For each donor, all data was normalised to the unstimulated media alone control at 4 h (A). Non-IPF (n = 5) and IPF (n = 4) HLFs were stimulated with 2 ng/ml of TGF beta for 8 and 24 h, total protein isolated and 20 μg protein/lane separated by SDS-PAGE. IL-33 protein expression by non-IPF (B) and IPF (C) HLFs was assessed by western blot and quantified relative to alpha -Tubulin via densitometry (D). Representative cropped western blots from non-IPF and IPF donors are shown. Whole IL-33 blots for the representative non-IPF and IPF donors are viewable in Additional file 1: Figs. S2 and Fig. S3 respectively. IL-33 release from non-IPF (n = 3) and IPF (n = 3) HLFs treated with 2 ng /ml TGF beta for 4, 8 or 24 h was measured by ELISA. Recombinant IL-33 was titrated as a positive control (E) and analysed alongside neat HLF supernatants (F) Non-IPF and IPF donors shown in black and grey respectively. Bars indicate median values and each data point represents a single donor with statistical analysis performed by Wilcoxon signed-rank test Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36949463), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Immunohistochemistry
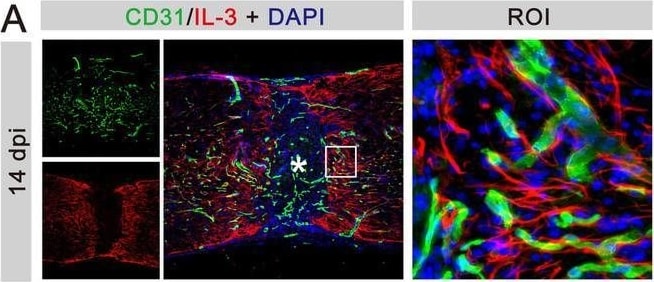
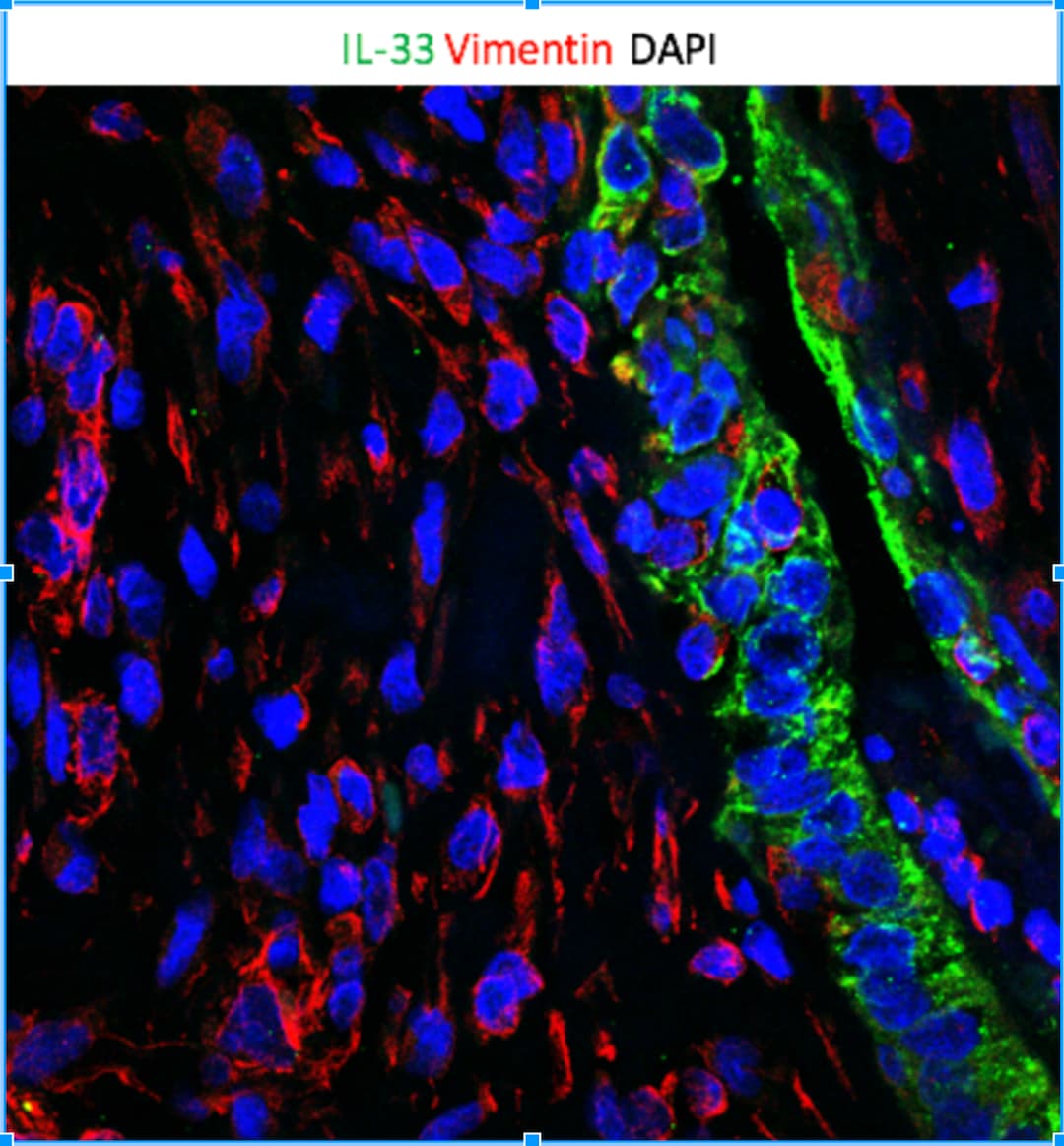
IL‐3 is specifically expressed in astrocytes at 14 dpi. (A) Immunofluorescent staining of CD31 (green), IL‐3 (red), and nuclei (blue) in sagittal sections at 14 dpi. (B) Immunofluorescent staining of PDGFR beta (green), IL‐3 (red), and nuclei (blue) in sagittal sections at 14 dpi. (C) Immunofluorescent staining of CD68 (green), IL‐3 (red), and nuclei (blue) in sagittal sections at 14 dpi. (D) Immunofluorescent staining of NG2 (green), IL‐3 (red), and nuclei (blue) in sagittal sections at 14 dpi. (E) Immunofluorescent staining of GFAP (green), IL‐3 (red), and nuclei (blue) in sagittal sections at 14 dpi. The region of interest (ROI) represents the boxed region on the left. Asterisks indicate the injured core. Scale bars: 200 μm (left panel) and 20 μm (right panel). n = 3 animals per group. (F) Quantification of the proportion of IL‐3+CD31+ cells in CD31+ cells, IL‐3+PDGFR beta + cells in PDGFR beta + cells, IL‐3+CD68+ cells in CD68+ cells, IL‐3+NG2+ cells in NG2+ cells, or IL‐3+GFAP+ cells in GFAP+ cells at 14 dpi. ND, no determined, **p < 0.01; ****p < 0.001 by one‐way ANOVA followed by Tukey's post hoc test. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/39697159), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human IL‑33 Antibody
Dual RNAscope ISH-IHC Compatible
Sample: Immersion fixed paraffin-embedded sections of human tonsil
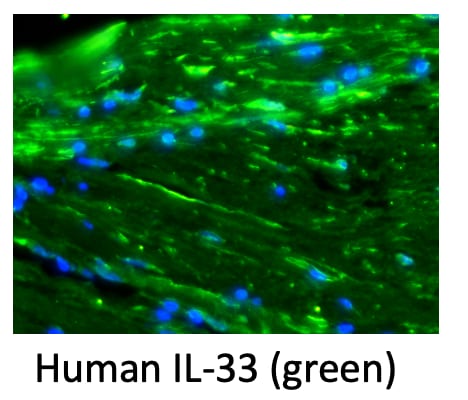
Immunocytochemistry
Sample: Immersion fixed human peripheral blood mononuclear cells treated with PMA and calcium ionomycin
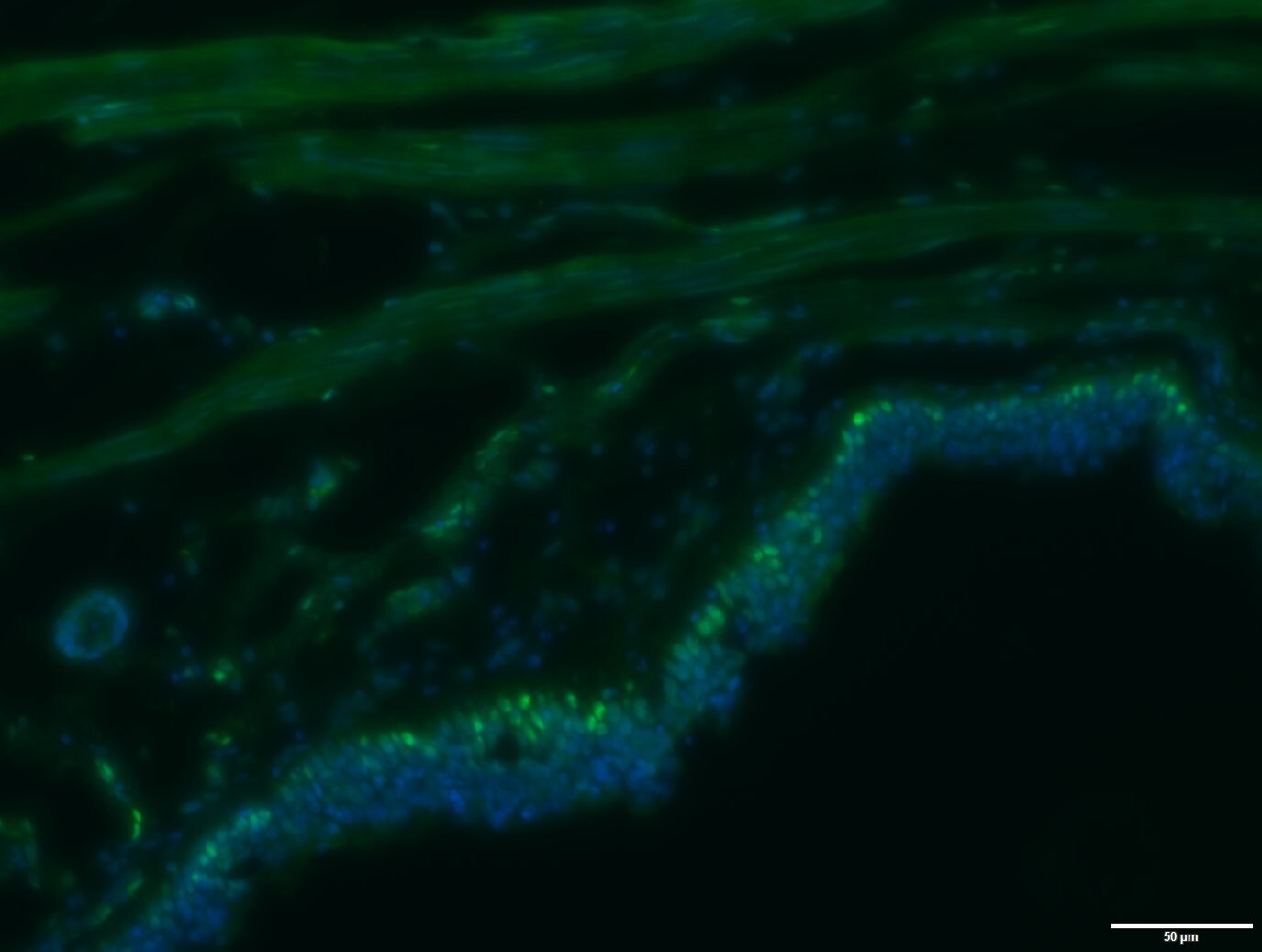
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of human tonsil
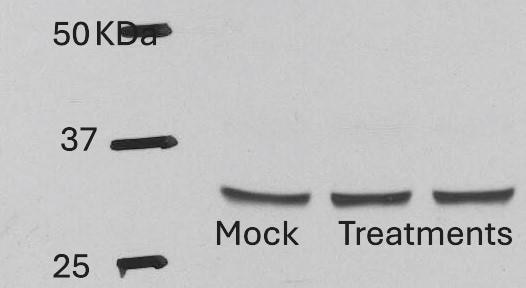
Western Blot
Sample: Recombinant Human IL‑33 (Catalog # 3625-IL)
Neutralization
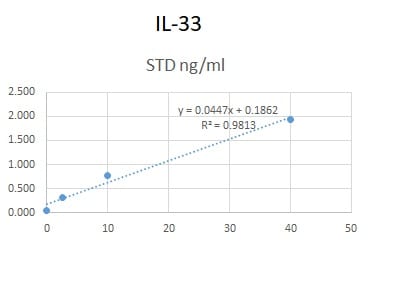
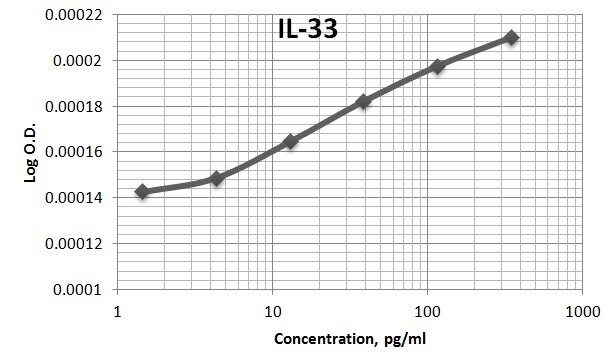
Human IL-33 Sandwich Immunoassay
Reviewed Applications
Read 8 reviews rated 4.6 using AF3625 in the following applications:
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-33
References
- Onda, H. et al. (1999) J. Cereb. Blood Flow Metab. 19:1279.
- Baekkevold, E.S. et al. (2003) Am. J. Pathol. 163:69.
- Schmitz, J. et al. (2005) Immunity 23:479.
- Black, R.A. et al. (1989) J. Biol. Chem. 264:5323.
- Xu, D. et al. (1998) J. Exp. Med. 187:787.
- Lohning, M. et al. (1998) Proc. Natl. Acad. Sci. USA 95:6930.
- Dinarello, C.A. (2005) Immunity 23:461.
- Chackerian, A.A. et al. (2007) J. Immunol. 179:2551.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional IL-33 Products
Product Documents for Human IL‑33 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IL‑33 Antibody
For research use only
Related Research Areas
Citations for Human IL‑33 Antibody
Customer Reviews for Human IL‑33 Antibody (8)
Have you used Human IL‑33 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Western BlotSample Tested: A549 human lung carcinoma cell lineSpecies: HumanVerified Customer | Posted 11/13/2024
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Aorta tissueSpecies: HumanVerified Customer | Posted 07/25/2023
-
Application: ELISASample Tested: Serum and PlasmaSpecies: HumanVerified Customer | Posted 06/09/2020We used this antibody in an in-house ELISA along with mAb (3625) and protein (3625-IL-010) to quantify IL-33 in human serum and plasma. This combination could not detect IL-33 in our samples but generated a good standard curve.
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: BRONCHUSSpecies: HumanVerified Customer | Posted 02/26/2019Staining was done on human airways after citrate antigen retrieval. OCT sections were used
-
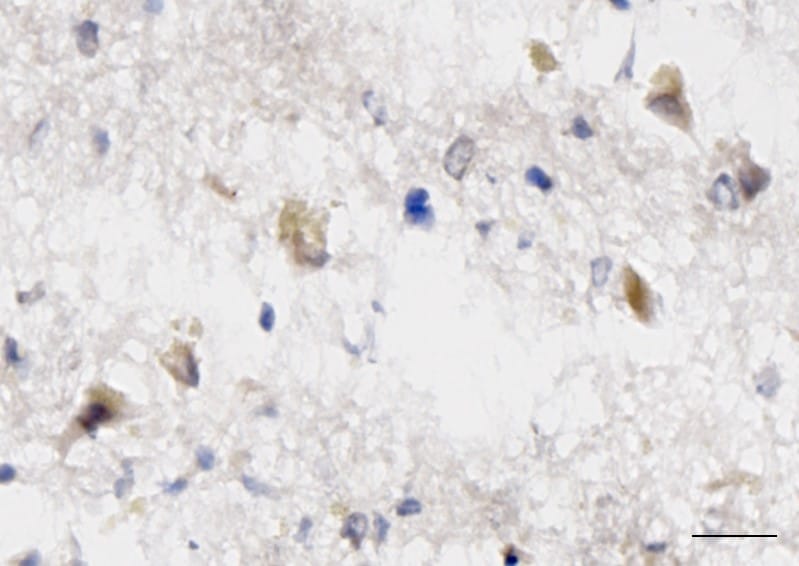
Application: ImmunohistochemistrySample Tested: Adult brainSpecies: HumanVerified Customer | Posted 03/27/2018Published in https://www.ncbi.nlm.nih.gov/pubmed/28169287 Used at 10ug/ml. Briefly, frozen brain sections were fixed in 4% PFA (Fisher Scientific), followed by antigen retrieval using heating in acid citric buffer (Vector, Burlingame, CA, USA). Endogenous avidin-biotin was blocked for 15 min (Vector). Sections were incubated with 10% horse serum in PBS (Biosera, Boussens, France) and Fc Receptor Blocking Solution was added (Human TruStain FcX Biolegend, London, UK). Primary antibodies were added overnight at 4 °C. IL-33 was detected with donkey anti-goat-biotin (ab6578, Abcam), followed by streptavidin-HRP and visualised with DAB. Section was counterstained with Hematoxylin.
-
Application: ELISASample Tested: Serum and PlasmaSpecies: HumanVerified Customer | Posted 11/09/2017AF3625 was used as both the capture and the detection antibody for the sandwich ELISA for IL-33. The immunoassay standard was 3625-IL. Assay had sensitivity of ~1pg/ml. Many healthy human samples were not detectable.
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Adult lungSpecies: HumanVerified Customer | Posted 10/18/2016
-
Application: ImmunocytochemistrySample Tested: See PMID 23172891Species: HumanVerified Customer | Posted 01/08/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- ISH-IHC Protocol for Chromogenic Detection on Formalin Fixed Paraffin Embedded (FFPE) Tissue
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for Human IL‑33 Antibody
-
Q: Does the Human IL-33 Antibody, Catalog # AF3625, detect full length IL-33 or the cleaved forms?
A: The immunogen sequence that this antibody recognizes is Ser112-Thr270 from Accession # O95760. This seqeunce corresponds to the cleaved form and is included in the full length sequence. Therefore, we would expect that this antibody detects both the full length and cleaved forms.
Associated Pathways
 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways
 Th2 Differentiation Pathway
Th2 Differentiation Pathway