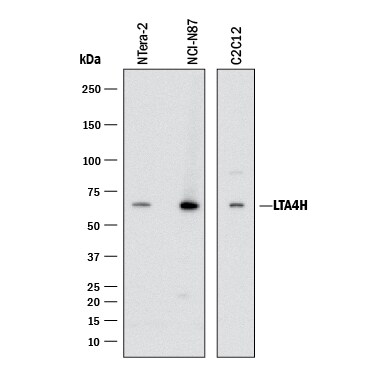
Leukotrienes are a family of lipid mediators important in a variety of allergic and inflammatory reactions. Synthesized by leukocytes, these molecules are divided into two classes, the spasmogenic cysteinyl leukotrienes and LTB4, a classical chemoattractant (1). Encoded by the LTA4H gene, Leukotriene A4 Hydrolase catalyzes the conversion of unstable epoxide LTA4 to LTB4, which is the final and committed step in LTB4 biosynthesis. As a bifunctional zinc metalloenzyme, LTA4H also acts as an arginyl aminopeptidase (2). LTA4H is a drug target for anti-inflammation, and for cancer prevention and therapy (1, 3).
The mature chain of human LTA4H consists of 610 amino acids (residues 2‑611). It is highly specific for LTA4, which also covalently modifies and inhibits the enzyme. The aminopeptidase activity is enhanced by monovalent anions (1). R&D Systems rhLTA4H corresponds to the mature chain, and is characterized by its arginyl aminopeptidase activity.