GALNT10 (N-Acetyl-Galactosaminyl Transferase 10; also UDP-Acetylgalactosaminyltransferase 10 and ppGalNAc-T10) is a member of the GalNAC transferase subfamily, glycosyltransferase 2 family of enzymes. It is widely expressed, being found in intestine, pancreas, thyroid and spleen. GALNT10 is found in the Golgi apparatus, and catalyzes the transfer of UDP-GalNAc onto either a Ser or Thr residue on a previously glycosylated peptide/polypeptide backbone. Human GALNT2 is a 603 amino acid (aa) type II transmembrane glycoprotein. It contains an 11 aa N-terminal cytoplasmic region and a 572 aa extracellular domain (aa 32-603). The ECD possesses two key parts, a catalytic region with two catalytic subdomains (aa 144-253 and 311-373), and a ricin B-type lectin domain that binds carbohydrates (aa 458-590). These two distinct domains have unique but complimentary properties. By itself, an active catalytic domain can do no more than place a GalNAc residue immediately adjacent to an existing C-terminal GalNAC residue. Alternatively, if the lectin domain is involved, this domain may recognize (bind to) any number of existing glycosylation sites, allowing for the subsequent attachment of GalNAc by the catalytic site onto a Ser or Thr residue quite distant from the initial lectin domain:CHO recognition site. There are at least three potential splice form variants. One shows a deletion of aa 190-251, a second contains a 13 aa substitution for aa 354-366, and a third possesses a 23 aa substitution for aa 1-352 coupled to a three aa substitution for Asn389. Over aa 71-603, human GALNT10 shares 96% aa sequence identity with mouse GALNT10.
Human/Mouse Polypeptide GalNAc Transferase 10/GALNT10 Antibody
R&D Systems | Catalog # AF7575

Key Product Details
Species Reactivity
Human, Mouse
Applications
Immunohistochemistry, Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Polypeptide GalNac Transferase 10/GALNT10
Leu71-Asn603
Accession # Q86SR1
Leu71-Asn603
Accession # Q86SR1
Specificity
Detects human and mouse Polypeptide GalNac Transferase 10/GALNT10 in Western blots. In direct ELISA, less than 1% cross-reactivity with recombinant human (rh) GALNT1 and rhGALNT4 is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Human/Mouse Polypeptide GalNAc Transferase 10/GALNT10 Antibody
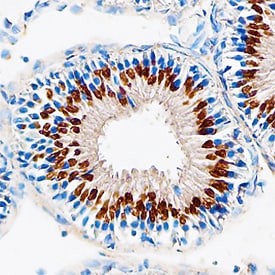
Polypeptide GalNac Transferase 10/ GALNT10 in Mouse Testis.
Polypeptide GalNac Transferase 10/GALNT10 was detected in perfusion fixed frozen sections of mouse testis using Sheep Anti-Human/ Mouse Polypeptide GalNac Transferase 10/ GALNT10 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF7575) at 5 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Sheep HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS019) and counterstained with hematoxylin (blue). Specific staining was localized to Golgi organelles in spermatocytes. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Applications for Human/Mouse Polypeptide GalNAc Transferase 10/GALNT10 Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Perfusion fixed frozen sections of mouse testis
Sample: Perfusion fixed frozen sections of mouse testis
Western Blot
Optimal dilutions of this antibody should be experimentally determined.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Polypeptide GalNAc Transferase 10/GALNT10
Long Name
UDP-GalNAc:polypeptide N-acetylgalactosaminyltransferase 10
Alternate Names
GalNAc-T10, PPGALNACT10, PPGANTASE10
Gene Symbol
GALNT10
UniProt
Additional Polypeptide GalNAc Transferase 10/GALNT10 Products
Product Documents for Human/Mouse Polypeptide GalNAc Transferase 10/GALNT10 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse Polypeptide GalNAc Transferase 10/GALNT10 Antibody
For research use only
Related Research Areas
Citations for Human/Mouse Polypeptide GalNAc Transferase 10/GALNT10 Antibody
Customer Reviews for Human/Mouse Polypeptide GalNAc Transferase 10/GALNT10 Antibody
There are currently no reviews for this product. Be the first to review Human/Mouse Polypeptide GalNAc Transferase 10/GALNT10 Antibody and earn rewards!
Have you used Human/Mouse Polypeptide GalNAc Transferase 10/GALNT10 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
