Human High-mobility group box 1 protein (HMGB1), previously known as HMG-1 or amphoterin, is a member of the high mobility group box family of non-histone chromosomal proteins (1‑3). Human HMGB1 is expressed as a 30 kDa, 215 amino acid (aa) single chain polypeptide containing three domains: two N-terminal globular, 70 aa positively charged DNA-binding domains (HMG boxes A and B), and a negatively charged 30 aa C-terminal region that contains only Asp and Glu (4, 5). Residues 27‑43 and 178‑184 contain a NLS. Posttranslational modifications of the molecule have been reported, with acetylation occurring on as many as 17 lysine residues (6). HMGB1 is expressed at high levels in almost all cells (2, 4). It was originally discovered as a nuclear protein that could bend DNA. Such bending stabilizes nucleosome formation and regulates the expression of select genes upon recruitment by DNA binding proteins (1, 7, 8). It is now known that HMGB1 can also act extracellularly, both as an inflammatory mediator that promotes monocyte migration and cytokine secretion, and as a mediator of T cell-dendritic cell interaction (1, 4, 7, 9, 10). The cytokine activity of HBMG1 is restricted to the HMG B box, (3) while the A box is associated with the helix-loop-helix domain of transcription factors (11). HMBG1 is released in response to cell death and as a secretion product. Although HMBG-1 does not possess a classic signal sequence, it appears to be secreted as an acetylated form via secretory endolysosome exocytosis (6, 12). Once secreted, HMGB1 transduces cellular signals through its high affinity receptor, RAGE and, possibly, TLR2 and TLR4 (1, 3, 4). Human HMGB1 is 100% aa identical to canine HMGB1 and 99% aa identical to mouse, rat, bovine and porcine HMGB1, respectively.
Human/Mouse/Rat HMGB1/HMG‑1 Antibody
R&D Systems | Catalog # MAB16901

Key Product Details
Species Reactivity
Validated:
Human, Mouse, Rat
Cited:
Mouse, Rat
Applications
Validated:
Immunohistochemistry, Western Blot, Simple Western
Cited:
Immunohistochemistry-Paraffin, Western Blot, In vivo assay
Label
Unconjugated
Antibody Source
Monoclonal Rat IgG2B Clone # 951420
Loading...
Product Specifications
Immunogen
Human HMGB1/HMG-1 synthetic peptide between amino acids Lys50-Ser100
Accession # P09429
Accession # P09429
Specificity
Detects human, mouse, and rat HMGB1/HMG-1 in Western blot.
Clonality
Monoclonal
Host
Rat
Isotype
IgG2B
Scientific Data Images for Human/Mouse/Rat HMGB1/HMG‑1 Antibody
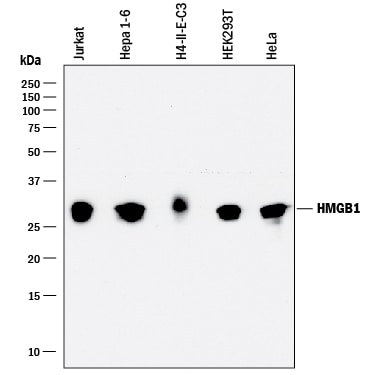
Detection of Human, Mouse, and Rat HMGB1/HMG‑1 by Western Blot.
Western blot shows lysates of Jurkat human acute T cell leukemia cell line, Hepa 1-6 mouse hepatoma cell line, H4-II-E-C3 rat hepatoma cell line, HEK293 human embryonic kidney cell line, and HeLa human cervical epithelial carcinoma cell line. PVDF membrane was probed with 0.1 µg/mL of Rat Anti-Human HMGB1/HMG-1 Monoclonal Antibody (Catalog # MAB16901) followed by HRP-conjugated Anti-Rat IgG Secondary Antibody (HAF005). A specific band was detected for HMGB1/HMG-1 at approximately 28 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.HMGB1/HMG‑1 in Human Prostate Cancer Tissue.
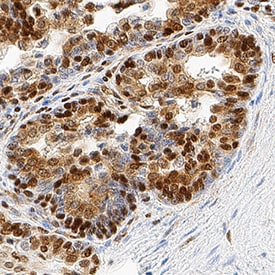
HMGB1/HMG-1 was detected in immersion fixed paraffin-embedded sections of human prostate cancer tissue using Rat Anti-Human/Mouse/Rat HMGB1/HMG-1 Monoclonal Antibody (Catalog # MAB16901) at 5 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Rat HRP-DAB Cell & Tissue Staining Kit (brown; CTS017) and counterstained with hematoxylin (blue). Specific staining was localized to nuclei and cytoplasm. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human and Mouse HMGB1/HMG‑1 by Simple WesternTM.
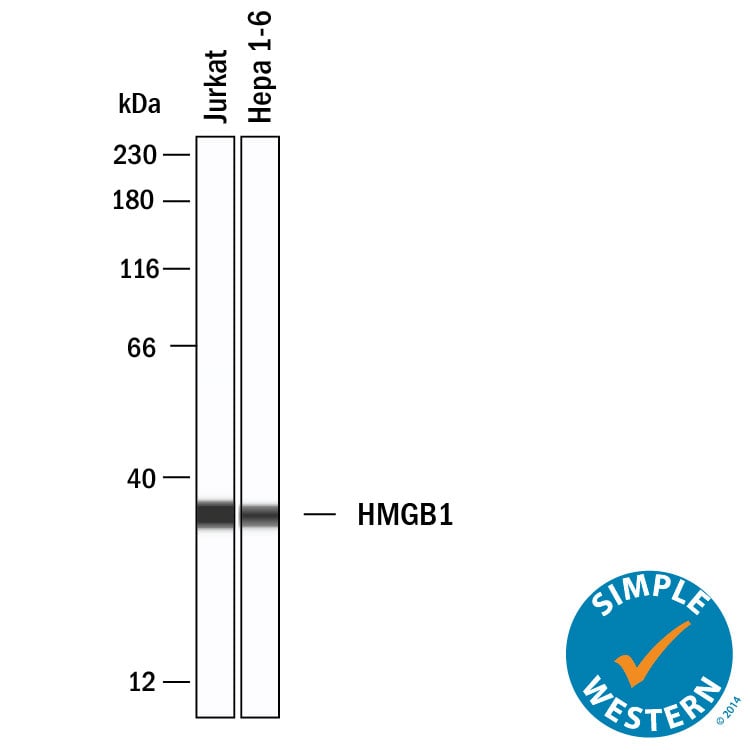
Simple Western shows lysates of Jurkat human acute T cell leukemia cell line and Hepa 1‑6 mouse hepatoma cell line, loaded at 0.2 mg/ml. A specific band was detected for HMGB1/HMG‑1 at approximately 35 kDa (as indicated) using 20 µg/mL of Rat Anti-Human/Mouse/Rat HMGB1/HMG‑1 Monoclonal Antibody (Catalog # MAB16901). This experiment was conducted under reducing conditions and using the 12-230kDa separation system.Detection of HMGB1/HMG-1 by Immunocytochemistry/ Immunofluorescence
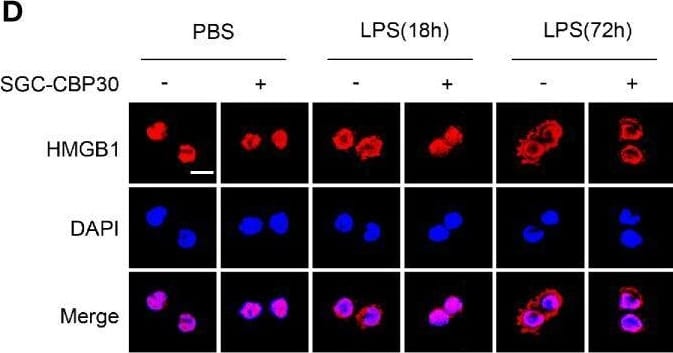
Inhibitory effect of SGC-CBP30 on HMGB1 release and expression in LPS-stimulated cells. THP-1 cells (A) and primary MPM cells (B) were treated with SGC-CBP30 (4 μM) at 8 h after stimulation with LPS (500 ng/ml). After LPS stimulation for the indicated time, HMGB1 release was measured by ELISA. THP-1 cells were treated with LPS (500 ng/ml) for 8 h and then incubated with SGC-CBP30 (4 μM). After the indicated time of LPS stimulation, nuclear and cytoplasmic fractions were analyzed by Western blot using anti-HMGB1 antibody (C). Cells were incubated with mouse anti-HMGB1 antibody and then incubated with Alexa Flour 555-conjugated anti-mouse (red) secondary antibody. The nuclei were counterstained with DAPI (blue). The location of HMGB1 was observed under a confocal laser microscope. Scale bar: 10 μm (D). The cell lysates were immunoprecipitated with anti-HMGB1 antibody, followed by immunoblotting with anti-acetyl lysine and anti-HMGB1 antibodies (E). Quantification of HMGB1 transcripts by real-time PCR with GAPDH as the internal control (F). Whole cell lysates were subjected to immunoblotting with anti-HMGB1 and anti-beta -actin antibodies (G). Data shown were representative of three independent experiments. Error bars indicate mean ± SD. *p < 0.05; **p < 0.01; ***p < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/33603756), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse/Rat HMGB1/HMG‑1 Antibody
Application
Recommended Usage
Immunohistochemistry
5-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human prostate cancer tissue
Sample: Immersion fixed paraffin-embedded sections of human prostate cancer tissue
Simple Western
20 µg/mL
Sample: Jurkat human acute T cell leukemia cell line and Hepa 1-6 mouse hepatoma cell line
Sample: Jurkat human acute T cell leukemia cell line and Hepa 1-6 mouse hepatoma cell line
Western Blot
0.1 µg/mL
Sample: Jurkat human acute T cell leukemia cell line, Hepa 1‑6 mouse hepatoma cell line, H4‑II‑E‑C3 rat hepatoma cell line, HEK293 human embryonic kidney cell line, and HeLa human cervical epithelial carcinoma cell line
Sample: Jurkat human acute T cell leukemia cell line, Hepa 1‑6 mouse hepatoma cell line, H4‑II‑E‑C3 rat hepatoma cell line, HEK293 human embryonic kidney cell line, and HeLa human cervical epithelial carcinoma cell line
Reviewed Applications
Read 1 review rated 5 using MAB16901 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: HMGB1/HMG-1
References
- Lotze, M.T. and K.J. Tracey (2005) Nat. Rev. Immunol. 5:331.
- Yang, H. et al. (2005) J. Leukoc. Biol. 78:1.
- Dumitriu, I.E. et al. (2005) Trends Immunol. 26:381.
- Degryse, B. and M. de Virgilio (2003) FEBS Lett. 553:11.
- Wen, L. et al. (1989) Nucleic Acids Res. 17:1197.
- Bonaldi, T. et al. (2003) EMBO J. 22:5551.
- Muller, S. et al. (2001) EMBO J. 20:4337.
- Bustin, M. (1999) Mol. Cell. Biol. 19:5237.
- Wang, H. et al. (1999) Science. 285:248.
- Dimitriu, I.E. et al. (2005) J. Immunol. 174:7506.
- Najima, Y. et al. (2005) J. Biol. Chem. 280:27523.
- Gardella, S. et al. (2002) EMBO Rep. 3:995.
Long Name
High Mobility Group Protein 1
Alternate Names
HMG-1, HMG1
Entrez Gene IDs
3146 (Human)
Gene Symbol
HMGB1
UniProt
Additional HMGB1/HMG-1 Products
Product Documents for Human/Mouse/Rat HMGB1/HMG‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse/Rat HMGB1/HMG‑1 Antibody
For research use only
Related Research Areas
Citations for Human/Mouse/Rat HMGB1/HMG‑1 Antibody
Customer Reviews for Human/Mouse/Rat HMGB1/HMG‑1 Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human/Mouse/Rat HMGB1/HMG‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
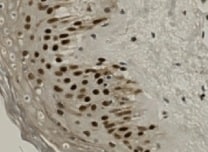
Application: ImmunohistochemistrySample Tested: Skin tissueSpecies: HumanVerified Customer | Posted 10/24/2021
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...