LMW-PTP (Low Molecular Weight Protein Tyrosine Phosphatase; also Red cell acid phosphatase 1/ACP1, HCPTP, and HAAP/Adipocyte acid phosphatase) is a 17‑18 kDa member of the LMWPTP family, PTP superfamily of enzymes. It is widely expressed, being found in cells diverse as B and T cells, endothelial cells and vascular smooth muscle cells. LMW-PTP dephosphorylates multiple substrates, often in a cell-dependent manner. Among the substrates for LMW-PTP include PDGR beta, EphA2 and beta -catenin. PMW-PTP is found intracellularly, generally associated with two cytoplasmic pools, one in the cytosol and the other accompaning the cytoskeleton. Human LMW-PTP is 158 amino acids (aa) in length. The catalytic region spans aa 9-154, and there are two acetylation sites at Ala2 and Lys156. Phosphorylation on Tyr132 and Tyr133 occurs on cytoskeleton-associated LMW-PTP, and has the effect of increasing its phosphatase activity. There are three alternative splice variants reported for LMW-PTP. One shows a 34 aa substitution for aa 41-74, a second shows a deletion of aa 41-74, while a third contains a 35 aa substitution for aa 78-158. The last two isoforms may function a negative regulators of LMW-PTP activity. Full-length LMW-PTP shares 86% aa sequence identity with mouse LMW-PTP.

Key Product Details
Species Reactivity
Human, Mouse, Rat
Applications
Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human LMW‑PTP/ACP1
Ala2-His158
Accession # P24666
Ala2-His158
Accession # P24666
Specificity
Detects human, mouse, and rat LMW‑PTP/ACP1 in Western blots.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Human/Mouse/Rat LMW‑PTP/ACP1 Antibody
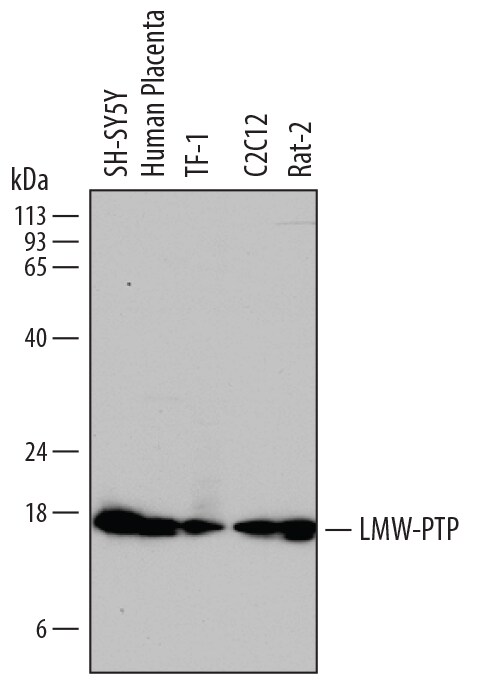
Detection of Human, Mouse, and Rat LMW‑PTP/ACP1 by Western Blot.
Western blot shows lysates of SH-SY5Y human neuroblastoma cell line, human placenta tissue, TF-1 human erythroleukemic cell line, C2C12 mouse myoblast cell line, and Rat-2 rat embryonic fibroblast cell line. PVDF membrane was probed with 1 µg/mL of Sheep Anti-Human/Mouse/Rat LMW-PTP/ACP1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF5075) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). A specific band was detected for LMW-PTP/ACP1 at approximately 18 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Applications for Human/Mouse/Rat LMW‑PTP/ACP1 Antibody
Application
Recommended Usage
Western Blot
1 µg/mL
Sample: SH‑SY5Y human neuroblastoma cell line, human placenta tissue, TF‑1 human erythroleukemic cell line, C2C12 mouse myoblast cell line, and Rat‑2 rat embryonic fibroblast cell line
Sample: SH‑SY5Y human neuroblastoma cell line, human placenta tissue, TF‑1 human erythroleukemic cell line, C2C12 mouse myoblast cell line, and Rat‑2 rat embryonic fibroblast cell line
Reviewed Applications
Read 1 review rated 5 using AF5075 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: LMW-PTP/ACP1
Long Name
Low Molecular Weight Phosphotyrosine Protein Phosphotase
Alternate Names
ACP1, HAAP, LMWPTP
Gene Symbol
ACP1
UniProt
Additional LMW-PTP/ACP1 Products
Product Documents for Human/Mouse/Rat LMW‑PTP/ACP1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse/Rat LMW‑PTP/ACP1 Antibody
For research use only
Related Research Areas
Customer Reviews for Human/Mouse/Rat LMW‑PTP/ACP1 Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human/Mouse/Rat LMW‑PTP/ACP1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
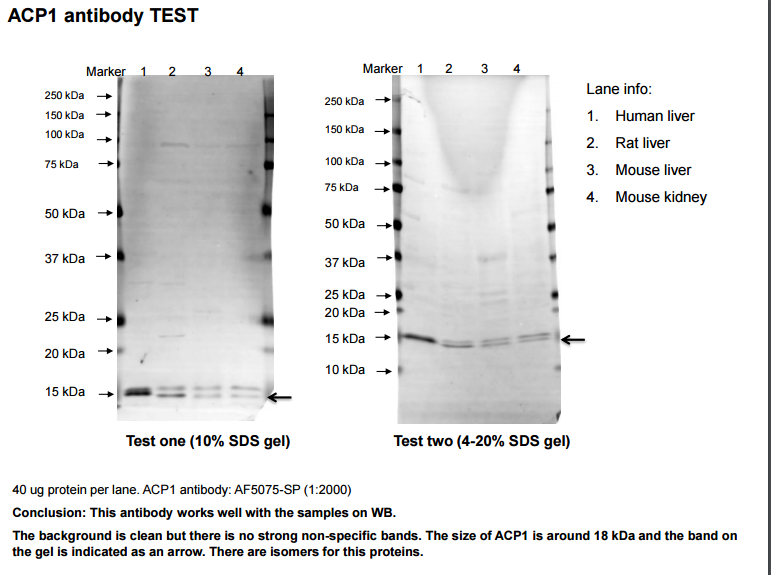
Application: Western BlotSample Tested: Liver and Adult kidneySpecies: Human, Mouse and RatVerified Customer | Posted 02/24/2017
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...