Vimentin is a 57 kDa class III intermediate filament (IF) protein that belongs to the intermediate filament family. It is the predominant IF in cells of mesenchymal origin such as vascular endothelium and blood cells (1-3). The human Vimentin cDNA encodes a 466 amino acid (aa) protein that contains head and tail regions with multiple regulatory Ser/Thr phosphorylation sites, and a central rod domain with three coiled-coil regions separated by linkers (1, 2). Human Vimentin shares 97-98% aa identity with mouse, rat, ovine, bovine, and canine Vimentin. Sixteen Vimentin coiled-coil dimers self-assemble to form intermediate (10-12 nm wide) filaments (4). These filaments then anneal longitudinally to form non-polarized fibers that support cell structure and withstand stress (4). IF fibers are highly dynamic, and half-life depends on the balance between kinase and phosphatase activity. For example, phosphorylation followed by dephosphorylation drives IF disintegration, followed by reorganization during mitosis (1, 5, 6). Interactions of head and tail domains link IFs with other structures such as actin and microtubule cytoskeletons (7). Vimentin is involved in positioning autophagosomes, lysosomes and the Golgi complex within the cell (8). It facilitates cell migration and motility by recycling internalized trailing edge integrins back to the cell surface at the leading edge (9-11). Vimentin helps maintain the lipid composition of cellular membranes, and caspase cleavage of Vimentin is a key event in apoptosis (8, 12). Phosphorylation promotes secretion of Vimentin by TNF-alpha -stimulated macrophages (13). Extracellular Vimentin has been shown to associate with several microbes, and appears to promote an antimicrobial oxidative burst (13, 14). Cell-associated Vimentin can also interact with NKp46 to recruit NK cells to tuberculosis-infected monocytes (15).
Human/Mouse/Rat Vimentin Biotinylated Antibody
R&D Systems | Catalog # BAM2105

Key Product Details
Species Reactivity
Human, Mouse, Rat
Applications
Immunocytochemistry
Label
Biotin
Antibody Source
Monoclonal Rat IgG2A Clone # 280618
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human Vimentin
Ser2-Glu466
Accession # P08670
Ser2-Glu466
Accession # P08670
Specificity
Vimentin antibodies are ideal for immunocytochemistry colocalization studies in intermediate filaments. The unconjugated antibody detects human Vimentin in Western blots.
Clonality
Monoclonal
Host
Rat
Isotype
IgG2A
Scientific Data Images for Human/Mouse/Rat Vimentin Biotinylated Antibody
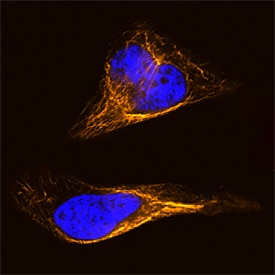
Vimentin in HeLa Human Cell Line.
Vimentin was detected in formaldehyde fixed HeLa human cervical epithelial carcinoma cell line using Rat Anti-Human/Mouse/Rat Vimentin Biotinylated Monoclonal Antibody (Catalog # BAM2105) at 8 µg/mL overnight at 4 ° C. Cells were stained using the NorthernLights™ 557-conjugated Streptavidin (orange; Catalog # NL999) and counterstained with DAPI (blue). Specific staining was localized to intermediate filaments. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Mouse Vimentin by Immunohistochemistry
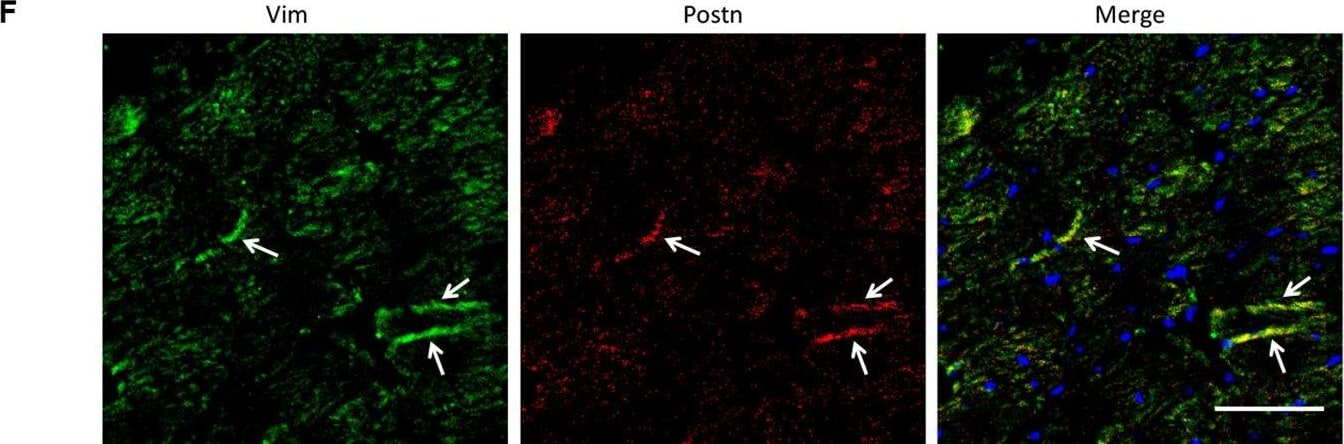
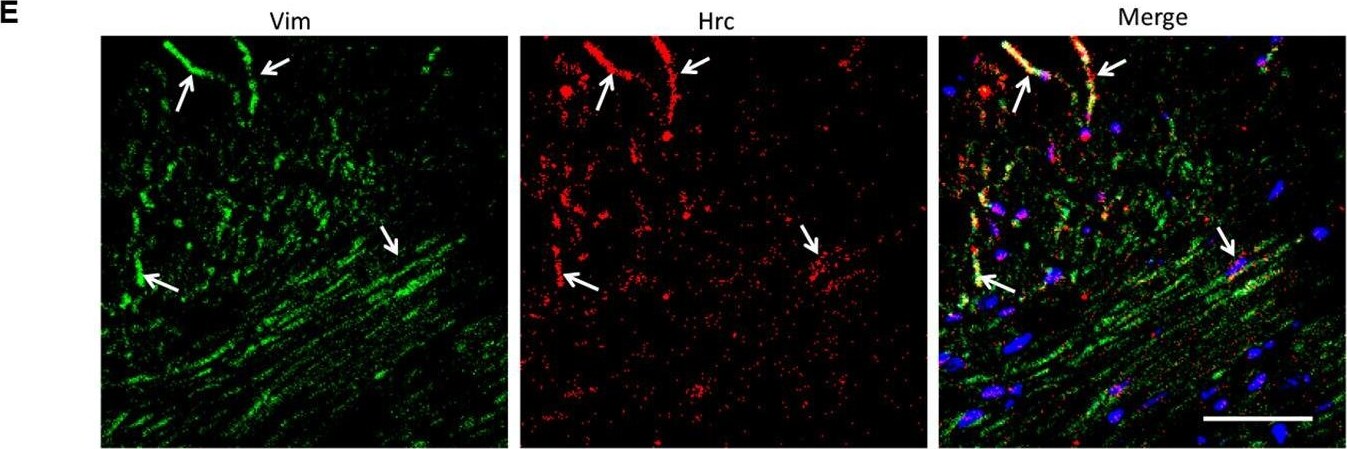
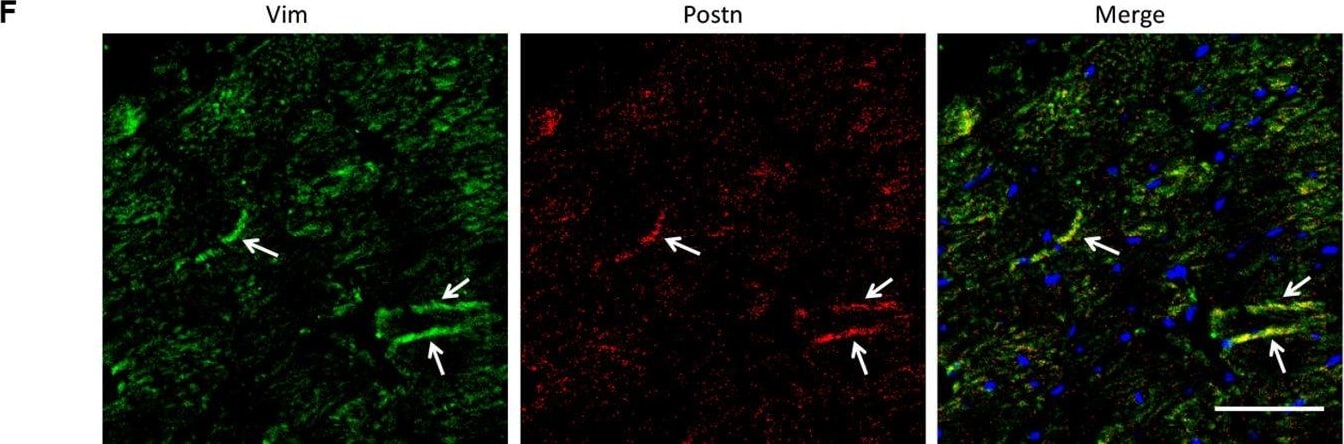
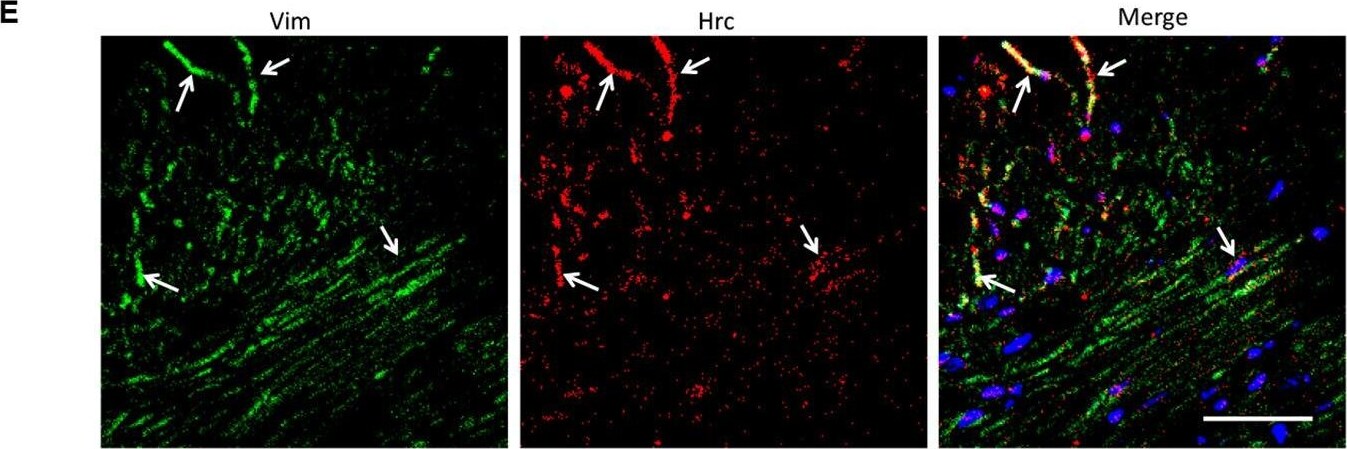
Analysis of the heterogeneity of the fibroblast subpopulations.(A) t-SNE plot of the 10 cardiac fibroblast subpopulations in the high-fat diet (HFD)/streptozotocin (STZ)-treated and control mice (n = 3428 fibroblasts from healthy control and n = 2988 fibroblasts from 6 diabetic mice). (B) Correlation heatmap of the gene-expression signatures of each fibroblast subpopulation. Color differences indicate subpopulations that were resolved through multiscale bootstrapping. (C) Heatmap showing the top 5 marker genes in each fibroblast subpopulation. The red color indicates high expression, while green color indicates low expression. (D) Heatmap showing the enriched Gene Ontology (GO) terms associated with cardiac fibrosis in each fibroblast population. (E, F) Representative immunofluorescence images of Hrc (E) and Postn (F) in mouse heart (n = 6 mice per group), scale bar = 100 µm. The details of the 10 transcriptionally distinct fibroblast subpopulations are listed in Supplementary file 15. The details of distinct signatures of each fibroblast subpopulation in the heart are listed in Supplementary file 16. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37010266), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Vimentin by Immunohistochemistry
Analysis of the heterogeneity of the fibroblast subpopulations.(A) t-SNE plot of the 10 cardiac fibroblast subpopulations in the high-fat diet (HFD)/streptozotocin (STZ)-treated and control mice (n = 3428 fibroblasts from healthy control and n = 2988 fibroblasts from 6 diabetic mice). (B) Correlation heatmap of the gene-expression signatures of each fibroblast subpopulation. Color differences indicate subpopulations that were resolved through multiscale bootstrapping. (C) Heatmap showing the top 5 marker genes in each fibroblast subpopulation. The red color indicates high expression, while green color indicates low expression. (D) Heatmap showing the enriched Gene Ontology (GO) terms associated with cardiac fibrosis in each fibroblast population. (E, F) Representative immunofluorescence images of Hrc (E) and Postn (F) in mouse heart (n = 6 mice per group), scale bar = 100 µm. The details of the 10 transcriptionally distinct fibroblast subpopulations are listed in Supplementary file 15. The details of distinct signatures of each fibroblast subpopulation in the heart are listed in Supplementary file 16. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37010266), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Vimentin by Immunohistochemistry
Analysis of the heterogeneity of the fibroblast subpopulations.(A) t-SNE plot of the 10 cardiac fibroblast subpopulations in the high-fat diet (HFD)/streptozotocin (STZ)-treated and control mice (n = 3428 fibroblasts from healthy control and n = 2988 fibroblasts from 6 diabetic mice). (B) Correlation heatmap of the gene-expression signatures of each fibroblast subpopulation. Color differences indicate subpopulations that were resolved through multiscale bootstrapping. (C) Heatmap showing the top 5 marker genes in each fibroblast subpopulation. The red color indicates high expression, while green color indicates low expression. (D) Heatmap showing the enriched Gene Ontology (GO) terms associated with cardiac fibrosis in each fibroblast population. (E, F) Representative immunofluorescence images of Hrc (E) and Postn (F) in mouse heart (n = 6 mice per group), scale bar = 100 µm. The details of the 10 transcriptionally distinct fibroblast subpopulations are listed in Supplementary file 15. The details of distinct signatures of each fibroblast subpopulation in the heart are listed in Supplementary file 16. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37010266), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Vimentin by Immunohistochemistry
Analysis of the heterogeneity of the fibroblast subpopulations.(A) t-SNE plot of the 10 cardiac fibroblast subpopulations in the high-fat diet (HFD)/streptozotocin (STZ)-treated and control mice (n = 3428 fibroblasts from healthy control and n = 2988 fibroblasts from 6 diabetic mice). (B) Correlation heatmap of the gene-expression signatures of each fibroblast subpopulation. Color differences indicate subpopulations that were resolved through multiscale bootstrapping. (C) Heatmap showing the top 5 marker genes in each fibroblast subpopulation. The red color indicates high expression, while green color indicates low expression. (D) Heatmap showing the enriched Gene Ontology (GO) terms associated with cardiac fibrosis in each fibroblast population. (E, F) Representative immunofluorescence images of Hrc (E) and Postn (F) in mouse heart (n = 6 mice per group), scale bar = 100 µm. The details of the 10 transcriptionally distinct fibroblast subpopulations are listed in Supplementary file 15. The details of distinct signatures of each fibroblast subpopulation in the heart are listed in Supplementary file 16. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37010266), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse/Rat Vimentin Biotinylated Antibody
Application
Recommended Usage
Immunocytochemistry
8-25 µg/mL
Sample: Formaldehyde fixed HeLa human cervical epithelial carcinoma cell line
Sample: Formaldehyde fixed HeLa human cervical epithelial carcinoma cell line
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein.
Shipping
The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Vimentin
References
- Omary, M.B. et al. (2006) Trends Biochem. Sci. 31:383.
- Ivaska, J. et al. (2007) Exp. Cell Res. 313:2050.
- Ferrari, S. et al. (1986) Mol. Cell. Biol. 6:3614.
- Sokolova, A.V. et al. (2006) Proc. Natl. Acad. Sci. USA 103:16206.
- Eriksson, J.E. et al. (2004) J. Cell Sci. 117:919.
- Li, Q-F. et al. (2006) J. Biol. Chem. 281:34716.
- Esue, O. et al. (2006) J. Biol. Chem. 281:30393.
- Styers, M.L. et al. (2005) Traffic 6:359.
- McInroy, L. and A. Maata (2007) Biochem. Biophys. Res. Commun. 360:109.
- Nieminen, M. et al. (2006) Nat. Cell Biol. 8:156.
- Ivaska, J. et al. (2005) EMBO J. 24:3834.
- Byun, Y. et al. (2001) Cell Death Differ. 8:443.
- Mor-Vaknin, N. et al. (2003) Nat. Cell Biol. 5:59.
- Zou, Y. et al. (2006) Biochem. Biophys. Res. Commun. 351:625.
- Garg, A. et al. (2006) J. Immunol. 177:6192.
Alternate Names
VIM
Gene Symbol
VIM
UniProt
Additional Vimentin Products
Product Documents for Human/Mouse/Rat Vimentin Biotinylated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse/Rat Vimentin Biotinylated Antibody
For research use only
Citations for Human/Mouse/Rat Vimentin Biotinylated Antibody
Customer Reviews for Human/Mouse/Rat Vimentin Biotinylated Antibody
There are currently no reviews for this product. Be the first to review Human/Mouse/Rat Vimentin Biotinylated Antibody and earn rewards!
Have you used Human/Mouse/Rat Vimentin Biotinylated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- View all Protocols, Troubleshooting, Illustrated assays and Webinars