Siglecs are sialic acid specific I-type lectins that belong to the immunoglobulin superfamily. Structurally, they are transmembrane proteins with an N-terminal Ig-like V‑set domain followed by varying numbers of Ig-like C2-set domains (1, 2). Human Siglec-1, also known as sialoadhesin and CD169, is a 175‑185 kDa glycoprotein. It contains a 1622 amino acid (aa) extracellular domain (ECD) with one Ig-like V‑set domain and 16 Ig-like C2-set domains, a 21 aa transmembrane segment, and a 44 aa cytoplasmic domain (3). Within the ECD, human Siglec-1 shares approximately 70% aa sequence identity with mouse and rat Siglec-1. Alternate splicing generates a potentially soluble form of the ECD, and a second isoform with a substituted cytoplasmic domain. Siglec-1 expression is restricted to lymph node and splenic macrophages, plus some tissue macrophages (3). The adhesive function of Siglec-1 is supported by the N-terminal Ig-like domain which shows a selectivity for alpha 2,3‑linked sialic acid residues (3‑5). Siglec-1 binds a number of sialylated molecules including the mannose receptor, MGL1, MUC1, PSGL-1, and different glycoforms of CD43 (6‑9). Its binding capacity can be masked by endogenous sialylated molecules (10, 11). The sialylated and sulfated N-linked carbohydrates that modify Siglec-1 itself are required for ligand binding (6, 7). Siglec-1 is expressed on dendritic cells following rhinovirus exposure, and these DC promote T cell anergy (12). It is also induced on circulating monocytes during systemic sclerosis and HIV-1 infection (13‑15). Siglec-1 can trap HIV-1 particles for trans infection of permissive cells (14).
Human Siglec‑1/CD169 Antibody
R&D Systems | Catalog # AF5197

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, Neutralization
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Flow Cytometry
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Siglec‑1/CD169
Ser20-Gln1641
Accession # Q9BZZ2
Ser20-Gln1641
Accession # Q9BZZ2
Specificity
Detects human Siglec‑1/CD169 in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human Siglec‑1/CD169 Antibody
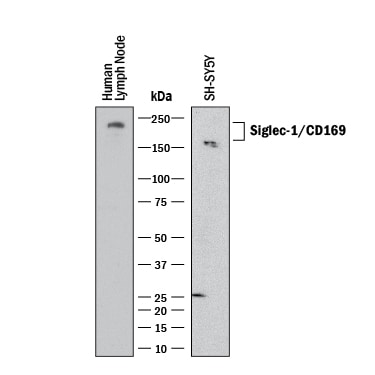
Detection of Human Siglec‑1/CD169 by Western Blot.
Western blot shows lysates of SH‑SY5Y human neuroblastoma cell line and human lymph node. PVDF membrane was probed with 0.5 µg/mL of Sheep Anti-Human Siglec‑1/CD169 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF5197) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (HAF016). A specific band was detected for Siglec‑1/CD169 at approximately 180-200 kDa (as indicated). This experiment was conducted under reducing conditions and using Western Blot Buffer Group 1.Cell Adhesion Mediated by Siglec‑1/CD169 and Neutralization by Human Siglec‑1/CD169 Antibody.
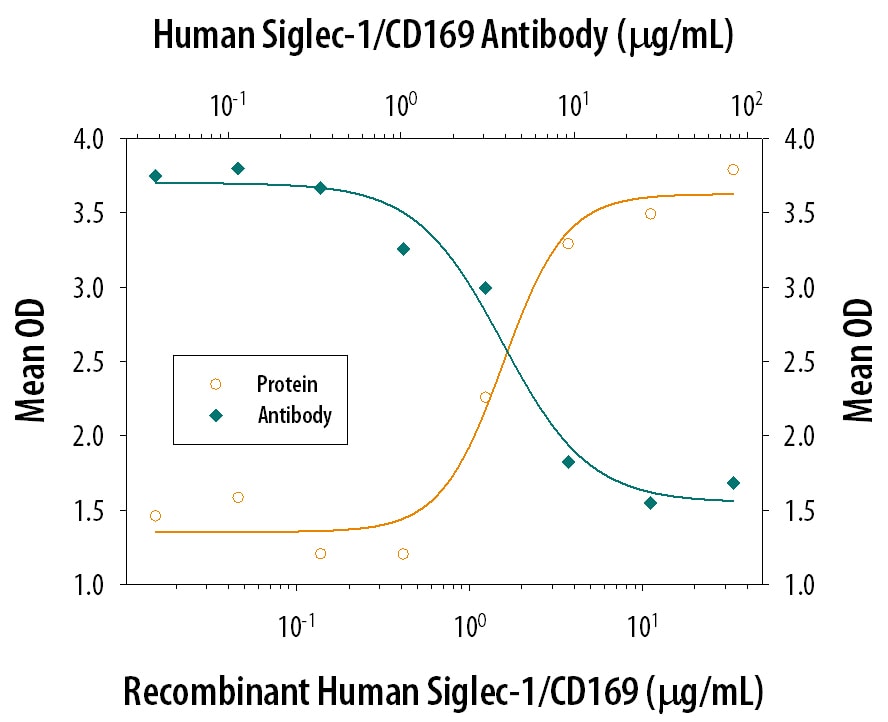
Recombinant Human Siglec-1/CD169 Fc Chimera (Catalog # 5197-SL), immobilized onto a microplate, supports the adhesion of human red blood cells in a dose-dependent manner (orange line). Adhesion elicited by Recombinant Human Siglec-1/CD169 Fc Chimera (5 µg/mL) is neutralized (green line) by increasing concentrations of Sheep Anti-Human Siglec-1/CD169 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF5197). The ND50 is typically 1.5-7.5 µg/mL.Applications for Human Siglec‑1/CD169 Antibody
Application
Recommended Usage
Western Blot
0.5 µg/mL
Sample: SH‑SY5Y human neuroblastoma cell line and human lymph node
Sample: SH‑SY5Y human neuroblastoma cell line and human lymph node
Neutralization
Measured by its ability to neutralize Siglec‑1/CD169-mediated adhesion of human red blood cells. Kelm, S. et al. (1994) Current Biology 4:965. The Neutralization Dose (ND50) is typically 1.5-7.5 µg/mL in the presence of 5 µg/mL Recombinant Human Siglec‑1/CD169 Fc Chimera.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Siglec-1/CD169
References
- Varki, A. and T. Angata (2006) Glycobiology 16:1R.
- Crocker, P.R. et al. (2007) Nat. Rev. Immunol. 7:255.
- Hartnell, A. et al. (2001) Blood 97:288.
- Nath, D. et al. (1995) J. Biol. Chem. 270:26184.
- Crocker, P.R. et al. (1991) EMBO J. 10:1661.
- Martinez-Pomares, L. et al. (1999) J. Biol. Chem. 274:35211.
- Kumamoto, Y. et al. (2004) J. Biol. Chem. 279:49274.
- Nath, D. et al. (1999) Immunology 98:213.
- van den Berg, T.K. et al. (2001) J. Immunol. 166:3637.
- Nakamura, K. et al. (2002) Glycobiology 12:209.
- Barnes, Y.C. et al. (1999) Blood 93:1245.
- Kirchberger, S. et al. (2005) J. Immunol. 175:1145.
- York, M.R. et al. (2007) Arthritis Rheum. 56:1010.
- Rempel, H. et al. (2008) PloS ONE 3:e1967.
- van der Kuyl, A.C. et al. (2007) Plos ONE 2:e257.
Long Name
Sialic Acid Binding Ig-like Lectin 1
Alternate Names
CD169, Siglec1
Gene Symbol
SIGLEC1
UniProt
Additional Siglec-1/CD169 Products
Product Documents for Human Siglec‑1/CD169 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Siglec‑1/CD169 Antibody
For research use only
Related Research Areas
Citations for Human Siglec‑1/CD169 Antibody
Customer Reviews for Human Siglec‑1/CD169 Antibody
There are currently no reviews for this product. Be the first to review Human Siglec‑1/CD169 Antibody and earn rewards!
Have you used Human Siglec‑1/CD169 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...