Signal regulatory protein alpha (SIRP alpha, designated CD172a), also called SHPS-1 (SHP substrate 1) and previously, MyD-1 (Myeloid/Dendritic-1), is a monomeric ~90 kDa type I transmembrane glycoprotein that belongs to the SIRP/SHPS (CD172) family of the immunoglobulin superfamily (1‑4). SIRPs are paired receptors, with similar extracellular domains but differing C-termini and functions (1, 2). The 503 amino acid (aa) human SIRP alpha contains a 342 aa extracellular domain (ECD), with one V-type, and two C1 type Ig domains, and three potential N glycosylation sites. It has a 110 aa cytoplasmic sequence with ITIM motifs that recruit tyrosine phosphatases SHP-1 and SHP-2 when phosphorylated (4). Human SIRP alpha has more than 40 described polymorphisms, including the prominent BIT (Brain Ig like molecule with Tyrosine-based activation motifs, also called SIRP alpha 2 or PTPNS) (5). One reported isoform lacks aa 1‑101, which eliminates most of the V type Ig domain. Human SIRP alpha ECD shares 61%, 60%, 71%, 72% and 73% aa identity with mouse, rat, porcine, bovine and equine SIRP alpha, respectively; it shares 84% and 76% aa identity with human SIRP beta 1 and SIRP gamma, respectively (2). SIRP alpha is expressed mainly on myeloid cells, including macrophages, neutrophils, dendritic and Langerhans cells (3‑6). It is also found on neurons, smooth muscle and endothelial cells (7‑9). SIRP alpha shows adhesion to the ubiquitous CD47/IAP (integrin associated protein), while SIRP gamma binds more weakly and SIRP alpha 1 does not bind at all (1, 2). Mouse and human SIRP alpha -CD47 binding only cross-reacts for specific polymorphisms and influences engraftment of xenotransplanted stem cells (6, 10). SIRP alpha engagement generally produces a negative regulatory signal (4). Low SIRP alpha recognition of CD47, which occurs on aged erythrocytes or platelets or xenogenic cells, promotes clearance of CD47low cells from circulation (11, 13). SIRP alpha recognition of surfactants SP-A and SP-D in the lung can inhibit alveolar macrophage cytokine production (14). The CD47 integrin-SIRP alpha interaction is reported to promote macrophage fusion during osteoclastogenesis (15).
Human SIRP alpha /CD172a
Antibody
R&D Systems | Catalog # AF4546

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot, Simple Western
Cited:
Western Blot, ELISA Capture
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Chinese hamster ovary cell line CHO-derived recombinant human SIRP alpha /CD172a
Gly27-Asn370
Accession # P78324
Gly27-Asn370
Accession # P78324
Specificity
Detects human SIRP alpha /CD172a in Western blots.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Human SIRP alpha /CD172a
Antibody
Detection of Human SIRP alpha /CD172a
by Western Blot.
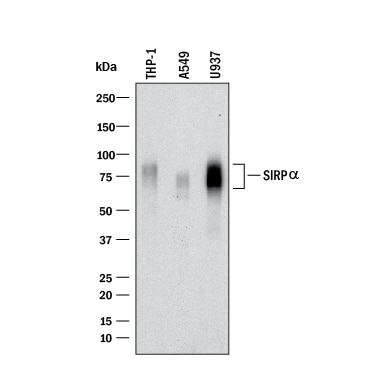
Western blot shows lysates of THP‑1 human acute monocytic leukemia cell line, A549 human lung carcinoma cell line, and U937 human histiocytic lymphoma cell line. PVDF membrane was probed with 0.5 µg/mL of Sheep Anti-Human SIRP alpha /CD172aAntigen Affinity-purified Polyclonal Antibody (Catalog # AF4546) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (HAF016). A specific band was detected for SIRP alpha /CD172a
at approximately 70-95 kDa (as indicated). This experiment was conducted under reducing conditions and using Western Blot Buffer Group 1.
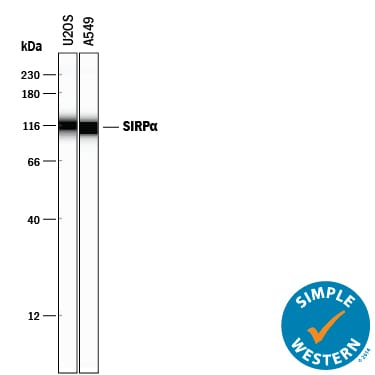
Detection of Human SIRP alpha /CD172a by Simple WesternTM.
Simple Western lane view shows lysates of U2OS human osteosarcoma cell line and A549 human lung carcinoma cell line, loaded at 0.2 mg/mL. A specific band was detected for SIRPa/CD172a at approximately 113-116 kDa (as indicated) using 50 µg/mL of Sheep Anti-Human SIRPa/CD172a Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4546) followed by 1:50 dilution of HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Applications for Human SIRP alpha /CD172a Antibody
Application
Recommended Usage
Simple Western
50 µg/mL
Sample: U2OS human osteosarcoma cell line and A549 human lung carcinoma cell line
Sample: U2OS human osteosarcoma cell line and A549 human lung carcinoma cell line
Western Blot
0.5 µg/mL
Sample: THP‑1 human acute monocytic leukemia cell line, A549 human lung carcinoma cell line, and U937 human histiocytic lymphoma cell line
Sample: THP‑1 human acute monocytic leukemia cell line, A549 human lung carcinoma cell line, and U937 human histiocytic lymphoma cell line
Reviewed Applications
Read 1 review rated 4 using AF4546 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: SIRP alpha/CD172a
References
- Barclay, A.N. & M.H. Brown (2006) Nat. Rev. Immunol. 6:457.
- vanBeek, E.M. et al. (2005) J. Immunol. 175:7781.
- Liu, Y. et al. (2005) J. Biol. Chem. 280:36132.
- Kharitonenkov, A. et al. (1997) Nature 386:181.
- Swissprot Accession # P78324.
- Miyashita, M. et al. (2004) Mol. Biol. Cell 15:3950.
- Wang, X.X. & K.H. Pfenninger (2005) J. Cell Sci. 119:172.
- Maile, L.A. et al. (2003) Mol. Biol. Cell 14:3519.
- Johansen, M.L. & E.J. Brown (2007) J. Biol. Chem. 282:24219.
- Takenaka, K. et al. (2007) Nat. Immunol. 8:1313.
- Ishikawa-Sekigami, T. et al. (2006) Biochem. Biophys. Res. Commun. 343:1197.
- Olsson, M. et al. (2005) Blood 105:3577.
- Ide, K. et al. (2007) Proc. Natl. Acad. Sci. USA 104:5062.
- Gardai, S.J. et al. (2003) Cell 115:13.
- Lundberg, P. et al. (2007) Biochem. Biophys. Res. Commun. 352:444.
Long Name
Signal-regulatory Protein alpha
Alternate Names
BIT, CD172a, MFR, MYD-1, SHPS1, SIRPA
Entrez Gene IDs
Gene Symbol
SIRPA
UniProt
Additional SIRP alpha/CD172a Products
Product Documents for Human SIRP alpha /CD172a Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human SIRP alpha /CD172a Antibody
For research use only
Citations for Human SIRP alpha /CD172a
Antibody
Customer Reviews for Human SIRP alpha /CD172a Antibody (1)
4 out of 5
1 Customer Rating
Have you used Human SIRP alpha /CD172a Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: ELISASample Tested: SerumSpecies: HumanVerified Customer | Posted 06/30/2021
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars