Uteroglobin, also called Clara cell secretory, phospholipid binding, 10 kDa or 16 kDa protein (CCSP, CCPBP, CC10 or CC16, respectively) is a small, non‑glycosylated secreted protein of the secretoglobin superfamily, (designated 1A, member 1) (1‑3). Its name is derived from its very high expression in the pre‑implantation uterus. It is produced by the non-ciliated, non-mucous secretory cells that predominate in lung bronchioles (Clara cells), and other non-ciliated epithelia that communicate with the external environment (1‑3). Expression is induced by steroid hormones such as estrogen, and enhanced by the non-steroid hormone prolactin (1). Uteroglobin is found in blood, urine and other body fluids (1). Human Uteroglobin cDNA encodes a 21 amino acid (aa) signal sequence and a 70 aa mature protein. It shares 53-56% aa identity with mouse, rat, bovine, canine, equine or rabbit Uteroglobin, and is active in mice (4). The mature protein forms a disulfide-linked head-to-tail homodimer of 16 kDa (2, 5). This homodimer is thought to form a binding pocket that binds hydrophobic ligands such as phospholipids, progesterone and retinols (5). Sequestering of prostaglandins and leukotrienes is anti-inflammatory, while sequestering of carcinogens such as polychlorinated bisphenols is anti-tumorigenic (6‑8). Other immunoregulatory activities of Uteroglobin include cell migration inhibition (by binding the chemotaxis-related formyl peptide receptor FPR2 on dendritic cells), and the inhibition of T cell differentiation to a Th2 phenotype (9). A single nucleotide polymorphism of Uteroglobin, A38G, confers increased risk of asthma (10). Transglutaminase can crosslink Uteroglobin, either to itself or to other proteins such as the adhesion molecule fibronectin (3, 11). Binding of fibronectin to Uteroglobin in the kidney is thought to protect against nephropathy, while binding of the lipocalin-1 receptor has been reported to suppress cancer cell motility and invasion (12, 13).
Human Uteroglobin/SCGB1A1 Antibody
R&D Systems | Catalog # AF4218

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Rat
Applications
Validated:
Immunohistochemistry
Cited:
Immunohistochemistry
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human Uteroglobin/SCGB1A1
Glu22-Asn91
Accession # NP_003348
Glu22-Asn91
Accession # NP_003348
Specificity
Detects human Uteroglobin/SCGB1A1 in direct ELISAs.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human Uteroglobin/SCGB1A1 Antibody
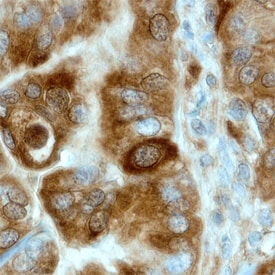
Uteroglobin/SCGB1A1 in Human Endometrial Cancer Tissue.
Uteroglobin/SCGB1A1 was detected in immersion fixed paraffin-embedded sections of human endometrial cancer tissue using Human Uteroglobin/SCGB1A1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4218) at 3 µg/mL overnight at 4 °C. Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (CTS013). Tissue was stained using the Anti-Sheep HRP-DAB Cell & Tissue Staining Kit (brown; CTS019) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm of epithelial cells. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Applications for Human Uteroglobin/SCGB1A1 Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections in human endometrial cancer tissue
Sample: Immersion fixed paraffin-embedded sections in human endometrial cancer tissue
Reviewed Applications
Read 1 review rated 5 using AF4218 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Uteroglobin/SCGB1A1
References
- Mukherjee, A.B. et al. (2007) Endocr. Rev. 28:707.
- Singh, G. et al. (1988) Biochim. Biophys. Acta 950:329.
- Mantile, G. et al. (1993) J. Biol. Chem. 268:20343.
- Wang, S-Z. et al. (2003) J. Immunol. 171:1051.
- Umland, T.C. et al. (1994) Nat. Struct. Biol. 1:538.
- Mandal, A.K. et al. (2004) J. Exp. Med. 199:1317.
- Watson, T.M. et al. (2001) Am. J. Physiol. Lung Cell Mol. Physiol. 281:L1523.
- Yang, Y. et al. (2004) J. Biol. Chem. 279:29336.
- Ray, R. et al. (2006) FEBS Lett. 580:6022.
- Martin, A.C. et al. (2006) Am. J. Respir. Crit. Care Med. 173:617.
- Antico, G. et al. (2006) J. Cell Physiol. 207:553.
- Zheng, F. et al. (1999) Nat. Med. 5:1018.
- Zhang, Z. et al. (2006) Gene 369:66.
Alternate Names
Blastokinin, CC10, CC16, CCPBP, CCSP, Clara, PCB-BP, SCGB1A1, UGB
Gene Symbol
SCGB1A1
UniProt
Additional Uteroglobin/SCGB1A1 Products
Product Documents for Human Uteroglobin/SCGB1A1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Uteroglobin/SCGB1A1 Antibody
For research use only
Related Research Areas
Citations for Human Uteroglobin/SCGB1A1 Antibody
Customer Reviews for Human Uteroglobin/SCGB1A1 Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human Uteroglobin/SCGB1A1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: ELISASample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 12/20/2017
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
