Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Hamster, Monkey, Primate - Macaca mulatta (Rhesus Macaque), Transgenic Mouse, Xenograft
Applications
Validated:
Immunohistochemistry, Western Blot
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Flow Cytometry, Immunofluorescence, Immunocytochemistry, Immunocytochemistry/ Immunofluorescence
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human VASA
Met1-Tyr145
Accession # Q9NQI0
Met1-Tyr145
Accession # Q9NQI0
Specificity
Detects human VASA in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human VASA Antibody
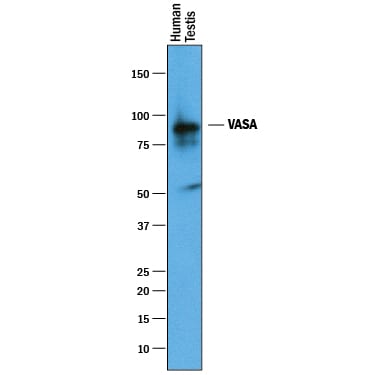
Detection of Human VASA by Western Blot.
Western blot shows lysates of human testis tissue. PVDF membrane was probed with 1 µg/mL of Goat Anti-Human VASA Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2030) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF019). A specific band was detected for VASA at approximately 85 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.VASA in Human Testis.
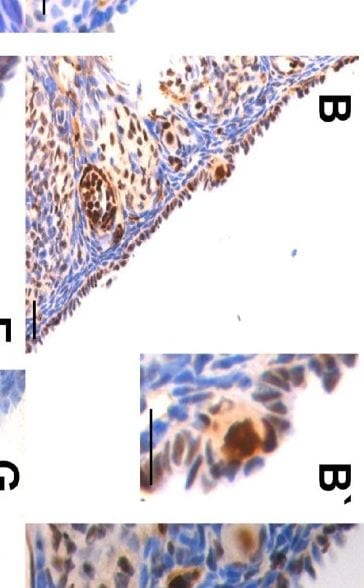
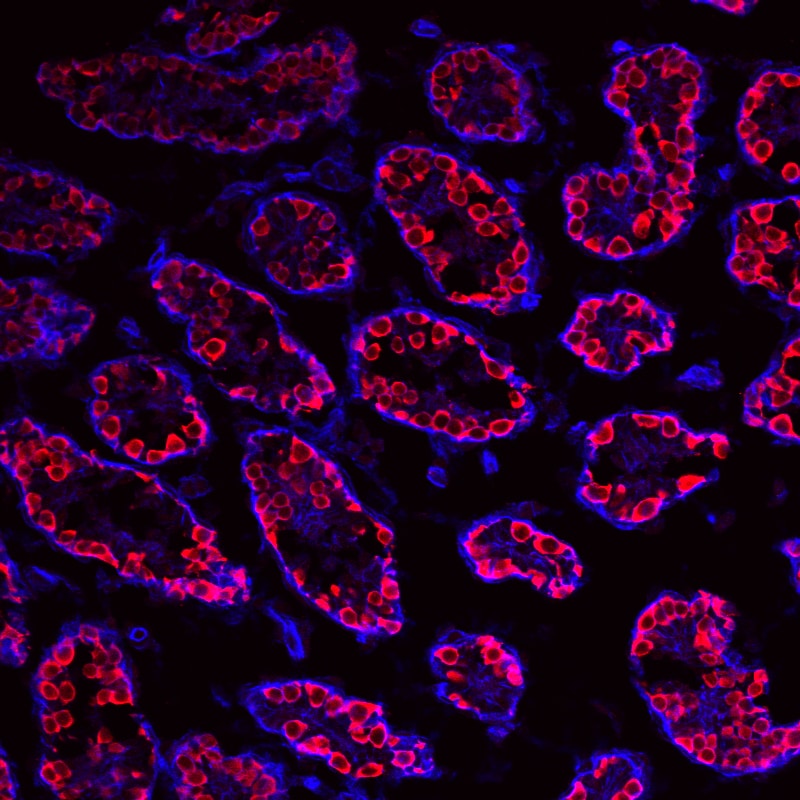
VASA was detected in paraffin-embedded sections of human testis using Goat Anti-Human VASA Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2030) at 10 µg/mL overnight at 4 °C. Before incubation with the primary antibody tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Mouse VASA by Immunohistochemistry
Immuno-staining on control and PMSG treated ovarian sections.A-C: Immunostaining with anti-PCNA antibody. Note minimal PCNA staining in control OSE. Increased staining is observed after PMSG treatment in both the OSE cells and the oocytes of PF (B-&C). Inset B’ is magnified image of the PF located in the OSE. At places, granulosa cells were also positive for PCNA (arrowhead). D-F: Immunostaining with anti-OCT-4 antibody in control (D) and 7D PMSG treated (E&F) ovarian sections. OCT-4 is localized in the ooplasm in control, while in 7D PMSD treated, positive staining was observed in ooplasm as well as in the nucleus of few oocytes (arrowhead) in PFs. G: Immunostaining with anti-human VASA antibody that cross-reacts with mouse MVH. MVH is distinctly localized in the ooplasm of PF and at places was also observed in a ‘germ cell nest’ (G’, arrow). At places some oocytes of primordial follicles in cohorts appeared connected without intervening granulosa cell (arrowhead) H-J: SCP-3 is localized in PF oocytes present in close vicinity of multilayer OSE. Bar: 20μm. Image collected and cropped by CiteAb from the following publication (https://ovarianresearch.biomedcentral.com/articles/10.1186/1757-2215-5-…), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human VASA Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human testis subjected to Antigen Retrieval Reagent-Basic (Catalog # CTS013)
Sample: Immersion fixed paraffin-embedded sections of human testis subjected to Antigen Retrieval Reagent-Basic (Catalog # CTS013)
Western Blot
1 µg/mL
Sample: Human testis tissue
Sample: Human testis tissue
Reviewed Applications
Read 6 reviews rated 5 using AF2030 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: VASA
References
- Zeeman, A.M. et al. (2002) Lab Invest. 82:159.
- Raz, E. (2000) Genome Biol. 1:REVIEWS1017.
- Castrillon, D. et al. (2001) Proc. Natl. Acad. Sci. USA 97:9585.
Long Name
DEAD [Asp-Glu-Ala-Asp] Box Polypeptide 41
Alternate Names
DDX4
Gene Symbol
DDX4
UniProt
Additional VASA Products
Product Documents for Human VASA Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human VASA Antibody
For research use only
Related Research Areas
Citations for Human VASA Antibody
Customer Reviews for Human VASA Antibody (6)
5 out of 5
6 Customer Ratings
Have you used Human VASA Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
6 reviews
Showing All
Filter By:
-
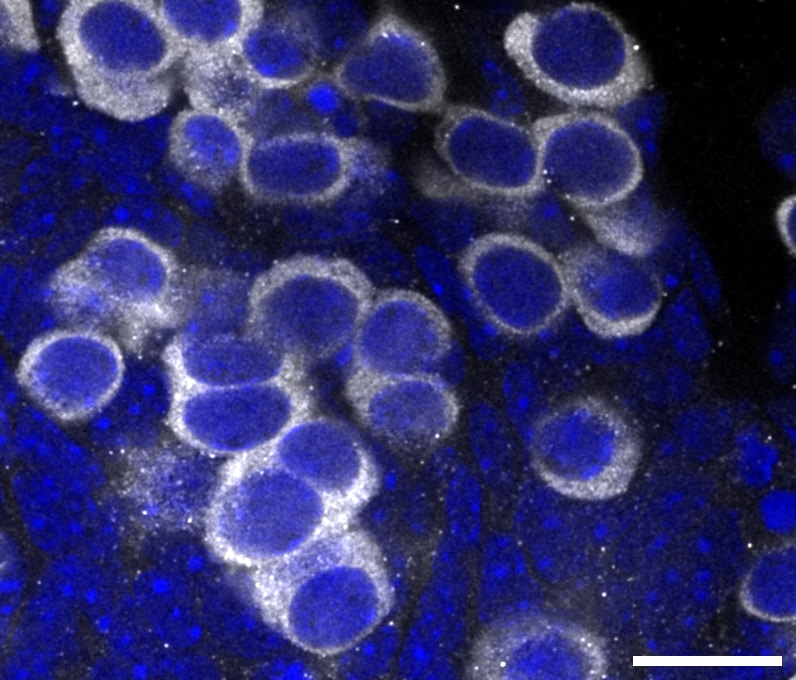
Application: ImmunofluorescenceSample Tested: Testis tissueSpecies: HumanVerified Customer | Posted 02/20/2025Vasa signaling specific label human germ cell.Sections were treated with xylene for deparaffinization and ethanol for rehydration. Antigen retrieval was achieved by microwaving in 10 mM citrate-based buffer at pH 6.0. Sections were washed by PBS with 0.05% Tween 20, followed by incubation with VASA antibodies overnight at 4°C and secondary antibodies at room temperature for 1 h.
-
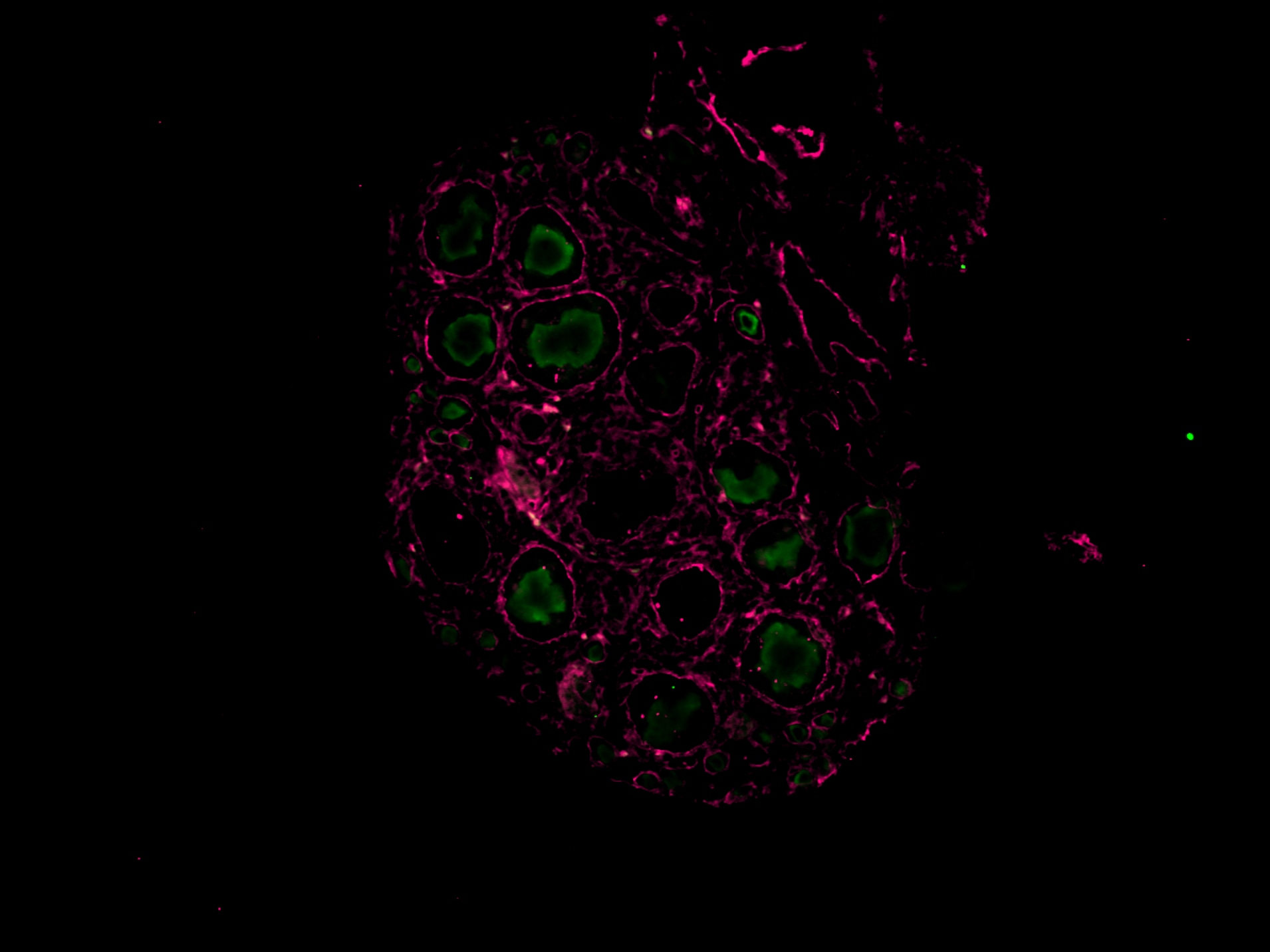
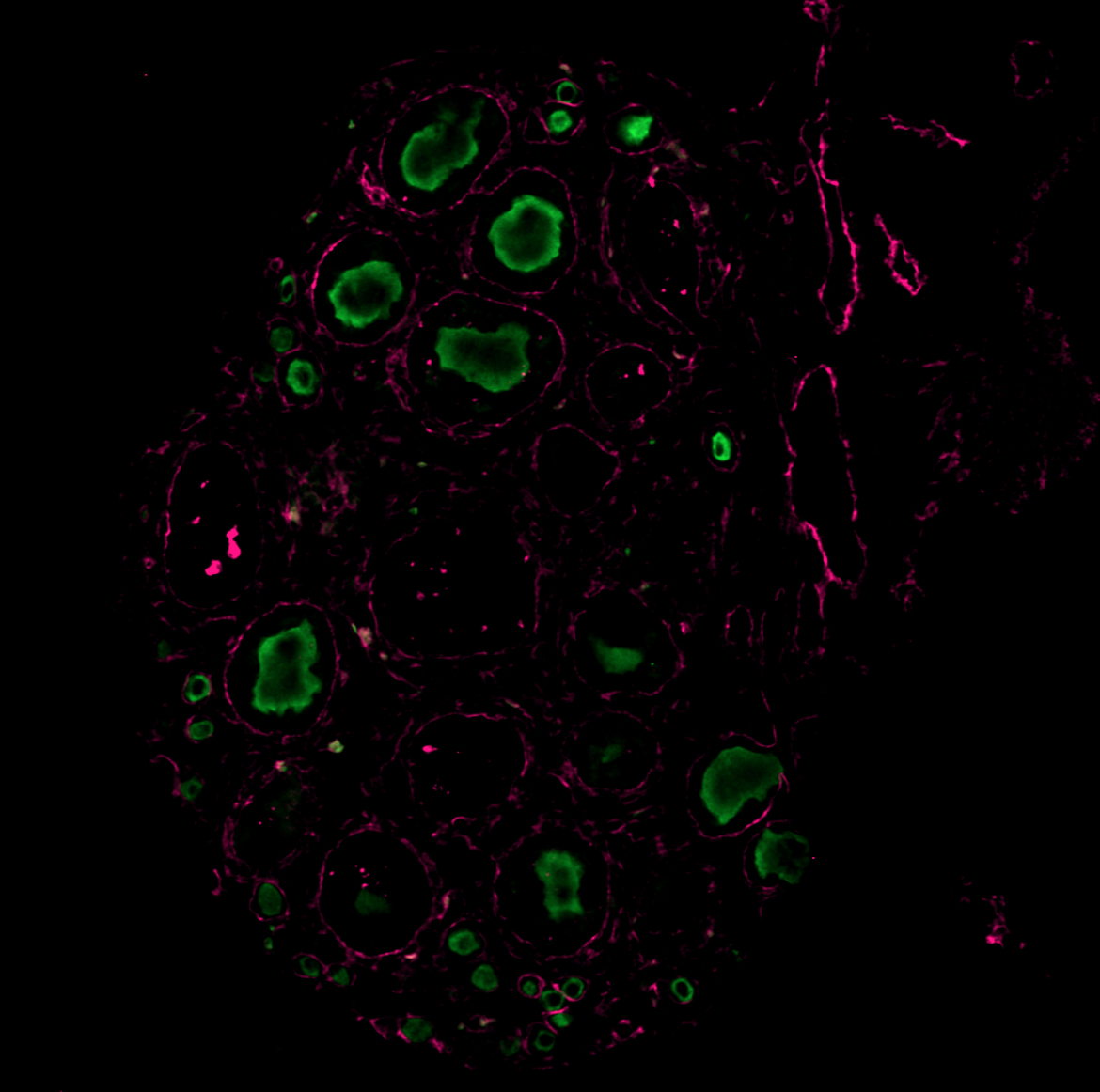
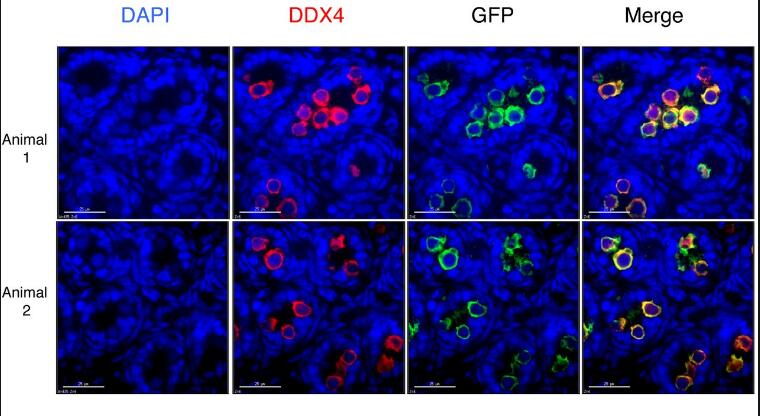
Application: ImmunohistochemistrySample Tested: Ovary tissueSpecies: MouseVerified Customer | Posted 11/15/2024Worked on mouse P14 ovary magenta (laminin), green (vasa), working concentration 1:100, IHC-P
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Ovary tissueSpecies: MouseVerified Customer | Posted 11/08/2024The antibody was tested on a paraffin-embedded section of a mouse P14 ovary, with successful staining observed at a dilution of 1:200. Green (VASA) Magenta (Laminin)
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Ovary tissueSpecies: MouseVerified Customer | Posted 11/16/2023Whole mount of Mouse Ovary (E16.5). Dilution: 1:300.
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Testis tissueSpecies: MouseVerified Customer | Posted 11/11/2019DDX4/VASA germ cell marker. IF1:500
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Testis tissueSpecies: MouseVerified Customer | Posted 04/13/2016Postnatal day 6 mouse testes were fixed in 4% paraformaldehyde. Tissue was embedded in O.C.T. and 5 micron sections cut. Sections were permeabilized with PBS containing 0.1% Triton X-100, then blocked for 30 min in 3% BSA in PBS containing 0.1% Triton X-100. Goat Anti-Human VASA Antigen Affinity-purified Polyclonal Antibody (AF2030) was diluted in blocking buffer to 1.25 µg/ml and applied to sections for 1 hour at RT. Sections were washed 3X with PBS containing 0.1% Triton X-100, then incubated with secondary antibody: Donkey anti-Goat IgG (H+L) Secondary Antibody, Alexa Fluor® 555 conjugate (Thermo Fisher A-21432) diluted to 1:500 for 1 hour at RT. Sections were washed 3X with PBS containing 0.1% Triton X-100 and mounted.
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...