LRRK2 Antibody - BSA Free
Novus Biologicals | Catalog # NB300-268

![Western Blot: LRRK2 AntibodyBSA Free [NB300-268] Western Blot: LRRK2 AntibodyBSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Western-Blot-NB300-268-img0023.jpg)
Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Localization
Clonality
Host
Isotype
Theoretical MW
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for LRRK2 Antibody - BSA Free
Immunohistochemistry-Frozen: LRRK2 Antibody - BSA Free [NB300-268]
Immunohistochemistry-Frozen: LRRK2 Antibody [NB300-268] - Staining as described in PMID 24312256. Image from verified customer review.Western Blot: LRRK2 AntibodyBSA Free [NB300-268]
LRRK2-Antibody-Western-Blot-NB300-268-img0028.jpgImmunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268]
LRRK2-Antibody-Immunohistochemistry-Paraffin-NB300-268-img0026.jpgFlow Cytometry: LRRK2 Antibody - BSA Free [NB300-268]
Flow Cytometry: LRRK2 Antibody [NB300-268] - Analysis using the Alexa Fluor 647 conjugate of NB300-268. Staining of LRRK2 in normal human peripheral blood cells (anticoagulated Lithium-Heparin) using anti-LRRK2 antibody conjugated with AF647. The primary antibody was used at a dilution of 1:200 and incubated for 20 min at room temperature. Image from verified customer review.Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268]
LRRK2-Antibody-Immunohistochemistry-Paraffin-NB300-268-img0024.jpgImmunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268]
Immunocytochemistry/Immunofluorescence: LRRK2 Antibody [NB300-268] - HeLa cells were fixed in 4% paraformaldehyde for 10 minutes and permeabilized in 0.05% Triton X-100 in PBS for 5 minutes. The cells were incubated with anti-LRRK2 Antibody NB300-268 at 2 ug/ml overnight at 4C and detected with an anti-mouse Dylight 488 (Green) at a 1:1000 dilution for 60 minutes. Nuclei were counterstained with DAPI (Blue). Cells were imaged using a 100X objective and digitally deconvolved.Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268]
Flow Cytometry: LRRK2 Antibody [NB300-268] - An intracellular stain was performed on U2OS cells with LRRK2 Antibody NB300-268 (blue) and a matched isotype control (orange). Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 2.5 ug/mL for 30 minutes at room temperature, followed by Rabbit IgG (H+L) Cross-Adsorbed Secondary Antibody, Dylight 550 (SA5-10033, Thermo Fisher).Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268]
Immunocytochemistry/Immunofluorescence: LRRK2 Antibody [NB300-268] - LRRK2 antibody was tested in SH-SY-5Y cells at a 1:200 dilution against DyLight 488 (Green). Alpha tubulin and nuclei were counterstained against DyLight 568 (Red), and DAPI (Blue), respectively.Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268]
Immunohistochemistry-Paraffin: LRRK2 Antibody [NB300-268] - Staining of human brain, Putamen, Neurons and Glia using a 60X magnification.Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268]
Flow Cytometry: LRRK2 Antibody [NB300-268] - An intracellular stain was performed on U2OS cells with LRRK Antibody NB300-268AF647 (blue) and a matched isotype control (orange). Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 2.5 ug/mL for 30 minutes at room temperature. Both antibodies were conjugated to Alexa Fluor 647.Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268]
Flow Cytometry: LRRK2 Antibody [NB300-268] - An intracellular stain was performed on Neuro2a cells with LRRK2 Antibody NB300-268 (blue) and a matched isotype control (orange). Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 2.5 ug/mL for 30 minutes at room temperature, followed by Rabbit IgG (H+L) Cross-Adsorbed Secondary Antibody, Dylight 550 (SA5-10033, Thermo Fisher).Western Blot: LRRK2 Antibody - BSA Free [NB300-268] -
LRRK2 kinase inhibition blocks Tat-induced S935 phosphorylation and inflammatory cytokine expression in BV-2 cells. (A) Western blot depicting pS935-LRRK2 and total LRRK2 protein levels 12 hours after saline or Tat-treatment ± LRRK2 kinase inhibition (LRRK2i).Immunocytochemistry/Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] -
The interaction and phosphorylation of LRRK2 and LC1. (A) Cellular co-localisation of endogenous LRRK2 and MAP1B (LC1). SKNSH cells were labelled with LRRK2 (green) and LC1 (red) antibodies and signals were detected by immunofluorescence. Detailed figure in Additional file 1.Western Blot: LRRK2 Antibody - BSA Free [NB300-268] -
Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - Western blotting of LRRK2. A. Recombinant LRRK2 (arrowhead) from transfected (+) HEK 293T & M17 cells was specifically recognized by all four LRRK2 antibodies (Ab1, Ab2, Ab3, & Ab4) used in this study. LRRK2 was not recognized in non-transfected cells (-). B. Recombinant LRRK2 (arrowhead) from transfected M17 cells is detected by anti-LRRK2 Ab4 (lane 1), but not after absorption of the antibody with its peptide antigen (lane 2). Brain LRRK2 was recognized by anti-LRRK2 Ab4 in two controls (lanes 3 & 4) & two PD cases (lanes 5 & 6). Cell lysates (10 μg protein) & brain homogenates (100 μg protein) were prepared & loaded on 6% SDS-PAGE gels for Western blot analysis using anti-LRRK2 antibodies Ab1–Ab4 in (A) & Ab4 in (B). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/17137507), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268] -
Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268] - Deletion of LRRK2 attenuates Mn-induced apoptosis & cell death in RAW 264.7 cells.(A) LRRK2 WT or KO RAW 264.7 cells were treated with Mn (250 μM) for the designated times, followed by flow cytometry analysis to determine Mn-induced apoptosis. Both early & late apoptotic cells (Q2 & Q3) were measured. (B) At the end of Mn exposure, cell viability was determined by MTT assay. @@@, p < 0.001; ###, p < 0.001; ***, p < 0.001 compared to the control (one-way ANOVA followed by Tukey’s post hoc test; n = 3, for apoptosis assay; n = 6, for MTT assay). The data shown are representative of 3 independent experiments. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30645642), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] -
Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 accumulates in globules in alpha S tg mice. (a & b) Double immunofluorescence for alpha S with parkin, PINK1, DJ-1, LRRK2, or negative control (the immunopeptide-preabsorbed anti-LRRK2 antibody) in alpha S tg mice (a) & P123H beta S tg mice (b). Note that alpha S-globules were immunopositive for LRRK2 (~79%, n = 22), whereas P123H beta S globules were negative for LRRK2. Representative images are shown for the thalamus ( alpha S) & basal ganglia (P123H beta S). Scale bar = 5 μm for all panels. (c) Triple immunofluorescence for alpha S, LRRK2 & Rab5B for basal ganglia in alpha S tg mice. LRRK2 & Rab5B were colocalized in axon terminal (arrow), but were not colocalized in the alpha S-globule (arrowhead) Scale bar = 10 μm for all panels. Image collected & cropped by CiteAb from the following publication (https://molecularbrain.biomedcentral.com/articles/10.1186/1756-6606-5-34), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] -
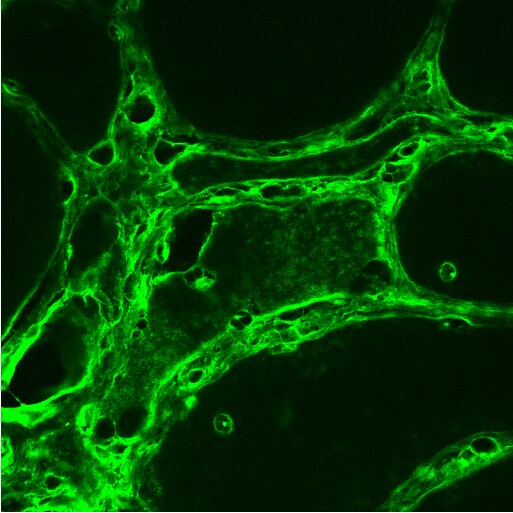
Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - Specificity of immunostaining for LRRK2. Specificity of the antibodies used for immunocytochemistry was determined by performing adsorptions with the corresponding peptide sequences. Immunochemical localization of LBs (arrows) by Ab4 against LRRK22500–2527 (A) & Ab1 against LRRK2900–100 (C) was blocked using respective peptide antigens (B & D). Adjacent sections with LB within pigmented neurons from the substantia nigra of a case of PD were used. Scale bar: 20 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/17137507), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] -
Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - Immunocytochemistry of LRRK2 in DLB. Cortical LBs (arrows) in DLB were positive for LRRK2 using both Ab4 against LRRK22500–2527 (A), & Ab1 against LRRK2900–100 (B). Neuronal cytoplasm (C) was also strongly labelled throughout the cortex by Ab4 & with a consistently more granular pattern by Ab1 (inset in C) in cases of DLB. Control cases display similar, though less intense neuronal labeling (D). Biochemically purified cortical LBs were strongly positive for staining by Ab4 (E) as well as by anti-alpha -synuclein (F), while they were unstained when omitting primary antibody (G). Large vessels (H) were also intensely labelled by Ab4. Scale bars: A, B = 25 μm; C, D = 100 μm; E-G = 25 μm; H = 50 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/17137507), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] -
Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - Immunocytochemistry of LRRK2 in DLB. Cortical LBs (arrows) in DLB were positive for LRRK2 using both Ab4 against LRRK22500–2527 (A), & Ab1 against LRRK2900–100 (B). Neuronal cytoplasm (C) was also strongly labelled throughout the cortex by Ab4 & with a consistently more granular pattern by Ab1 (inset in C) in cases of DLB. Control cases display similar, though less intense neuronal labeling (D). Biochemically purified cortical LBs were strongly positive for staining by Ab4 (E) as well as by anti-alpha -synuclein (F), while they were unstained when omitting primary antibody (G). Large vessels (H) were also intensely labelled by Ab4. Scale bars: A, B = 25 μm; C, D = 100 μm; E-G = 25 μm; H = 50 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/17137507), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] -
Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 accumulates in globules in alpha S tg mice. (a & b) Double immunofluorescence for alpha S with parkin, PINK1, DJ-1, LRRK2, or negative control (the immunopeptide-preabsorbed anti-LRRK2 antibody) in alpha S tg mice (a) & P123H beta S tg mice (b). Note that alpha S-globules were immunopositive for LRRK2 (~79%, n = 22), whereas P123H beta S globules were negative for LRRK2. Representative images are shown for the thalamus ( alpha S) & basal ganglia (P123H beta S). Scale bar = 5 μm for all panels. (c) Triple immunofluorescence for alpha S, LRRK2 & Rab5B for basal ganglia in alpha S tg mice. LRRK2 & Rab5B were colocalized in axon terminal (arrow), but were not colocalized in the alpha S-globule (arrowhead) Scale bar = 10 μm for all panels. Image collected & cropped by CiteAb from the following publication (https://molecularbrain.biomedcentral.com/articles/10.1186/1756-6606-5-34), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] -
Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 accumulates in globules in alpha S tg mice. (a & b) Double immunofluorescence for alpha S with parkin, PINK1, DJ-1, LRRK2, or negative control (the immunopeptide-preabsorbed anti-LRRK2 antibody) in alpha S tg mice (a) & P123H beta S tg mice (b). Note that alpha S-globules were immunopositive for LRRK2 (~79%, n = 22), whereas P123H beta S globules were negative for LRRK2. Representative images are shown for the thalamus ( alpha S) & basal ganglia (P123H beta S). Scale bar = 5 μm for all panels. (c) Triple immunofluorescence for alpha S, LRRK2 & Rab5B for basal ganglia in alpha S tg mice. LRRK2 & Rab5B were colocalized in axon terminal (arrow), but were not colocalized in the alpha S-globule (arrowhead) Scale bar = 10 μm for all panels. Image collected & cropped by CiteAb from the following publication (https://molecularbrain.biomedcentral.com/articles/10.1186/1756-6606-5-34), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: LRRK2 Antibody - BSA Free [NB300-268] -
Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - Western blotting of LRRK2. A. Recombinant LRRK2 (arrowhead) from transfected (+) HEK 293T & M17 cells was specifically recognized by all four LRRK2 antibodies (Ab1, Ab2, Ab3, & Ab4) used in this study. LRRK2 was not recognized in non-transfected cells (-). B. Recombinant LRRK2 (arrowhead) from transfected M17 cells is detected by anti-LRRK2 Ab4 (lane 1), but not after absorption of the antibody with its peptide antigen (lane 2). Brain LRRK2 was recognized by anti-LRRK2 Ab4 in two controls (lanes 3 & 4) & two PD cases (lanes 5 & 6). Cell lysates (10 μg protein) & brain homogenates (100 μg protein) were prepared & loaded on 6% SDS-PAGE gels for Western blot analysis using anti-LRRK2 antibodies Ab1–Ab4 in (A) & Ab4 in (B). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/17137507), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] -
Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - Immunocytochemistry of LRRK2 in DLB. Cortical LBs (arrows) in DLB were positive for LRRK2 using both Ab4 against LRRK22500–2527 (A), & Ab1 against LRRK2900–100 (B). Neuronal cytoplasm (C) was also strongly labelled throughout the cortex by Ab4 & with a consistently more granular pattern by Ab1 (inset in C) in cases of DLB. Control cases display similar, though less intense neuronal labeling (D). Biochemically purified cortical LBs were strongly positive for staining by Ab4 (E) as well as by anti-alpha -synuclein (F), while they were unstained when omitting primary antibody (G). Large vessels (H) were also intensely labelled by Ab4. Scale bars: A, B = 25 μm; C, D = 100 μm; E-G = 25 μm; H = 50 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/17137507), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] -
Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - Immunocytochemistry of LRRK2 in DLB. Cortical LBs (arrows) in DLB were positive for LRRK2 using both Ab4 against LRRK22500–2527 (A), & Ab1 against LRRK2900–100 (B). Neuronal cytoplasm (C) was also strongly labelled throughout the cortex by Ab4 & with a consistently more granular pattern by Ab1 (inset in C) in cases of DLB. Control cases display similar, though less intense neuronal labeling (D). Biochemically purified cortical LBs were strongly positive for staining by Ab4 (E) as well as by anti-alpha -synuclein (F), while they were unstained when omitting primary antibody (G). Large vessels (H) were also intensely labelled by Ab4. Scale bars: A, B = 25 μm; C, D = 100 μm; E-G = 25 μm; H = 50 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/17137507), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] -
Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - Immunocytochemistry of LRRK2 in DLB. Cortical LBs (arrows) in DLB were positive for LRRK2 using both Ab4 against LRRK22500–2527 (A), & Ab1 against LRRK2900–100 (B). Neuronal cytoplasm (C) was also strongly labelled throughout the cortex by Ab4 & with a consistently more granular pattern by Ab1 (inset in C) in cases of DLB. Control cases display similar, though less intense neuronal labeling (D). Biochemically purified cortical LBs were strongly positive for staining by Ab4 (E) as well as by anti-alpha -synuclein (F), while they were unstained when omitting primary antibody (G). Large vessels (H) were also intensely labelled by Ab4. Scale bars: A, B = 25 μm; C, D = 100 μm; E-G = 25 μm; H = 50 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/17137507), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: LRRK2 Antibody - BSA Free [NB300-268] -
Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 deletion attenuates Mn-induced pro-apoptotic protein levels.(A) After cells (LRRK2 WT & LRRK2 KO) were exposed to Mn (250 μM) for up to 24 h, total protein was extracted & followed by western blot analysis to determine protein levels of Bax & Daxx. beta -actin was used as a loading control. (B) The expression levels of Bax & Daxx modulated by Mn were quantified relative to its control levels in LRRK2 WT & KO cells. **, p < 0.01; ***, p < 0.001 (one-way ANOVA followed by Tukey’s post hoc test; n = 3). The data shown are representative of 3 independent experiments. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30645642), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] -
Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] - The interaction & phosphorylation of LRRK2 & LC1. (A) Cellular co-localisation of endogenous LRRK2 & MAP1B (LC1). SKNSH cells were labelled with LRRK2 (green) & LC1 (red) antibodies & signals were detected by immunofluorescence. Detailed figure in Additional file 1. (B) LRRK2 kinase assay. The LRRK2 wild-typed (WT) kinase interacted & phosphorylated the LC1 domain unlike the LRRK2 kinase-dead protein. (C) Western blotting of LRRK2 & LC1 proteins after a kinase assay. Both LRRK2 & LC1 proteins were probed for phospho-threonine & phospho-serine signals respectively. Image collected & cropped by CiteAb from the following publication (http://molecularbrain.biomedcentral.com/articles/10.1186/1756-6606-7-29), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: LRRK2 Antibody - BSA Free [NB300-268] -
Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - Mn increases LRRK2 kinase activity & expression in RAW 264.7 & HMC3 cells.(A) Two-hundred (200) μg of protein were collected from cell lysates, followed by western blot analysis to determine the presence of LRRK2 in LRRK2 WT, KO RAW 264.7 & HMC3 cells. beta -actin was used as a loading control. (B,C) LRRK2 WT or HMC3 cells were treated with Mn (250 μM) for the designated times, followed by protein extraction & western blot analysis as described in the Methods section. Protein levels of LRRK2 & phosphorylated LRRK2 (S1292) in LRRK2 WT RAW 264.7 (B) & HMC3 (C) cells were quantified. (D,E) Effect of Mn on LRRK2 mRNA levels in LRRK2 WT RAW 264.7 (D) & HMC3 (E) cells were assessed as described in the Methods section. GAPDH was used as a loading control. (F) After pre-treatment with LRRK2 inhibitors GSK (1 μM) & MLi-2 (50 nM) for 90 min, RAW 264.7 cells were exposed to Mn (250 μM) for 20 min, followed by western blot analysis to detect phosphorylation of LRRK2 (S1292). ###, p < 0.001; *, p < 0.05; **, p < 0.01; ***, p < 0.001 compared to the control (one-way ANOVA followed by Tukey’s post hoc test; n = 3). The data shown are representative of 3 independent experiments. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30645642), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] -
Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - Immunocytochemistry of LRRK2 in PD. Four antibodies raised against sequences corresponding to various regions shown on the schematic diagram of LRRK2 (red bars) were used on brainstem sections of PD (A-H) & age-matched controls (I-L). Intense immunolabeling of brainstem LBs in cases of PD was seen with Ab1 against LRRK2900–100 (A) & Ab4 against LRRK22500–2527(D). Both rim (A & D) & core (inset in D) of LBs were recognized. In contrast, LBs were not labelled in any case using antibodies directed against LRRK21246–1265 (Ab2, B) or LRRK21838–2133 (Ab3, C), for which the antigenic sites are located within the folded domains. The cell bodies of both pigmented & no-pigmented neurons (arrows) as well as axons (arrowheads) contain LRRK2, seen only using antibodies against sites outside the folded domains (Ab1, E & Ab4, H). In contrast, Ab4 staining was much less intense in control tissue (L). The muscle layer of both large & small vessels was consistently found to contain high levels of LRRK2 in almost all PD cases (seen in E, G, & H) & strikingly the vessels are the only structure immunolabeled with Ab3 recognizing LRRK21838–2133 in all cases studied (G & K). Ab2 to LRRK21246–1265 did not recognize LBs, cell bodies or vessels in any case (B, F, & J). Scale bars: A-D & inset = 10 μm; E-L = 100 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/17137507), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] -
Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - Immunocytochemistry of LRRK2 in PD. Four antibodies raised against sequences corresponding to various regions shown on the schematic diagram of LRRK2 (red bars) were used on brainstem sections of PD (A-H) & age-matched controls (I-L). Intense immunolabeling of brainstem LBs in cases of PD was seen with Ab1 against LRRK2900–100 (A) & Ab4 against LRRK22500–2527(D). Both rim (A & D) & core (inset in D) of LBs were recognized. In contrast, LBs were not labelled in any case using antibodies directed against LRRK21246–1265 (Ab2, B) or LRRK21838–2133 (Ab3, C), for which the antigenic sites are located within the folded domains. The cell bodies of both pigmented & no-pigmented neurons (arrows) as well as axons (arrowheads) contain LRRK2, seen only using antibodies against sites outside the folded domains (Ab1, E & Ab4, H). In contrast, Ab4 staining was much less intense in control tissue (L). The muscle layer of both large & small vessels was consistently found to contain high levels of LRRK2 in almost all PD cases (seen in E, G, & H) & strikingly the vessels are the only structure immunolabeled with Ab3 recognizing LRRK21838–2133 in all cases studied (G & K). Ab2 to LRRK21246–1265 did not recognize LBs, cell bodies or vessels in any case (B, F, & J). Scale bars: A-D & inset = 10 μm; E-L = 100 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/17137507), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: LRRK2 Antibody - BSA Free [NB300-268] -
Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - Mn increases LRRK2 kinase activity & expression in RAW 264.7 & HMC3 cells.(A) Two-hundred (200) μg of protein were collected from cell lysates, followed by western blot analysis to determine the presence of LRRK2 in LRRK2 WT, KO RAW 264.7 & HMC3 cells. beta -actin was used as a loading control. (B,C) LRRK2 WT or HMC3 cells were treated with Mn (250 μM) for the designated times, followed by protein extraction & western blot analysis as described in the Methods section. Protein levels of LRRK2 & phosphorylated LRRK2 (S1292) in LRRK2 WT RAW 264.7 (B) & HMC3 (C) cells were quantified. (D,E) Effect of Mn on LRRK2 mRNA levels in LRRK2 WT RAW 264.7 (D) & HMC3 (E) cells were assessed as described in the Methods section. GAPDH was used as a loading control. (F) After pre-treatment with LRRK2 inhibitors GSK (1 μM) & MLi-2 (50 nM) for 90 min, RAW 264.7 cells were exposed to Mn (250 μM) for 20 min, followed by western blot analysis to detect phosphorylation of LRRK2 (S1292). ###, p < 0.001; *, p < 0.05; **, p < 0.01; ***, p < 0.001 compared to the control (one-way ANOVA followed by Tukey’s post hoc test; n = 3). The data shown are representative of 3 independent experiments. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30645642), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: LRRK2 Antibody - BSA Free [NB300-268] -
Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - Mn increases LRRK2 kinase activity & expression in RAW 264.7 & HMC3 cells.(A) Two-hundred (200) μg of protein were collected from cell lysates, followed by western blot analysis to determine the presence of LRRK2 in LRRK2 WT, KO RAW 264.7 & HMC3 cells. beta -actin was used as a loading control. (B,C) LRRK2 WT or HMC3 cells were treated with Mn (250 μM) for the designated times, followed by protein extraction & western blot analysis as described in the Methods section. Protein levels of LRRK2 & phosphorylated LRRK2 (S1292) in LRRK2 WT RAW 264.7 (B) & HMC3 (C) cells were quantified. (D,E) Effect of Mn on LRRK2 mRNA levels in LRRK2 WT RAW 264.7 (D) & HMC3 (E) cells were assessed as described in the Methods section. GAPDH was used as a loading control. (F) After pre-treatment with LRRK2 inhibitors GSK (1 μM) & MLi-2 (50 nM) for 90 min, RAW 264.7 cells were exposed to Mn (250 μM) for 20 min, followed by western blot analysis to detect phosphorylation of LRRK2 (S1292). ###, p < 0.001; *, p < 0.05; **, p < 0.01; ***, p < 0.001 compared to the control (one-way ANOVA followed by Tukey’s post hoc test; n = 3). The data shown are representative of 3 independent experiments. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30645642), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: LRRK2 Antibody - BSA Free [NB300-268] -
Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - Mn increases LRRK2 kinase activity & expression in RAW 264.7 & HMC3 cells.(A) Two-hundred (200) μg of protein were collected from cell lysates, followed by western blot analysis to determine the presence of LRRK2 in LRRK2 WT, KO RAW 264.7 & HMC3 cells. beta -actin was used as a loading control. (B,C) LRRK2 WT or HMC3 cells were treated with Mn (250 μM) for the designated times, followed by protein extraction & western blot analysis as described in the Methods section. Protein levels of LRRK2 & phosphorylated LRRK2 (S1292) in LRRK2 WT RAW 264.7 (B) & HMC3 (C) cells were quantified. (D,E) Effect of Mn on LRRK2 mRNA levels in LRRK2 WT RAW 264.7 (D) & HMC3 (E) cells were assessed as described in the Methods section. GAPDH was used as a loading control. (F) After pre-treatment with LRRK2 inhibitors GSK (1 μM) & MLi-2 (50 nM) for 90 min, RAW 264.7 cells were exposed to Mn (250 μM) for 20 min, followed by western blot analysis to detect phosphorylation of LRRK2 (S1292). ###, p < 0.001; *, p < 0.05; **, p < 0.01; ***, p < 0.001 compared to the control (one-way ANOVA followed by Tukey’s post hoc test; n = 3). The data shown are representative of 3 independent experiments. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30645642), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: LRRK2 Antibody - BSA Free [NB300-268] -
Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 kinase inhibition blocks Tat-induced S935 phosphorylation & inflammatory cytokine expression in BV-2 cells. (A) Western blot depicting pS935-LRRK2 & total LRRK2 protein levels 12 hours after saline or Tat-treatment ± LRRK2 kinase inhibition (LRRK2i). (B) Tat treatment significantly increased pS935-LRRK2, which was attenuated by LRRK2i. (C) We measured TNF-alpha, IL-6, MCP-1 & IL-10 mRNA levels by qRT-PCR 12 hours post saline or Tat treatment ± LRRK2i. LRRK2i attenuated Tat-induced TNF-alpha, IL-6, MCP-1 & IL-10 expression for all groups (*P <0.05, ***P <0.001, one way ANOVA, Newman-Keuls post-test). ANOVA, analysis of variance; LRRK2, leucine-rich repeat kinase2; pS935, phosphorylation of serine 935; Tat, trans activator of transcription. Image collected & cropped by CiteAb from the following publication (http://jneuroinflammation.biomedcentral.com/articles/10.1186/1742-2094-…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] -
Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - Immunocytochemistry of LRRK2 in PD. Four antibodies raised against sequences corresponding to various regions shown on the schematic diagram of LRRK2 (red bars) were used on brainstem sections of PD (A-H) & age-matched controls (I-L). Intense immunolabeling of brainstem LBs in cases of PD was seen with Ab1 against LRRK2900–100 (A) & Ab4 against LRRK22500–2527(D). Both rim (A & D) & core (inset in D) of LBs were recognized. In contrast, LBs were not labelled in any case using antibodies directed against LRRK21246–1265 (Ab2, B) or LRRK21838–2133 (Ab3, C), for which the antigenic sites are located within the folded domains. The cell bodies of both pigmented & no-pigmented neurons (arrows) as well as axons (arrowheads) contain LRRK2, seen only using antibodies against sites outside the folded domains (Ab1, E & Ab4, H). In contrast, Ab4 staining was much less intense in control tissue (L). The muscle layer of both large & small vessels was consistently found to contain high levels of LRRK2 in almost all PD cases (seen in E, G, & H) & strikingly the vessels are the only structure immunolabeled with Ab3 recognizing LRRK21838–2133 in all cases studied (G & K). Ab2 to LRRK21246–1265 did not recognize LBs, cell bodies or vessels in any case (B, F, & J). Scale bars: A-D & inset = 10 μm; E-L = 100 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/17137507), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268] -
Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268] - Inhibition of LRRK2 kinase activity attenuates Mn-induced apoptosis.(A) After pre-treatment with GSK (1 μM) for 90 min, cells (HMC3) were exposed to Mn (250 μM) for the designated time periods, followed by annexin V & PI staining & flow cytometry analysis to determine apoptosis. Early & late apoptotic cells (Q2 & Q3) were analyzed. (B,C) After pre-treatment with GSK (1 μM) for 90 min, cells (LRRK2 WT RAW 264.7 & HMC3) were exposed to Mn for designated time periods, followed by the MTT assay to determine cell viability, as described in the Methods section, (@@@, p < 0.001; *, p < 0.05; ***, p < 0.001 compared to the control (one-way ANOVA followed by Tukey’s post hoc test; n = 3, apoptosis assay; n = 6, MTT assay). The data shown are representative of 3 independent experiments. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30645642), licensed under a CC-BY license. Not internally tested by Novus Biologicals.LRRK2 in U-2 OS Human Cell Line.
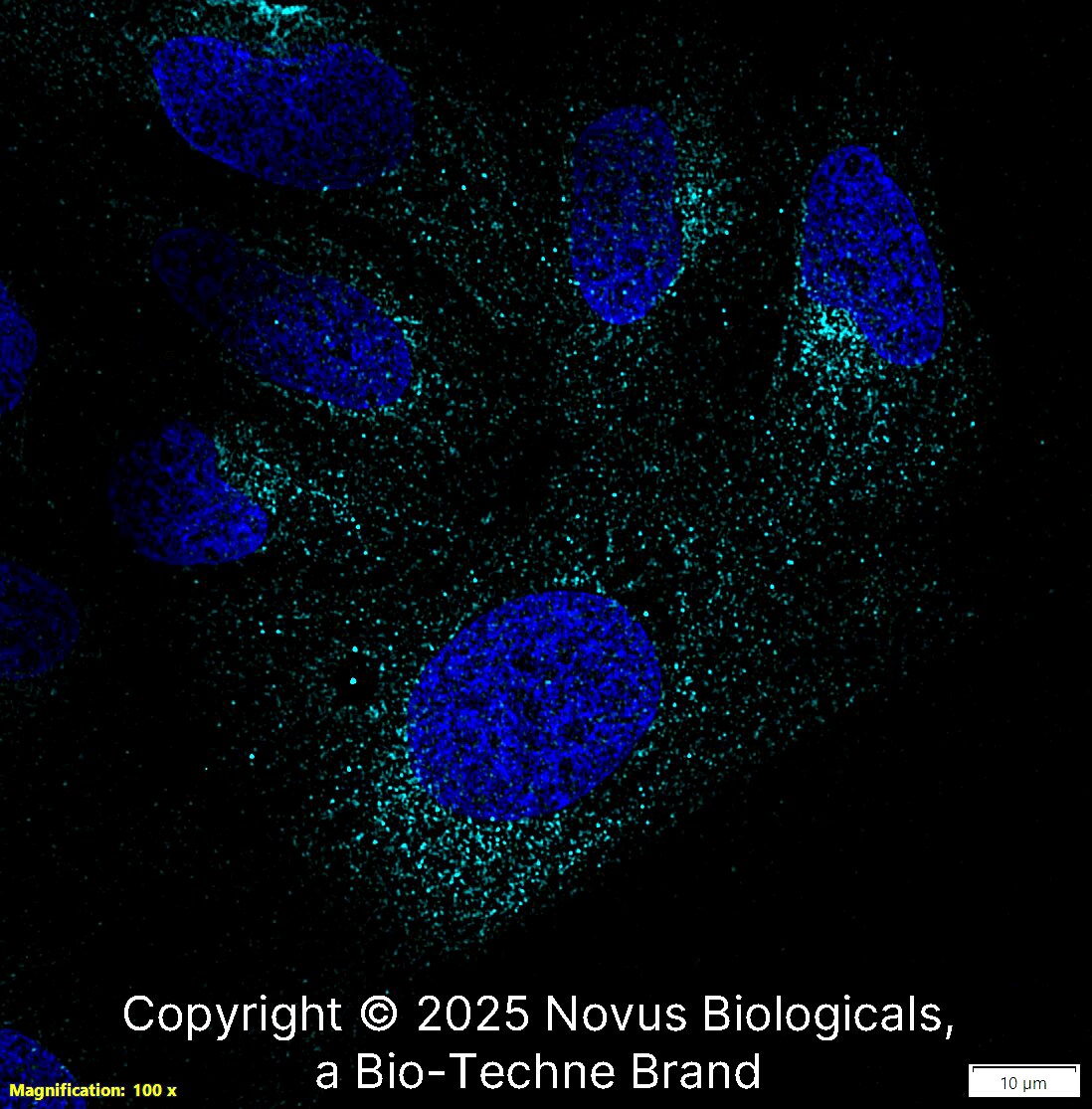
LRRK2 was detected in immersion fixed U-2 OS human osteosarcoma cell line using Rabbit anti-LRRK2 Antigen Affinity Purified Polyclonal Antibody conjugated to Alexa Fluor® 647 (Catalog # NB300-268AF647) (light blue) at 10 µg/mL overnight at 4C. Cells were counterstained with DAPI (blue). Cells were imaged using a 100X objective and digitally deconvolved.Applications for LRRK2 Antibody - BSA Free
Flow (Intracellular)
Flow Cytometry
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Paraffin
Immunoprecipitation
Western Blot
Reviewed Applications
Read 1 review rated 5 using NB300-268 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: LRRK2
Long Name
Alternate Names
Gene Symbol
UniProt
Additional LRRK2 Products
Product Documents for LRRK2 Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for LRRK2 Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for LRRK2 Antibody - BSA Free
Customer Reviews for LRRK2 Antibody - BSA Free (1)
Have you used LRRK2 Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Immunohistochemistry-FrozenSample Tested:Species: HumanVerified Customer | Posted 01/28/2014
There are no reviews that match your criteria.
Protocols
View specific protocols for LRRK2 Antibody - BSA Free (NB300-268):
Sample Preparation.
1. Grow cells to 60-85% confluency. Flow cytometry requires between 2 x 105 and 1 x 106 cells for optimal performance.
2. If cells are adherent, harvest gently by washing once with staining buffer and then scraping. Avoid using trypsin as this can disrupt certain epitopes of interest. If enzymatic harvest is required, use Accutase, Collagenase, or TrypLE Express for a less damaging option.
3. Reserve 100 uL for counting, then transfer cell volume into a 50 mL conical tube and centrifuge for 8 minutes at 400 RCF.
a. Count cells using a hemocytometer and a 1:1 trypan blue exclusion stain to determine cell viability before starting the flow protocol. If cells appear blue, do not proceed.
4. Re-suspend cells to a concentration of 1 x 106 cells/mL in staining buffer (NBP2-26247).
5. Aliquot out 1 mL samples in accordance with your experimental samples.
Tip: When cell surface and intracellular staining are required in the same sample, it is advisable that the cell surface staining be performed first since the fixation and permeablization steps might reduce the availability of surface antigens.
Intracellular Staining.
Tip: When performing intracellular staining, it is important to use appropriate fixation and permeabilization reagents based upon the target and its subcellular location. Generally, our Intracellular Flow Assay Kit (NBP2-29450) is a good place to start as it contains an optimized combination of reagents for intracellular staining as well as an inhibitor of intracellular protein transport (necessary if staining secreted proteins). Certain targets may require more gentle or transient permeabilization protocols such as the commonly employed methanol or saponin-based methods.
Protocol for Cytoplasmic Targets:
Optional: Perform cell surface staining as described in the previous section.
1. Fix the cells by adding 100 uL fixation solution (such as 4% PFA) to each sample for 10-15 minutes.
2. Permeabilize cells by adding 100 uL of a permeabization buffer to every 1 x 106 cells present in the sample. Mix well and incubate at room temperature for 15 minutes.
a. For cytoplasmic targets, use a gentle permeabilization solution such as 1X PBS + 0.5% Saponin or 1X PBS + 0.5% Tween-20.
b. To maintain the permeabilized state throughout your experiment, use staining buffer + 0.1% of the permeabilization reagent (i.e. 0.1% Tween-20 or 0.1% Saponin).
3. Following the 15 minute incubation, add 2 mL of the staining buffer + 0.1% permeabilizer to each sample.
4. Centrifuge for 5 minutes at 400 RCF.
5. Discard supernatant and re-suspend in 1 mL of staining buffer + 0.1% permeabilizer.
6. Stain each sample at 1 uL/ 1 x 106 cells of primary antibody or 1-3 uL/ 1 x 106 cells for directly conjugated antibodies. Mix well and incubate at room temperature for 30 minutes- 1 hour. Gently mix samples every 10-15 minutes.
7. Following the primary/conjugate incubation, add 2 mL/sample of staining buffer +0.1% permeabilizer and centrifuge for 5 minutes at 400 RCF.
8. Remove supernatant and re-suspend each sample in 2 mL staining buffer + 0.1% permeabilizer, repeat wash for 5 minutes at 400 RCF.
9. If using a directly conjugated antibody, after the second wash, re-suspend cell pellet to a final volume of 500 uL per sample and proceed with flow analysis.
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and wash the cells briefly in PBS. Add 10% formalin to the dish and fix at room temperature for 10 minutes.
2. Remove the formalin and wash the cells in PBS.
3. Permeablize the cells with 0.1% Triton X100 or other suitable detergent for 10 min.
4. Remove the permeablization buffer and wash three times for 10 minutes each in PBS. Be sure to not let the specimen dry out.
5. To block nonspecific antibody binding, incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
6. Add primary antibody at appropriate dilution and incubate overnight at 4C.
7. Remove primary antibody and replace with PBS. Wash three times for 10 minutes each.
8. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
9. Remove secondary antibody and replace with PBS. Wash three times for 10 minutes each.
10. Counter stain DNA with DAPi if required.
Antigen Unmasking:
Bring slides to a boil in 10 mM sodium citrate buffer (pH 6.0) then maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench-top for 30 minutes (keep slides in the sodium citrate buffer at all times).
Staining:
1. Wash sections in deionized water three times for 5 minutes each.
2. Wash sections in PBS for 5 minutes.
3. Block each section with 100-400 ul blocking solution (1% BSA in PBS) for 1 hour at room temperature.
4. Remove blocking solution and add 100-400 ul diluted primary antibody. Incubate overnight at 4 C.
5. Remove antibody solution and wash sections in wash buffer three times for 5 minutes each.
6. Add 100-400 ul HRP polymer conjugated secondary antibody. Incubate 30 minutes at room temperature.
7. Wash sections three times in wash buffer for 5 minutes each.
8. Add 100-400 ul DAB substrate to each section and monitor staining closely.
9. As soon as the sections develop, immerse slides in deionized water.
10. Counterstain sections in hematoxylin.
11. Wash sections in deionized water two times for 5 minutes each.
12. Dehydrate sections.
13. Mount coverslips.
Protocol for immunoprecipitation of LRRK2 followed by LRRK2 autophosphorylation kinase assay
Cell lysis
3X15 cm plates of SH-SY5Y cells were grown to 80% confluency. The plates were washed twice with PBS and placed on ice. Remaining PBS was aspirated off after tilting plate to remove all PBS. 1.5 ml of cold lysis buffer (buffer A) was added to each plate. The plates were allowed to incubate on ice 5 min until the cells detached. The lysis buffer and cells for each plate were then vigorously passed 6X through a 30.5 guage needle. Lysis buffer and cells were transferred to 3X1.5 ml eppendorf tubes and spun 5 min. at 5,000 rpm in a 4 degree eppendorf microfuge. Lysates were removed from pelleted debris and transferred to new 1.5 ml eppendorf tubes and recentrifuged 10 min. at 13,000 rpm. Lysate was transferred to three new tubes and 1/2 lysate volume of buffer A (-) NaCl was added to each tube.
Preclear
10 ug of rabbit IgG were added to the lysate for each tube and the lysate was vortexed followed by rotating at 4 degrees for 2 hours. 20 ul of protein A sepharose beads (Amersham cat#: 17-0469-01) were added. The tubes were vortexed and then rotated for 1.5 hours at 4 degrees. Lysates were separated from protein A beads beads by low (200 rpm) spin for 2 min and transferred to new eppendorf tubes. A repeat of the protein A sepharose incubation was carried out to remove residual rabbit IgG followed by removal of the protein A beads.
Immunoprecipitation with LRRK2 Ab
To two of the tubes containing precleared lysate were added 7 ul of LRRK2 Ab (7ug). To the remaining tube was added 7ug of rabbit IgG. The tubes were vortexed and allowed to rotate overnight at 4 degrees. The following morning 30 ul of protein A sepharose was added to each tube, the tubes were vortexed and rotated at 4 degrees for 2 hours. The protein A beads were then isolated by brief, low speed centrifugation and were washed 3X in 500ul buffer A (-) NaCl. This was followed by two washes in kinase buffer (buffer B). Protein A beads were resuspended in 1 volume (30 ul) of buffer B for a total of 60ul of immunoprecipitate.
Autophosphorylation kinase reaction, gel electrophoresis and phosphoimaging
On ice, 40 ul of immunoprecipitate from each tube was transferred to a.5ml kinase reaction tube. Each of the three reactions was supplemented with a 5 ul mixture that gave a final reaction concentration of 15 uM cold ATP and 5uCi ATP. The reaction mixtures were vortexed and transferred to a rotator in a 30 degree incubator. The autophosphorylation incubation was allowed to go for 30 minutes and the reaction tubes were taken off the rotator and vortexed every five minutes. The reactions were then halted by addition of 11ul of 5X SDS gel running sample buffer to each of the three samples. 40ul of each sample was then run on a 7% acrylamide-acetate mini-gel. Once the 200Kd molecular weight marker band had run half way down the gel, the gel was stopped dried and exposed blanked to a phosphoimaging cassette (Molecular Dynamics). Following 24 hour exposure, the cassette was assessed for radioactivity on a Storm analyzer.
Buffers
Buffer A (make 10ml both - and + NaCl solutions) = lysis buffer
50mM Tris pH 7.4
150mM NaCl
0.2% NP40
Protease inhibitor cocktail (stock = 100X, Sigma)
0.5mM vanadate
15mM EDTA
adjust to 10 ml with H2O
Buffer B = kinase buffer
10mM hepes
10mM MgCl2
50mM NaCl
protease inhibitor
vanadate
(1mM NaN3 if storing overnight or longer)
1. Perform SDS-PAGE on samples to be analyzed, loading 10-25 ug of total protein per lane.
2. Transfer proteins to PVDF membrane according to the instructions provided by the manufacturer of the membrane and transfer apparatus.
3. Stain the membrane with Ponceau S (or similar product) to assess transfer success, and mark molecular weight standards where appropriate.
4. Rinse the blot TBS -0.05% Tween 20 (TBST).
5. Block the membrane in 5% Non-fat milk in TBST (blocking buffer) for at least 1 hour.
6. Wash the membrane in TBST three times for 10 minutes each.
7. Dilute primary antibody in blocking buffer and incubate overnight at 4C with gentle rocking.
8. Wash the membrane in TBST three times for 10 minutes each.
9. Incubate the membrane in diluted HRP conjugated secondary antibody in blocking buffer (as per manufacturer's instructions) for 1 hour at room temperature.
10. Wash the blot in TBST three times for 10 minutes each (this step can be repeated as required to reduce background).
11. Apply the detection reagent of choice in accordance with the manufacturer's instructions.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for LRRK2 Antibody - BSA Free
-
Q: I am wondering what concentration of Lrrk2 blocking peptide (NB300-268PEP) I should use for FACS as a control?
A:
We do have a protocol for how to incubate the primary with the blocking peptide to perform a negative control competition assay. While this was written with WB and IHC in mind, it should work the same in inhibiting cell binding for flow.
-
Q: Is the immunogen of NB300-268 within amino acids 922 and 1143 of LRRK2 (Uniprot ID#Q5S007)?
A:
The immunogen used to generate NB300-268 was a synthetic peptide made to a C-terminal region within residues 2500-2527 of the human LRRK2 protein (UniProt# Q5S007). We do have an alternative antibody that you may be interested in, with catalog number NB110-55289. The immunogen used to generate this product was synthetic peptide made to an internal region of the mouse LRRK2 protein sequence (between residues 900-1000), although this does not recognize human LRRK2. If you are looking to detect mouse LRRK2 this would be a suitable product for you.
-
Q: I am wondering what concentration of Lrrk2 blocking peptide (NB300-268PEP) I should use for FACS as a control?
A:
We do have a protocol for how to incubate the primary with the blocking peptide to perform a negative control competition assay. While this was written with WB and IHC in mind, it should work the same in inhibiting cell binding for flow.
-
Q: Is the immunogen of NB300-268 within amino acids 922 and 1143 of LRRK2 (Uniprot ID#Q5S007)?
A:
The immunogen used to generate NB300-268 was a synthetic peptide made to a C-terminal region within residues 2500-2527 of the human LRRK2 protein (UniProt# Q5S007). We do have an alternative antibody that you may be interested in, with catalog number NB110-55289. The immunogen used to generate this product was synthetic peptide made to an internal region of the mouse LRRK2 protein sequence (between residues 900-1000), although this does not recognize human LRRK2. If you are looking to detect mouse LRRK2 this would be a suitable product for you.

![Immunohistochemistry-Frozen: LRRK2 Antibody - BSA Free [NB300-268] Immunohistochemistry-Frozen: LRRK2 Antibody - BSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Immunohistochemistry-Frozen-NB300-268-img0014.jpg)
![Western Blot: LRRK2 AntibodyBSA Free [NB300-268] Western Blot: LRRK2 AntibodyBSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Western-Blot-NB300-268-img0028.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Immunohistochemistry-Paraffin-NB300-268-img0026.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Immunohistochemistry-Paraffin-NB300-268-img0025.jpg)
![Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268] Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Flow-Cytometry-NB300-268-img0019.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Immunohistochemistry-Paraffin-NB300-268-img0024.jpg)
![Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Immunocytochemistry-Immunofluorescence-NB300-268-img0031.jpg)
![Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268] Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Flow-Cytometry-NB300-268-img0030.jpg)
![Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Immunocytochemistry-Immunofluorescence-NB300-268-img0015.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Immunohistochemistry-Paraffin-NB300-268-img0020.jpg)
![Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268] Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Flow-Cytometry-NB300-268-img0022.jpg)
![Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268] Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268]](https://resources.rndsystems.com/images/products/LRRK2-Antibody-Flow-Cytometry-NB300-268-img0029.jpg)
![Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-27122023125330.jpg)
![Immunocytochemistry/Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-271220231253364.jpg)
![Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202415371926.jpg)
![Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-31020241539739.jpg)
![Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-31020241538242.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202415392599.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202415304221.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-31020241534387.jpg)
![Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-31020241517133.jpg)
![Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202415304292.jpg)
![Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-31020241536376.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202415175254.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202415345320.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202415384173.jpg)
![Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-31020241535611.jpg)
![Immunocytochemistry/ Immunofluorescence: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202415382414.jpg)
![Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-31020241621814.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202416205168.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202416205158.jpg)
![Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202416205187.jpg)
![Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-31020241621810.jpg)
![Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-31020241621239.jpg)
![Western Blot: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-310202416212357.jpg)
![Immunohistochemistry-Paraffin: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-31020241621837.jpg)
![Flow Cytometry: LRRK2 Antibody - BSA Free [NB300-268] - LRRK2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb300-268_rabbit-polyclonal-lrrk2-antibody-31020241621844.jpg)