Amphiregulin (AR), also known as Schwannoma-derived growth factor (SDGF), is a member of the epidermal growth factor (EGF) family of growth factors which includes, AR, EGF, transforming growth factor-a (TGF-a), heparin binding EGF-like growth factor (HB-EGF), betacellulin (BTC), epiregulin, and the neuregulins-1 through -4. All EGF family members are synthesized as type I transmembrane precursors and contain one or several EGF domains in their extracellular region. The bioactive form of the proteins is released by proteolytic cleavage. The ErbB family of receptors that includes ErbB1‑B4, mediates the biological activities of the EGF family ligands. AR was originally isolated from the conditioned media of PMA-treated MCF-7 human breast carcinoma cell line. AR mRNA expression can be detected in numerous carcinoma cell lines and in the epithelial cells of various human tissues including colon, stomach, breast, ovary and kidney. AR stimulates the proliferation of keratinocytes, mammary epithelial cells, fibroblasts, astrocytes and glial cells. AR is also a growth inhibitor for certain tumor cells. The gene for AR has been mapped to human chromosome 4q13-q21 and mouse chromosome 5. Human and mouse AR cDNA encode 252 and 248 amino acid residue type I membrane proteins, respectively. The two proteins share approximately 69% sequence identity. Mouse AR also shares 81% amino acid sequence homology with rat AR. Several secreted isoforms of AR that vary in length and/or glycosylation level can be found in cell conditioned media. The 98 amino acid residue recombinant AR has better receptor binding and biological activity than the C-terminal truncated forms of the protein.
Mouse Amphiregulin Antibody
R&D Systems | Catalog # AF989

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Mouse, Guinea Pig, Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, ELISA Capture (Matched Antibody Pair), Neutralization
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Neutralization, Flow Cytometry, In vivo assay
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse Amphiregulin
Ser94-Lys191
Accession # P31955
Ser94-Lys191
Accession # P31955
Specificity
Detects mouse Amphiregulin in ELISAs and Western Blots. In sandwich immunoassays, less than 1% cross-reactivity with recombinant human Amphiregulin is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse Amphiregulin Antibody
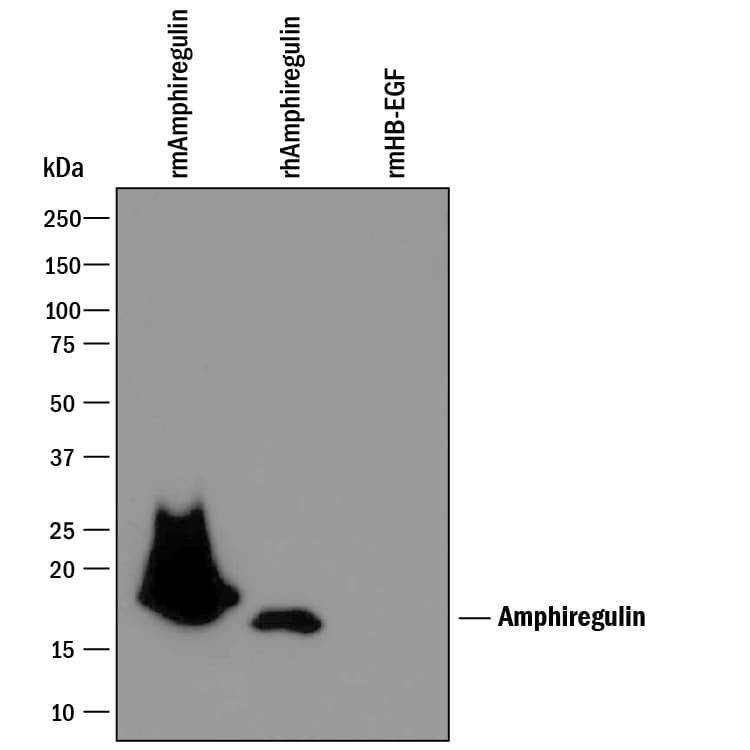
Detection of Recombinant Human and Mouse Amphiregulin by Western Blot.
Western blot shows 25 ng of Recombinant Mouse Amphiregulin (Catalog # 989-AR), Recombinant Human Amphiregulin (Catalog # 262-AR), and Recombinant Mouse HB-EGF. PVDF Membrane was probed with 0.1 µg/mL of Goat Anti-Mouse Amphiregulin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF989) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). A specific band was detected for Amphiregulin at approximately 16-25 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 3.Amphiregulin in Mouse Skin.
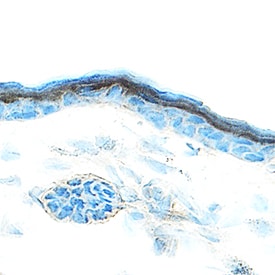
Amphiregulin was detected in immersion fixed frozen sections of mouse skin using Goat Anti-Mouse Amphiregulin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF989) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to keratinocytes. View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Cell Proliferation Induced by Amphiregulin and Neutralization by Mouse Amphiregulin Antibody.
Recombinant Mouse Amphiregulin (Catalog # 989-AR) stimulates proliferation in the Balb/3T3 mouse embryonic fibroblast cell line in a dose-dependent manner (orange line). Proliferation elicited by Recombinant Mouse Amphiregulin (50 ng/mL) is neutralized (green line) by increasing concentrations of Mouse Amphiregulin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF989). The ND50 is typically 0.3-1.5 µg/mL.Detection of Amphiregulin by Immunocytochemistry/ Immunofluorescence
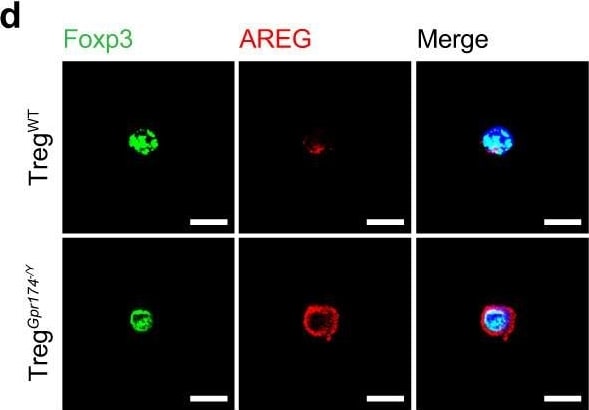
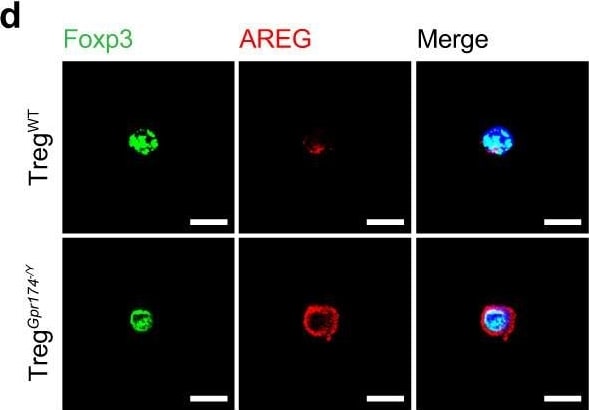
GPR174 regulates neovascularization by inhibiting AREG expression in Tregs. d Representative immunofluorescent images of AREG (red) & DAPI (blue) staining in Tregs isolated from ischemic muscle of WT & Gpr174−/Y mice 7 days after HLI. Scale bar, 10 μm. Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36473866), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Flow Cytometry
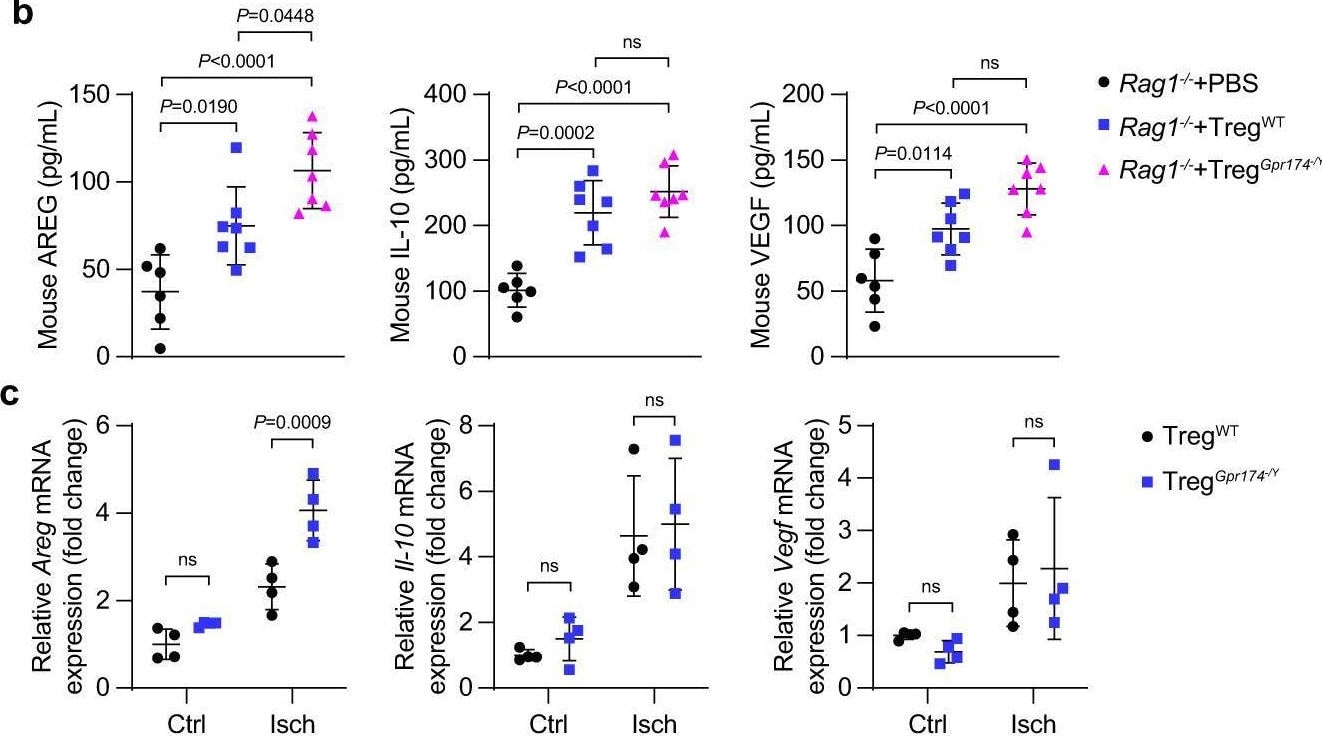
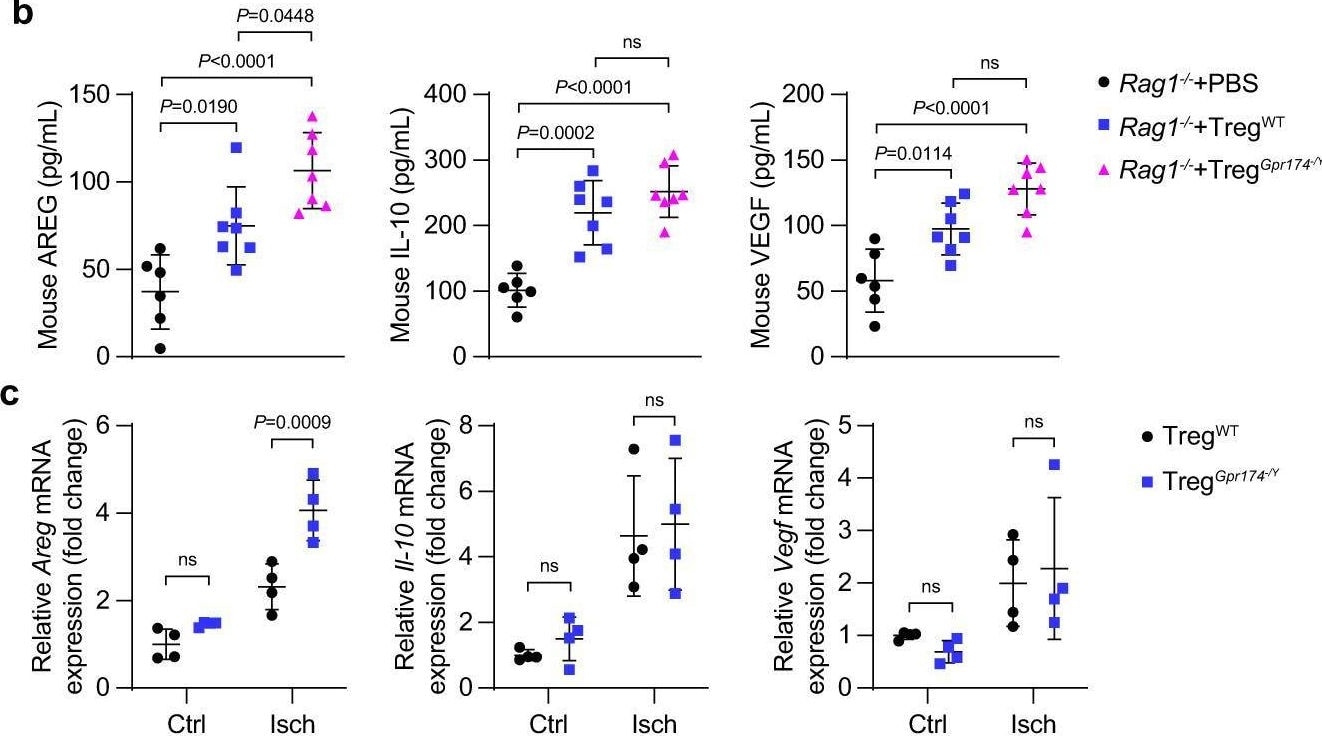
GPR174 regulates neovascularization by inhibiting AREG expression in Tregs. b Serum AREG, IL-10, & VEGF protein content in Rag1−/− mice receiving Tregs 7 days after adoptive transplantation experiments (n = 6 for PBS → Rag1−/− mice; n = 7 for wild-type Tregs→Rag1−/− mice; n = 7 for GPR174-deficient Tregs→Rag1−/− mice). Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36473866), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Immunocytochemistry/ Immunofluorescence
GPR174 regulates neovascularization by inhibiting AREG expression in Tregs. d Representative immunofluorescent images of AREG (red) & DAPI (blue) staining in Tregs isolated from ischemic muscle of WT & Gpr174−/Y mice 7 days after HLI. Scale bar, 10 μm. Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36473866), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Flow Cytometry
GPR174 regulates neovascularization by inhibiting AREG expression in Tregs. b Serum AREG, IL-10, & VEGF protein content in Rag1−/− mice receiving Tregs 7 days after adoptive transplantation experiments (n = 6 for PBS → Rag1−/− mice; n = 7 for wild-type Tregs→Rag1−/− mice; n = 7 for GPR174-deficient Tregs→Rag1−/− mice). Image collected & cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36473866), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Western Blot
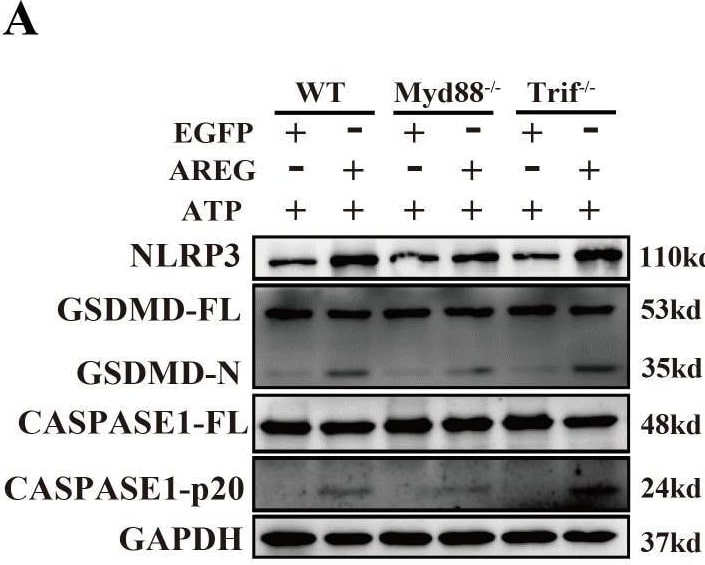
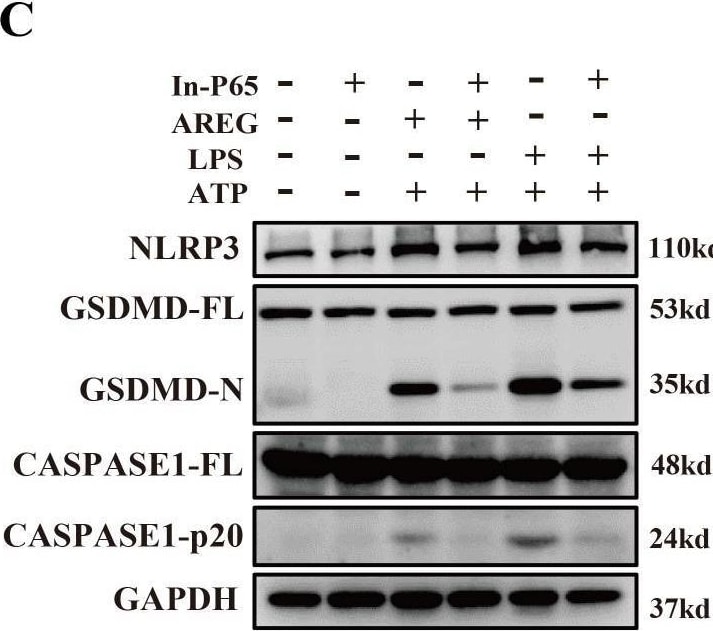
MyD88 silencing and NF kappa B inhibition restrain extracellular AREG-induced macrophage pyroptosis. NLRP3, CASPASE1-p20, and GSDMD-N expression levels were detected in extracellular AREG-induced Myd88−/− and Trif−/−BMDM via Western blot (A, B). NLRP3, CASPASE1-p20, and GSDMD-N expression levels were detected in the inhibitor of NF kappa B (P65) (20 μM) pretreating extracellular AREG-induced BMDM for 2 h via Western blot (C, D). Data are presented as mean ± SEM (n ≥ 3). *P < 0.05, **P < 0.01, ***P < 0.001 vs. Control. ns, no significient; AREG, amphiregulin; BMDM, bone marrow-derived macrophages; EGFR, epidermal growth factor receptor; GSDMD, gasdermin D. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40292295), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Western Blot
MyD88 silencing and NF kappa B inhibition restrain extracellular AREG-induced macrophage pyroptosis. NLRP3, CASPASE1-p20, and GSDMD-N expression levels were detected in extracellular AREG-induced Myd88−/− and Trif−/−BMDM via Western blot (A, B). NLRP3, CASPASE1-p20, and GSDMD-N expression levels were detected in the inhibitor of NF kappa B (P65) (20 μM) pretreating extracellular AREG-induced BMDM for 2 h via Western blot (C, D). Data are presented as mean ± SEM (n ≥ 3). *P < 0.05, **P < 0.01, ***P < 0.001 vs. Control. ns, no significient; AREG, amphiregulin; BMDM, bone marrow-derived macrophages; EGFR, epidermal growth factor receptor; GSDMD, gasdermin D. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40292295), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Western Blot
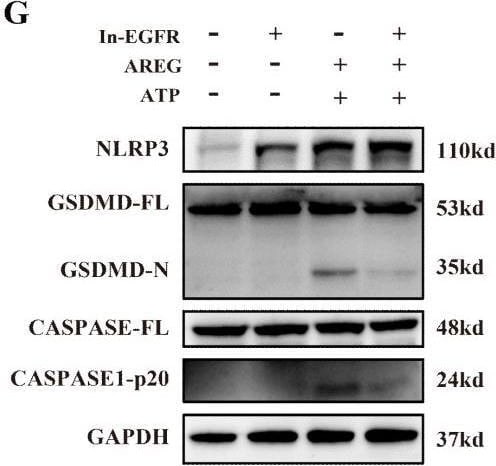
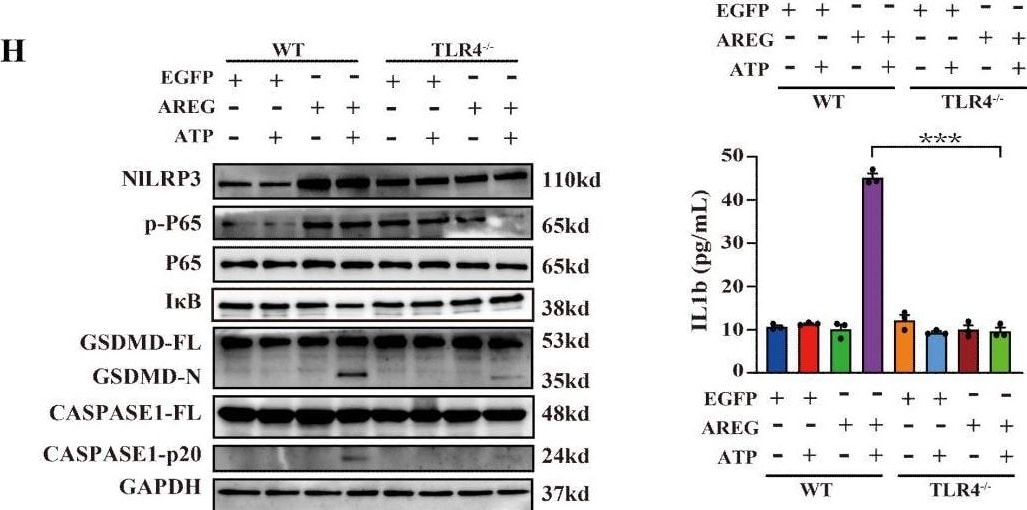
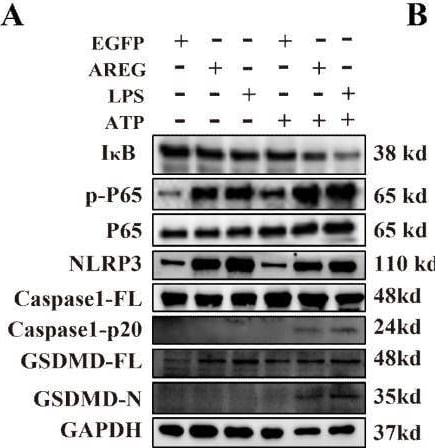
EGFR inhibition and TLR4 silencing impair AREG-induced macrophage pyroptosis. BMDM was stimulated with AREG+ATP or LPS+ATP, and the expression of NLRP3, p-P65, p-I kappa B, CASPASE-1-p20, and GSDMD-N was detected via Western blot (A-C). BMDM was stimulated with AREG+ATP or LPS+ATP, and oligomerization of ASC was detected using immunofluorescence (D, E). Experimental diagram of AREG-induced macrophage pyroptosis. For the priming step, BMDM was treated with AREG for 2.5 as the first signal and the ATP as the second signal (F). NLRP3, CASPASE-1-p20, and GSDMD-N expressions were detected in the EGFR inhibitor (1 mM) pretreating AREG +ATP-induced BMDM for 4 h through Western blot (G, J). NLRP3, CASPASE-1-p20, and GSDMD-N expressions were detected in AREG +ATP-induced TLR4−/−BMDM via Western blot (H, K). The expression of IL-1b and IL-18 was detected in the supernatant of AREG +ATP-induced TLR4−/−BMDM via ELISA (I). Data are presented as mean ± SEM (n ≥ 3). *P < 0.05, **P < 0.01, ***P < 0.001 vs. Control. ns, no significant; AREG, amphiregulin; BMDM, bone marrow-derived macrophages; EGFR, epidermal growth factor receptor; GSDMD, gasdermin D; ATP, adenosine triphosphate. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40292295), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Immunocytochemistry/ Immunofluorescence
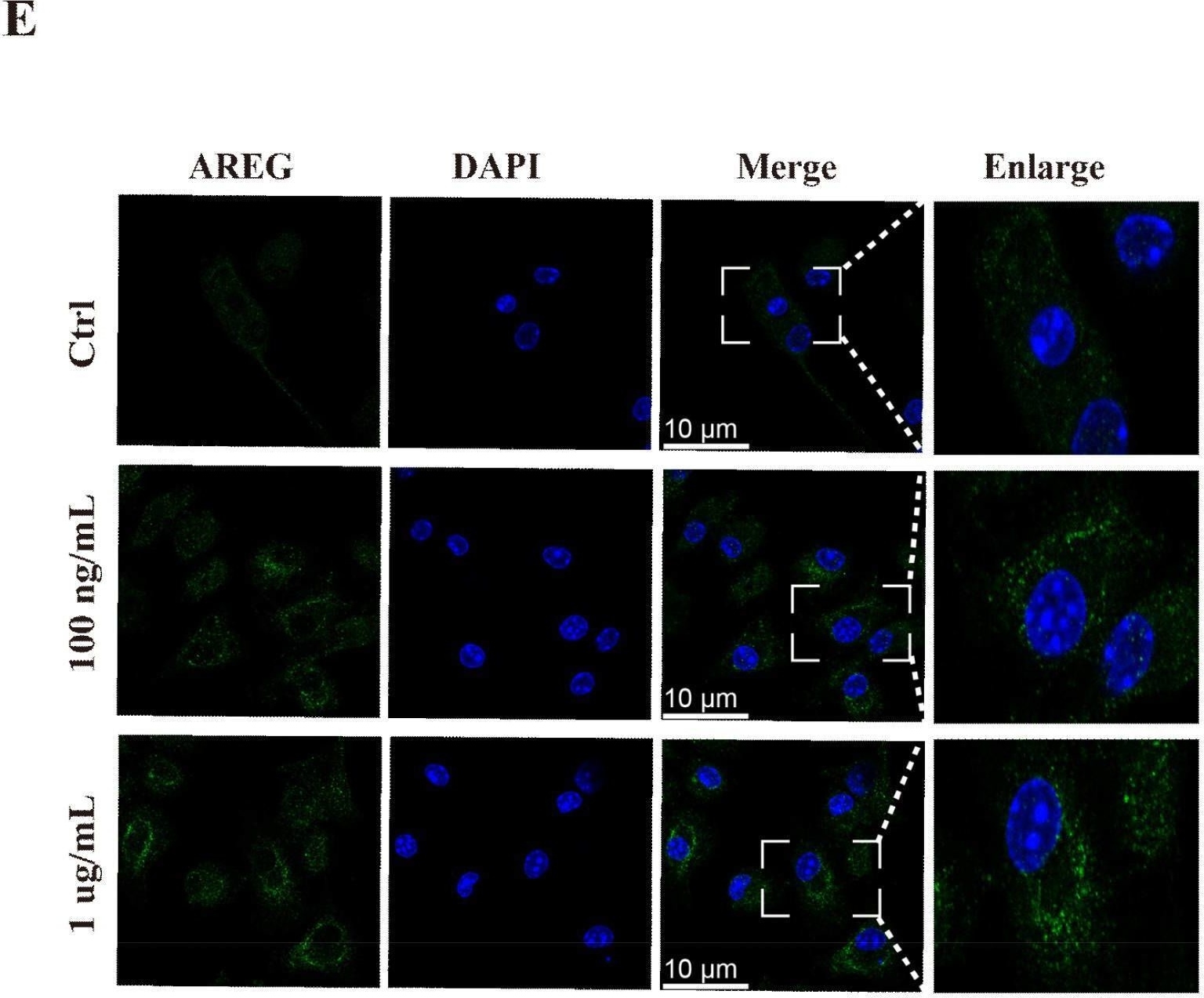
Dynamic expression of AREG in sepsis. RAW264.7 cells were stimulated with LPS (100 ng/mL or 1 μg/mL) for 1, 3, 6, 12, and 24 h. AREG mRNA and protein expressions were detected using RT-PCR and ELISA (A-C). WT C57BL/6 mice were intraperitoneally injected with LPS (20 mg/kg) and constructed with the CLP model, whereas AREG protein expression in serum was detected via ELISA (D). BMDM was stimulated with LPS (100 ng/mL or 1 μg/mL) for 12 h, and AREG protein expression was detected via Immunofluorescence (E). Data presented as mean ± SEM (n ≥ 3). *P < 0.05, **P < 0.01, ***P < 0.001 vs. Control. AREG, amphiregulin; WT, wild type; BMDM, bone marrow-derived macrophages; CLP, cecal ligation and puncture. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40292295), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Western Blot
EGFR inhibition and TLR4 silencing impair AREG-induced macrophage pyroptosis. BMDM was stimulated with AREG+ATP or LPS+ATP, and the expression of NLRP3, p-P65, p-I kappa B, CASPASE-1-p20, and GSDMD-N was detected via Western blot (A-C). BMDM was stimulated with AREG+ATP or LPS+ATP, and oligomerization of ASC was detected using immunofluorescence (D, E). Experimental diagram of AREG-induced macrophage pyroptosis. For the priming step, BMDM was treated with AREG for 2.5 as the first signal and the ATP as the second signal (F). NLRP3, CASPASE-1-p20, and GSDMD-N expressions were detected in the EGFR inhibitor (1 mM) pretreating AREG +ATP-induced BMDM for 4 h through Western blot (G, J). NLRP3, CASPASE-1-p20, and GSDMD-N expressions were detected in AREG +ATP-induced TLR4−/−BMDM via Western blot (H, K). The expression of IL-1b and IL-18 was detected in the supernatant of AREG +ATP-induced TLR4−/−BMDM via ELISA (I). Data are presented as mean ± SEM (n ≥ 3). *P < 0.05, **P < 0.01, ***P < 0.001 vs. Control. ns, no significant; AREG, amphiregulin; BMDM, bone marrow-derived macrophages; EGFR, epidermal growth factor receptor; GSDMD, gasdermin D; ATP, adenosine triphosphate. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40292295), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Western Blot
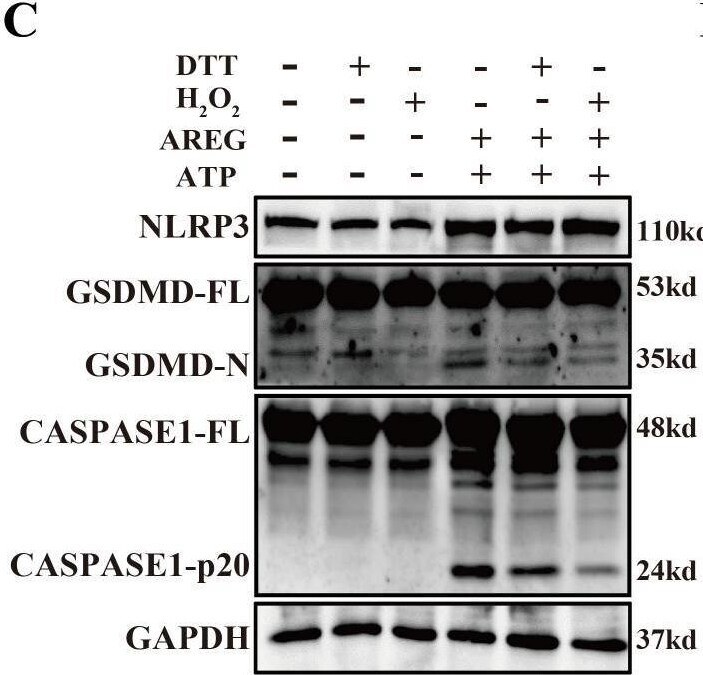
DTT-pretreated extracellular AREG restrains macrophage pyroptosis. NLRP3, CASPASE1-p20, and GSDMD-N expressions were detected in LPS-induced BMDM after DTT (1 mM) or H2O2 (100 μM) pretreating LPS for 1 h (A, B). NLRP3, Caspase1-p20, and GSDMD-N expression were detected in AREG-induced BMDM after DTT (1 mM) or H2O2 (100 μM) pretreating AREG for 1 h (C, D). Data are presented as mean ± SEM (n ≥ 3).*P < 0.05, **P < 0.01, ***P < 0.001 vs. Control. ns, no significient; AREG, amphiregulin; BMDM, bone marrow-derived macrophages; EGFR, epidermal growth factor receptor; GSDMD, gasdermin D. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40292295), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Western Blot
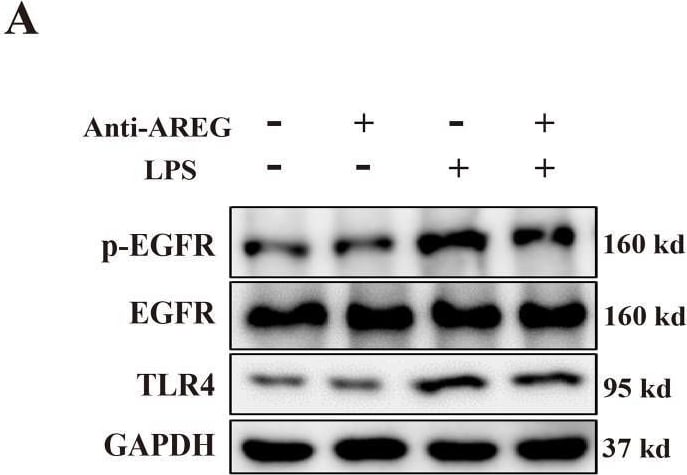
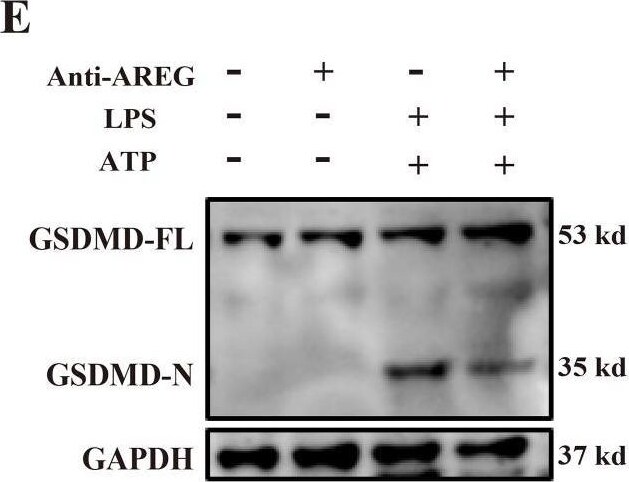
Neutralizing extracellular AREG decreases LPS-induced TLR4 expression and pyroptosis in macrophages. LPS-induced BMDM was pretreated with a neutralizing antibody of AREG. p-EGFR, TLR4, and GSDMD-N expression levels were detected via Western blot and immunofluorescence (A-C, E, F), TLR4 expression and ASC oligomerization was detected through immunofluorescence (D, G, H). Formation of pyrosomes (red arrows) was detected using electron microscopy, scale bars, 2 μm (I). Data are presented as mean ± SEM (n ≥ 3).*P < 0.05, **P < 0.01, ***P < 0.001 vs. Control. AREG, amphiregulin. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40292295), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Western Blot
EGFR inhibition and TLR4 silencing impair AREG-induced macrophage pyroptosis. BMDM was stimulated with AREG+ATP or LPS+ATP, and the expression of NLRP3, p-P65, p-I kappa B, CASPASE-1-p20, and GSDMD-N was detected via Western blot (A-C). BMDM was stimulated with AREG+ATP or LPS+ATP, and oligomerization of ASC was detected using immunofluorescence (D, E). Experimental diagram of AREG-induced macrophage pyroptosis. For the priming step, BMDM was treated with AREG for 2.5 as the first signal and the ATP as the second signal (F). NLRP3, CASPASE-1-p20, and GSDMD-N expressions were detected in the EGFR inhibitor (1 mM) pretreating AREG +ATP-induced BMDM for 4 h through Western blot (G, J). NLRP3, CASPASE-1-p20, and GSDMD-N expressions were detected in AREG +ATP-induced TLR4−/−BMDM via Western blot (H, K). The expression of IL-1b and IL-18 was detected in the supernatant of AREG +ATP-induced TLR4−/−BMDM via ELISA (I). Data are presented as mean ± SEM (n ≥ 3). *P < 0.05, **P < 0.01, ***P < 0.001 vs. Control. ns, no significant; AREG, amphiregulin; BMDM, bone marrow-derived macrophages; EGFR, epidermal growth factor receptor; GSDMD, gasdermin D; ATP, adenosine triphosphate. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40292295), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Amphiregulin by Western Blot
Neutralizing extracellular AREG decreases LPS-induced TLR4 expression and pyroptosis in macrophages. LPS-induced BMDM was pretreated with a neutralizing antibody of AREG. p-EGFR, TLR4, and GSDMD-N expression levels were detected via Western blot and immunofluorescence (A-C, E, F), TLR4 expression and ASC oligomerization was detected through immunofluorescence (D, G, H). Formation of pyrosomes (red arrows) was detected using electron microscopy, scale bars, 2 μm (I). Data are presented as mean ± SEM (n ≥ 3).*P < 0.05, **P < 0.01, ***P < 0.001 vs. Control. AREG, amphiregulin. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40292295), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse Amphiregulin Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed frozen sections of mouse skin
Sample: Immersion fixed frozen sections of mouse skin
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse Amphiregulin (Catalog # 989-AR)
Sample: Recombinant Mouse Amphiregulin (Catalog # 989-AR)
Neutralization
Measured by its ability to neutralize Amphiregulin-induced proliferation in the Balb/3T3 mouse embryonic fibroblast cell line. The Neutralization Dose (ND50) is typically 0.3-1.5 µg/mL in the presence of 50 ng/mL Recombinant Mouse Amphiregulin.
Mouse Amphiregulin Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Amphiregulin
References
- Thompson, S.A. et al. (1996) J. Biol. Chem. 271:17927.
- Sonoda, H. et al. (1992) Biochem. Biophys. Res. Commun. 185:103.
- Normanno, N. et al. (2001) Frontiers in Bioscience 6:685.
Alternate Names
AREG
Gene Symbol
AREG
UniProt
Additional Amphiregulin Products
Product Documents for Mouse Amphiregulin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Amphiregulin Antibody
For research use only
Related Research Areas
Citations for Mouse Amphiregulin Antibody
Customer Reviews for Mouse Amphiregulin Antibody
There are currently no reviews for this product. Be the first to review Mouse Amphiregulin Antibody and earn rewards!
Have you used Mouse Amphiregulin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways