Cathepsin D is a lysosomal aspartic protease of the pepsin family (4). Mouse Cathepsin D is synthesized as a precursor protein, consisting of a signal peptide (residues 1‑20), a propeptide (residues 21‑64), and a mature chain (residues 65‑410) (1‑3). It is expressed in most cells and overexpressed in breast cancer cells (5). It is a major enzyme in protein degradation in lysosomes, and also involved in the presentation of antigenic peptides. Mice deficient in this enzyme showed a progressive atrophy of the intestinal mucosa, a massive destruction of lymphoid organs, and a profound neuronal ceroid lipofucinosis, indicating that Cathepsin D is essential for proteolysis of proteins regulating cell growth and tissue homeostasis (6). Cathepsin D secreted from human prostate carcinoma cells is responsible for the generation of angiostatin, a potent endogeneous inhibitor of angiogenesis (6).
Mouse Cathepsin D Biotinylated Antibody
R&D Systems | Catalog # BAF1029

Key Product Details
Species Reactivity
Mouse
Applications
Immunohistochemistry, Western Blot
Label
Biotin
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse Cathepsin D
Ile21-Leu410
Accession # Q3UCD9
Ile21-Leu410
Accession # Q3UCD9
Specificity
Detects mouse Cathepsin D in Western blots. In Western blots, less than 1% cross-reactivity with recombinant mouse (rm) Cathepsin A, rmCathepsin B, rmCathepsin C, rmCathepsin H, and rmCathepsin X/Z/P is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Mouse Cathepsin D Biotinylated Antibody
Detection of Mouse Cathepsin D by Western Blot
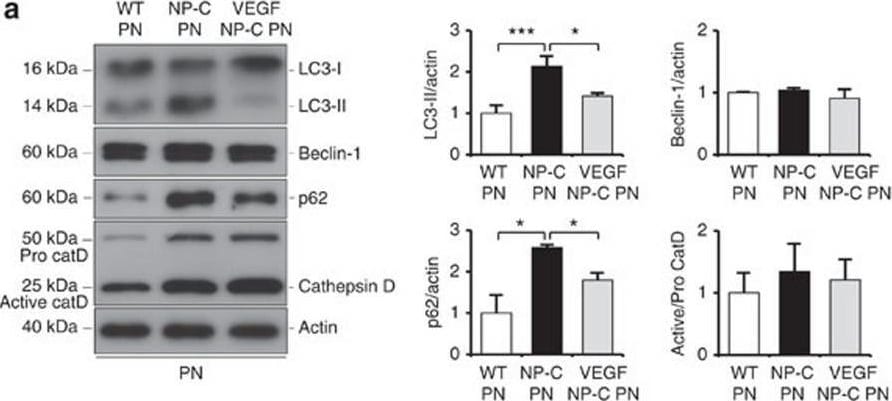
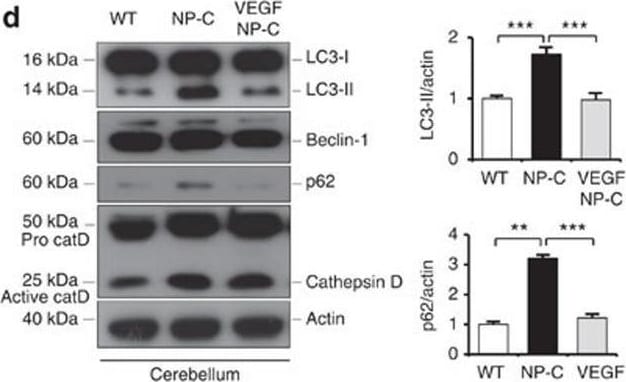
VEGF replenishment reverses defective autophagy in NP–C mice.(a) Western blot analysis of LC3, beclin-1, p62 and cathepsin D in primary cultured PNs derived from WT, NP–C and VEGF/NP–C mice (WT, n=5; NP–C, n=6; and VEGF/NP–C, n=6). (b) Immunocytochemistry of LC3 in WT, NP–C and VEGF/NP–C PNs (n=6 per group; scale bar, 20 μm). (c) Cathepsin D activity in primary cultured PNs (WT, n=5; NP–C, n=6; and VEGF/NP–C, n=6). (d) Western blot analysis of LC3, beclin-1, p62 and cathepsin D in the cerebellums of 6-week-old WT, NP–C and VEGF/NP–C mice (WT, n=6; NP–C, n=7; and VEGF/NP–C, n=7). (e) Cathepsin D activity in the cerebellums of WT, NP–C and VEGF/NP–C mice (WT, n=5; NP–C, n=6; and VEGF/NP–C, n=6). (f) EM images and quantification data of the cerebellum (n=5 per group; low-magnification scale bar, 1 μm; high-magnification scale bar, 200 nm). Arrow indicates autophagic vacuole. (g) Western blot analysis of Rab5 and Rab7 levels in the cerebellum (n=6 per group). (h) Cerebellar sections were immunostained with anti-active caspase-3 and the number of active caspase-3-positive cells in PCL was quantified (n=5 per group; scale bar, 50 μm). a–g, one-way analysis of variance, Tukey’s post hoc test. h, Student’s t-test. *P<0.05, **P<0.01, ***P<0.005. All error bars indicate s.e.m. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25417698), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Cathepsin D by Western Blot
VEGF replenishment reverses defective autophagy in NP–C mice.(a) Western blot analysis of LC3, beclin-1, p62 and cathepsin D in primary cultured PNs derived from WT, NP–C and VEGF/NP–C mice (WT, n=5; NP–C, n=6; and VEGF/NP–C, n=6). (b) Immunocytochemistry of LC3 in WT, NP–C and VEGF/NP–C PNs (n=6 per group; scale bar, 20 μm). (c) Cathepsin D activity in primary cultured PNs (WT, n=5; NP–C, n=6; and VEGF/NP–C, n=6). (d) Western blot analysis of LC3, beclin-1, p62 and cathepsin D in the cerebellums of 6-week-old WT, NP–C and VEGF/NP–C mice (WT, n=6; NP–C, n=7; and VEGF/NP–C, n=7). (e) Cathepsin D activity in the cerebellums of WT, NP–C and VEGF/NP–C mice (WT, n=5; NP–C, n=6; and VEGF/NP–C, n=6). (f) EM images and quantification data of the cerebellum (n=5 per group; low-magnification scale bar, 1 μm; high-magnification scale bar, 200 nm). Arrow indicates autophagic vacuole. (g) Western blot analysis of Rab5 and Rab7 levels in the cerebellum (n=6 per group). (h) Cerebellar sections were immunostained with anti-active caspase-3 and the number of active caspase-3-positive cells in PCL was quantified (n=5 per group; scale bar, 50 μm). a–g, one-way analysis of variance, Tukey’s post hoc test. h, Student’s t-test. *P<0.05, **P<0.01, ***P<0.005. All error bars indicate s.e.m. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25417698), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse Cathepsin D Biotinylated Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Perfusion fixed frozen sections of mouse kidney, liver, and thymus
Sample: Perfusion fixed frozen sections of mouse kidney, liver, and thymus
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse Cathepsin D (Catalog # 1029-AS)
Sample: Recombinant Mouse Cathepsin D (Catalog # 1029-AS)
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein.
Shipping
The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Cathepsin D
References
- Diedrich, et al. (1990) Nucl. Acid Res. 18:7184.
- Grusby, et al. (1990) Nucl. Acid Res. 18:4008.
- Hetman, et al. (1994) DNA Cell Biol. 13:419.
- Conner (2004) in Handbook of Proteolytic Enzymes (Barrett, et al. eds) Elsevier Academic Press, San Diego, p. 43.
- Rochefort, et al. (2000) Clin. Chim. Acta. 291:157.
- Tsukuba, et al. (2000) Mol. Cells 10:601.
Alternate Names
CTSD
Gene Symbol
CTSD
UniProt
Additional Cathepsin D Products
Product Documents for Mouse Cathepsin D Biotinylated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Cathepsin D Biotinylated Antibody
For research use only
Citations for Mouse Cathepsin D Biotinylated Antibody
Customer Reviews for Mouse Cathepsin D Biotinylated Antibody
There are currently no reviews for this product. Be the first to review Mouse Cathepsin D Biotinylated Antibody and earn rewards!
Have you used Mouse Cathepsin D Biotinylated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways