Mouse Chemerin, also known as Tazarotene-induced Gene-2 (TIG2), is a new, but distant member of the cystatin superfamily (1-3). Members of this superfamily contain at least two intrachain disulfide bonds and an alpha -helical structure over a distance of about 100 amino acids (2, 3). Chemerin is synthesized as a 162 amino acid (aa) precursor that contains a hydrophobic N-terminal sequence, an intervening 140 aa cystatin-fold containing domain, and a six aa C-terminal prosegment (4-6). Within the cystatin-fold domain there are three intrachain disulfide bonds that contribute to the characteristic fold (4, 7). The precursor molecule is described as undergoing proteolytic processing at both termini by unknown proteases. The N-terminal 16 residue hydrophobic segment is described as being either a signal sequence or a transmembrane (TM) segment for a type II TM protein (5, 8). In either case it gives rise to a soluble proform that undergoes further processing at the C‑terminus (5). In mouse, the C-terminal six residues are cleaved, giving rise to a monomeric, 16 kDa heparin-binding bioactive molecule (aa 17-156) (5-7). A shorter form has been described in human (7). The activity seems to be concentrated in the nine aa’s preceding the prosegment (aa 148-156). Retention of the prosegment blocks activity (4). The 140 aa mature segment is known to bind to the G-protein coupled receptor termed ChemR23 (5, 7). Binding results in macrophage and immature dendritic cell chemotaxis (5). The distribution of this receptor is limited to immune APCs, and it is assumed that Chemerin is an inflammatory molecule. It is unclear which cells are actually producing Chemerin, but keratinocytes, endothelial cells and osteoclasts are potential candidates (1, 7). Mature mouse Chemerin shares 67%, 84%, and 82% aa sequence identity to human, rat, and hamster Chemerin, respectively (6). There is apparently cross-species activity for the protein (6).

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Mouse, Fish - Danio rerio (Zebrafish), Transgenic Mouse
Applications
Validated:
Western Blot, Neutralization
Cited:
Immunohistochemistry-Paraffin, Western Blot, Neutralization, Immunocytochemistry, ELISA Development (Capture)
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse Chemerin
Thr17-Ser156
Accession # Q9DD06
Thr17-Ser156
Accession # Q9DD06
Specificity
Detects mouse Chemerin in direct ELISAs and Western blots. In direct ELISAs and Western blots, approximately 20% cross-reactivity with recombinant human Chemerin is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse Chemerin Antibody
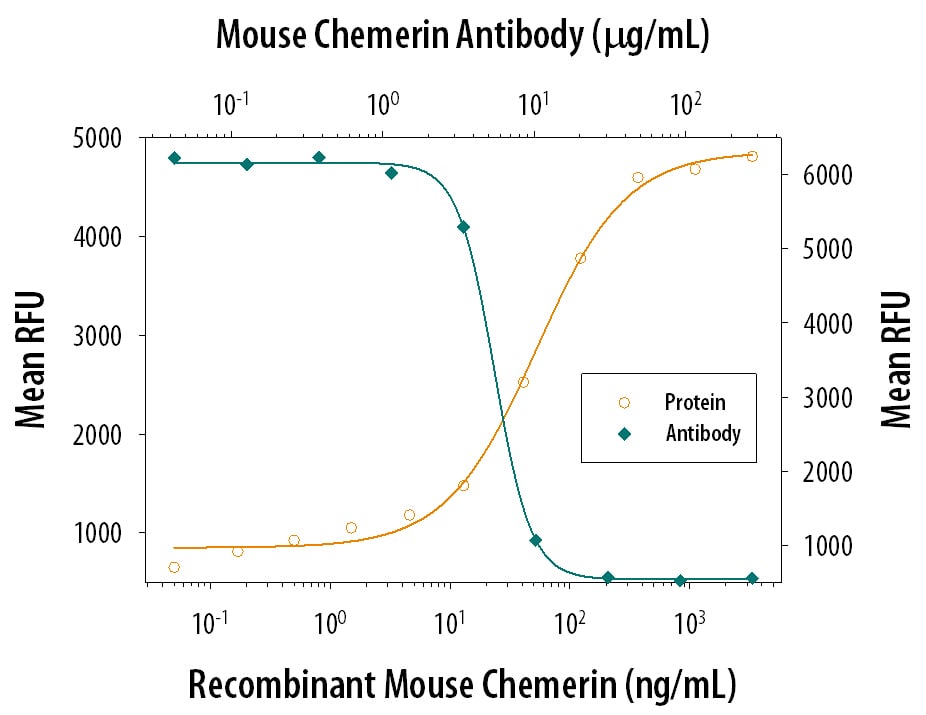
Chemotaxis Induced by Chemerin and Neutralization by Mouse Chemerin Antibody.
Recombinant Mouse Chemerin (2325-CM) chemo-attracts the BaF3 mouse pro-B cell line transfected with human Chem R23 in a dose-dependent manner (orange line). The amount of cells that migrated through to the lower chemotaxis chamber was measured by Resazurin (AR002). Chemotaxis elicited by Recombinant Mouse Chemerin (100 ng/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Mouse Chemerin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2325). The ND50 is typically 3-9 µg/mL.Applications for Mouse Chemerin Antibody
Application
Recommended Usage
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse Chemerin (Catalog # 2325-CM)
Sample: Recombinant Mouse Chemerin (Catalog # 2325-CM)
Neutralization
Measured by its ability to neutralize Chemerin-induced chemotaxis in the BaF3 mouse pro‑B cell line transfected with human Chem R23. The Neutralization Dose (ND50) is typically 3-9 µg/mL in the presence of 100 ng/mL Recombinant Mouse Chemerin.
Reviewed Applications
Read 1 review rated 4 using AF2325 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Chemerin
References
- Nagpal, S. et al. (1997) J. Invest. Dermatol. 109:91.
- Storici, P. et al. (1996) Eur. J. Biochem. 238:769.
- Zanetti, M. (2004) J. Leukoc. Biol. 75:39.
- Wittamer, V. et al. (2004) J. Biol. Chem. 279:9956.
- Wittamer, V. et al. (2003) J. Exp. Med. 198:977.
- Busmann, A. et al. (2004) J. Chromatog. B 811:217.
- Meder, W. et al. (2003) FEBS Lett. 555:495.
- Yokoyama-Kobayashi, M. et al. (1999) Gene 228:161.
Long Name
Retinoic Acid Receptor Responder Protein 2
Alternate Names
RARRES2, TIG-2
Gene Symbol
RARRES2
UniProt
Additional Chemerin Products
Product Documents for Mouse Chemerin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Chemerin Antibody
For research use only
Related Research Areas
Citations for Mouse Chemerin Antibody
Customer Reviews for Mouse Chemerin Antibody (1)
4 out of 5
1 Customer Rating
Have you used Mouse Chemerin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Western BlotSample Tested: See PMID 23766088Species: MouseVerified Customer | Posted 01/06/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
