Dentin matrix protein 1 (DMP-1) is a member of the SIBLING family of proteins that includes bone sialoprotein, dentin sialophosphoprotein, MEPE, and osteopontin. These are highly phosphorylated integrin-binding proteins that are rich in acidic amino acids and function in the formation of calcified bone and tooth matrix (1, 2). Its phosphate content, spacing of acidic residues, and calcium-dependent dimerization of DMP-1 contribute to its ability to sequester calcium phosphate clusters and promote hydroxyapatite (HA) crystal formation (3-5). Mature mouse DMP-1 is 487 amino acids (aa) in length. It contains a poly-Pro segment (aa 41-44) and an RGD binding motif (aa 350-352). DMP-1 may be cleaved by BMP-1 family proteases at a single site which is conserved in human, generating a 37 kDa N-terminal (aa 17‑212) and a 57 kDa C-terminal (aa 213-503) fragment (6). The N-terminal fragment in rat carries chondroitin sulfate (7). The C-terminal fragment alone can nucleate HA crystals, while crystal growth into a needle-like morphology is inhibited by the N-terminal fragment (3, 4). Crystal maturation is dependent on the presence of type I collagen (4). DMP-1 is required for odontoblast differentiation as well as dentin formation (8). Unphosphorylated DMP-1 is retained intracellularly where it is targeted to the nucleus. Here, it activates the transcription of odontoblast and osteoblast specific genes (9, 10). Early in osteoblast maturation, nuclear DMP-1 is extensively phosphorylated by casein kinase II, triggering its secretion (9). DMP-1 mutations in humans are associated with hypophosphatemia and FGF-23 over-expression (11, 12). DMP-1 induces the activation of pro-MMP-9 and displaces mature MMP-9 from TIMP1 (13). DMP-1 tethers MMP-9 to the cell surface via CD44 and integrins alpha V beta 3 and alpha V beta 5, promoting tumor cell invasiveness in vitro (14). Full length DMP-1 circulates in human serum in a tight complex with complement factor H (13, 14). When first bound to CD44 or integrin alpha V beta 3, DMP-1 can anchor factor H to the cell surface and protect the cell from complement-mediated lysis (15). Mature mouse DMP-1 shares 63%, 61%, and 87% aa sequence identity with bovine, human, and rat DMP-1, respectively.
Mouse DMP‑1 Antibody
R&D Systems | Catalog # AF4386

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Mouse, Rat
Applications
Validated:
Immunohistochemistry, Western Blot
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Flow Cytometry
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse DMP-1
Leu17-Tyr503
Accession # Q2HJ09
Leu17-Tyr503
Accession # Q2HJ09
Specificity
Detects mouse DMP-1 in direct ELISAs and Western blots. In direct ELISAs, approximately 25% cross-reactivity with recombinant human DMP-1 is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Mouse DMP‑1 Antibody
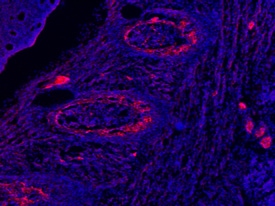
DMP‑1 in Embryonic Mouse Rib.
DMP-1 was detected in immersion fixed frozen sections of embryonic mouse rib (E15.5) using 10 µg/mL Sheep Anti-Mouse DMP-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4386) overnight at 4 °C. Tissue was stained with the NorthernLights™ 557-conjugated Anti-Sheep IgG Secondary Antibody (red; NL010) and counterstained with DAPI (blue). View our protocol for Fluorescent IHC Staining of Frozen Tissue Sections.Detection of Mouse DMP-1 by Immunohistochemistry
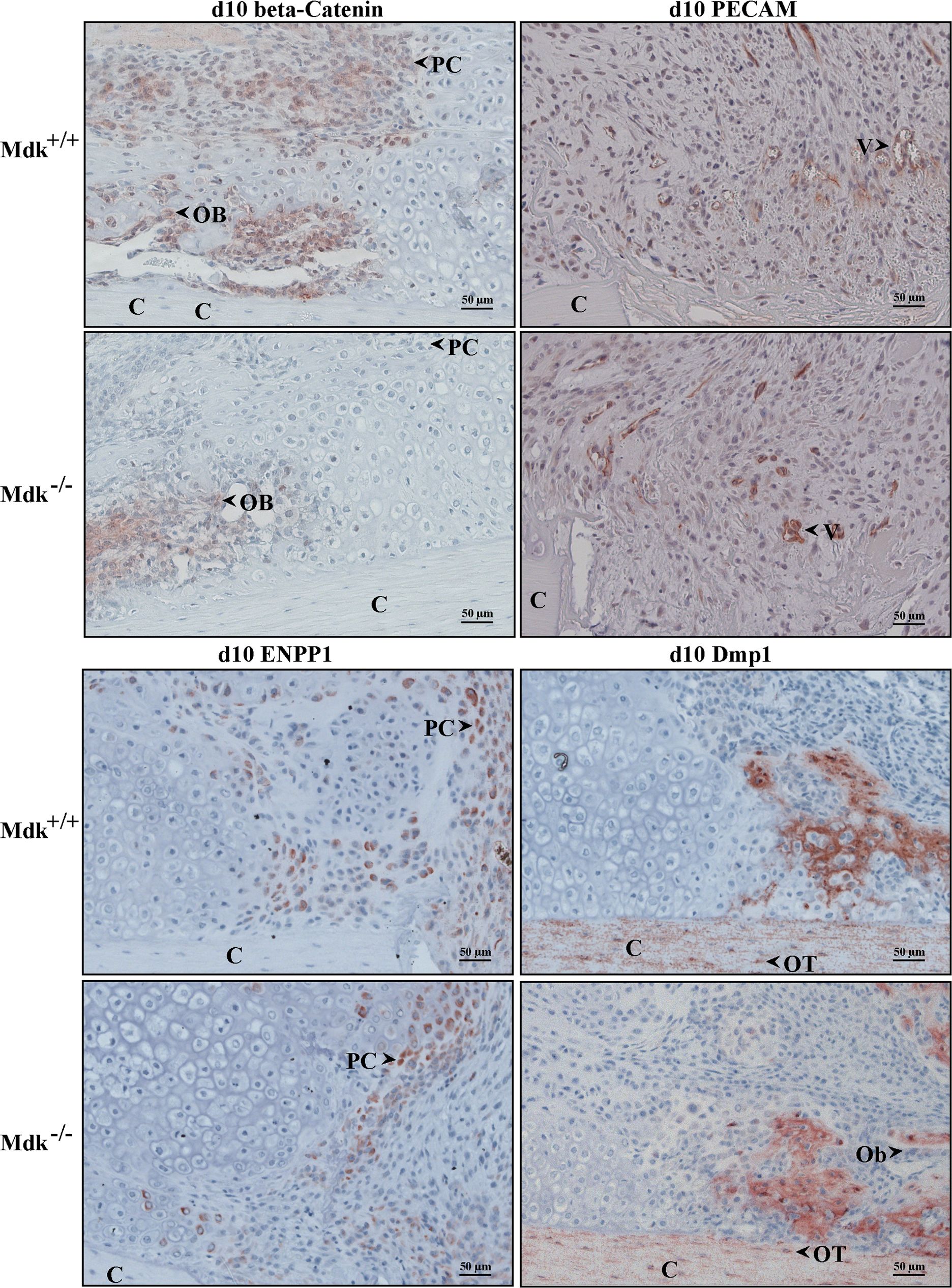
Immunohistochemical staining showing reduced beta-catenin levels in chondrocytes of Mdk-deficient mice.Sections of fractured femurs from four mice of each time point and genotype group were stained for each antigen and counterstained using hematoxylin. Representative images are shown; C = cortex; PC = proliferating chondrocyte; Ob = osteoblast; V = vessel; OT = osteocyte; scale bar 50 µm; 200-fold magnification. Beta-catenin staining of the periosteal callus at day 10. PECAM staining of the periosteal fracture callus bridging the osteotomy gap showing the endothelial cells of the newly formed vessels in an area of proliferating chondrocytes at day 10. Enpp1 staining of the osteotomy gap at day 10 showing positively stained proliferating chondrocytes. Dmp1 staining of the periosteal callus at day 10 showing positively stained cortex, osteocytes and areas of new bone formation. Osteoblasts were Dmp1 negative. (n = 4 per group). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25551381), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse DMP‑1 Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed frozen sections of embryonic mouse rib (E15.5)
Sample: Immersion fixed frozen sections of embryonic mouse rib (E15.5)
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse DMP‑1 (Catalog # 4386-DM)
Sample: Recombinant Mouse DMP‑1 (Catalog # 4386-DM)
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: DMP-1
References
- Qin, C. et al. (2004) Crit. Rev. Oral Biol. Med. 15:126.
- MacDougall, M. et al. (1998) J. Bone Miner. Res. 13:422.
- He, G. et al. (2003) Nat. Mater. 2:552.
- Gajjeraman, S. et al. (2007) J. Biol. Chem. 282:1193.
- He, G. et al. (2005) Biochemistry 44:16140.
- Steiglitz, B.M. et al. (2004) J. Biol. Chem. 279:980.
- Qin, C. et al. (2006) J. Biol. Chem. 281:8034.
- Lu, Y. et al. (2007) Dev. Biol. 303:191.
- Narayanan, K. et al. (2003) J. Biol. Chem. 278:17500.
- Narayanan, K. et al. (2006) J. Biol. Chem. 281:19064.
- Lorenz-Depiereux, B. et al. (2006) Nat. Genet. 38:1248.
- Feng, J.Q. et al. (2006) Nat. Genet. 38:1310.
- Fedarko, N.S. et al. (2004) FASEB J. 18:735.
- Karadag, A. et al. (2005) Cancer Res. 65:11545.
- Jain, A. et al. (2002) J. Biol. Chem. 277:13700.
Long Name
Dentin Matrix Protein 1
Alternate Names
DMP1
Gene Symbol
DMP1
UniProt
Additional DMP-1 Products
Product Documents for Mouse DMP‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse DMP‑1 Antibody
For research use only
Citations for Mouse DMP‑1 Antibody
Customer Reviews for Mouse DMP‑1 Antibody
There are currently no reviews for this product. Be the first to review Mouse DMP‑1 Antibody and earn rewards!
Have you used Mouse DMP‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...