Endocan (endothelial cell proteoglycan; also known as endothelial-cell specific molecule-1 [ESM-1]), is a 50 kDa, monomeric, secreted, cysteine-rich proteoglycan identified initially in endothelial cells of the kidney and lung (1). Mouse Endocan is synthesized as a 184 amino acid (aa) precursor that contains a 21 aa signal sequence and a 20 kDa, 163 aa mature region (2). The N-terminal 2/3 of the molecule contains 18 cysteine residues and there are no potential N-linked glycosylation sites. Based on human Endocan, there are at least two potential O-linked glycosylation sites, one of which will likely be utilized on Ser at position # 136 of the mature molecule (3). The posttranslational modification is approximately 30 kDa in size. It consists of a single dermatan sulfate chain that contains 4-O sulfated N-acetyl galactosamine with alpha -iduronate. This chain is suggested to bind HGF and contribute to HGF mitogenic activity (4). Mature mouse Endocan is 96% and 74% aa identical to rat and human Endocan, respectively. In human, there is a potential for an alternate splice variant. It shows a deletion of aa’s 82 through 131, a range which would not remove the dermatan sulfate attachment site (4). It is not known if such a splice form exists in mouse. Endocan is expressed by endothelial cells, adipocytes, bronchial epithelium and distal renal tubular epithelium (1, 5, 6). It is upregulated by TNF-alpha and VEGF, (1, 7) and is known to bind to LFA-1 ( alpha L beta 2) on the surface of PBMCs, blocking LFA-1 interaction with ICAM-1 (8). Normal circulating levels of Endocan are approximately 1 ng/mL (6).
Mouse Endocan/ESM‑1 Antibody
R&D Systems | Catalog # AF1999

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Mouse, Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Neutralization
Cited:
Immunohistochemistry, Immunohistochemistry-Frozen, Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse Endocan/ESM-1
Trp20-Arg184
Accession # Q9QYY7
Trp20-Arg184
Accession # Q9QYY7
Specificity
Detects mouse Endocan/ESM-1 in direct ELISAs and Western blots. In direct ELISAs and Western blots, approximately 25% cross-reactivity with recombinant human Endocan is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse Endocan/ESM‑1 Antibody
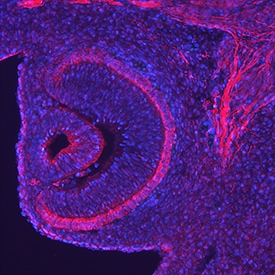
Endocan/ESM‑1 in Mouse Embryo.
Endocan/ESM-1 was detected in perfusion fixed frozen sections of mouse embryo (13 d.p.c.) using Goat Anti-Mouse Endocan/ESM-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1999) at 5 µg/mL overnight at 4 °C. Tissue was stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to the retina. View our protocol for Fluorescent IHC Staining of Frozen Tissue Sections.Cell Adhesion Mediated by Endocan/ESM‑1 and Neutralization by Mouse Endocan/ESM‑1 Antibody.
Recombinant Mouse Endocan/ESM-1 (Catalog # 1999-EC), immobilized onto a microplate, supports the adhesion of the Jurkat human acute T cell leukemia cell line in a dose-dependent manner (orange line). Adhesion elicited by Recombinant Mouse Endocan/ESM-1 (25 µg/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Mouse Endocan/ESM-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1999). The ND50 is typically 1-5 µg/mL.Detection of Mouse Endocan/ESM-1 by Immunocytochemistry/Immunofluorescence
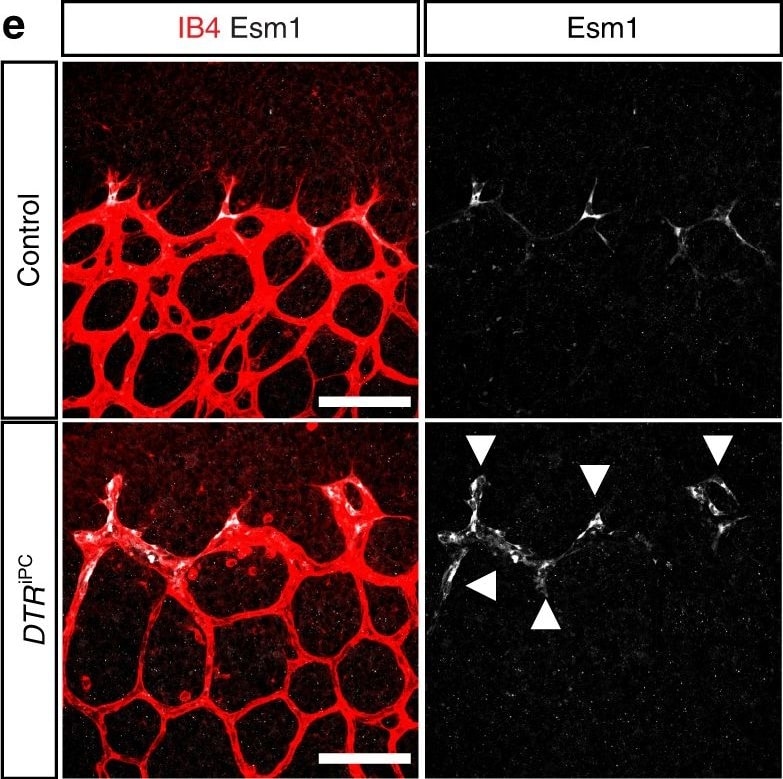
Endothelial changes after pericyte depletion. a–f Maximum intensity projection of confocal images from control and DTRiPC P6 retinas stained for IB4 (red) in combination with VEGF-A a, VEGFR2 b, VEGFR3 c, Tie2 d, Esm1 e, and Dll4 f (all in white), as indicated. Note local increase of VEGFR2, VEGFR3, and Esm1 (arrowheads in b, c, e) but not Tie2 or VEGF-A at the edge of the vessel plexus. Dll4 expression in DTRiPC sprouts is increased in some regions (arrowheads) but absent in others (arrows). Scale bar, 100 µm. g–j Quantitation of VEGF-A immunosignals area and intensity g, signal intensity for VEGFR2 h and VEGFR3 i and proportion of Esm1+ area with respect to vascular area j in the P6 control and DTRiPC angiogenic front. Error bars, s.e.m. p-values, Student’s t-test Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29146905), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Endocan/ESM-1 by Immunocytochemistry/Immunofluorescence
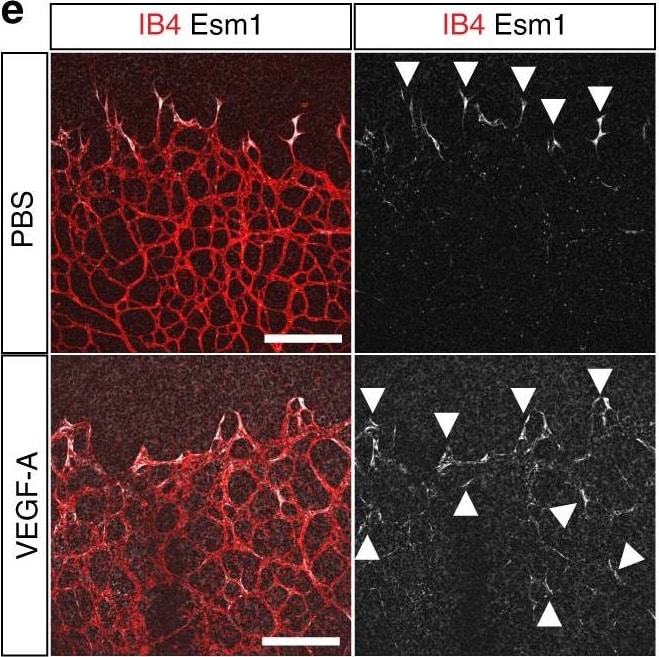
Vascular alterations after intraocular VEGF-A injection. a Morphology of IB4-stained P6 wild-type retinal vessels at 4 h after administration of human VEGF-A165 (0.5 µl at a concentration of 5 μg μl−1). Note blunt appearance of the vessel front after VEGF-A injection but not for vehicle (PBS) control. Scale bar, 200 µm. b Quantitation of sprouts and filopodia at the front of the P6 vessel plexus after injection of VEGF-A165 or vehicle control. Error bars, s.e.m. p-values, Student’s t-test. c PDGFR beta + (green) pericytes are unaffected by short-term VEGF-A administration, whereas VEGFR2 immunosignals (white) are increased in IB4+ (red) ECs (arrowheads). Images shown correspond to insets in a. Scale bar, 100 µm. d Quantitation of VEGFR2 immunosignals intensity in the peripheral plexus of P6 retinas after injection of VEGF-A165 or vehicle control. Error bars, s.e.m. p-values, Student’s t-test. e Confocal images showing increased Esm1 immunostaining (white) in IB4+ (red) ECs in the peripheral plexus (arrowheads) after VEGF-A injection in P6 pups. Scale bar, 200 µm. f VEGF-A165 injection-mediated increase of Esm1 immunosignals (normalized to IB4+ EC area) in the peripheral capillary plexus but not at the edge of the angiogenic front in comparison to PBS-injected controls at P6. Error bars, s.e.m. p-values, Student’s t-test. NS, not statistically significant. g Short-term VEGF-A165 administration leads to clustering of Erg1+ (green) and IB4+ (red) ECs, as indicated, in thick sprout-like structures of P6 retinas. Panels in the center and on the right (scale bar, 20 µm) show higher magnification of the insets on the left (scale bar, 100 µm). Dashed lines in panels on the right outline IB4+ vessels. h Quantitation of EC density in the leading front vessel and emerging sprouts of the P6 angiogenic front after injection of VEGF-A165 or vehicle control. Error bars, s.e.m. p-values, Student’s t-test Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29146905), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Endocan/ESM-1 by Immunocytochemistry/Immunofluorescence
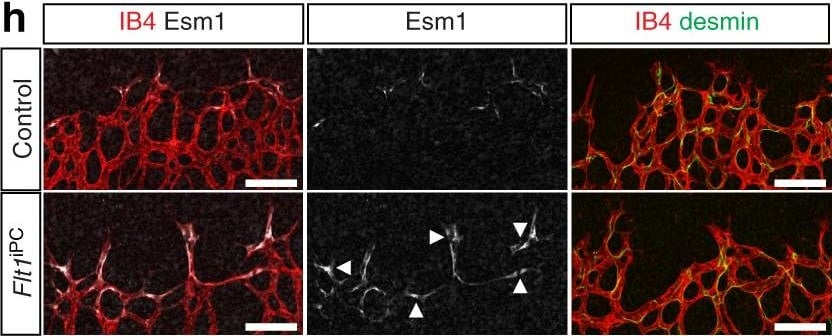
Inactivation of Flt1 in PDGFR beta + cells. a Experimental scheme of tamoxifen administration for the generation of Flt1iPC mutants. b P6 control, Flt1iPC/+ and Flt1iPC retinas stained with isolectin B4 (IB4). Dashed circles indicate vessel-covered (yellow) and peripheral avascular (white) areas in the overview pictures (top). Scale bar, 500 µm. c Quantitation of body weight and radial outgrowth of the retinal vasculature in control, Flt1iPC/+ and Flt1iPC P6 pups. Error bars, s.e.m. p-values, one-way ANOVA. NS, not statistically significant. d Confocal images of the IB4-stained P6 control, Flt1iPC/+ and Flt1iPC retinal angiogenic front illustrating differences in sprout number and morphology. Scale bar, 100 µm. e Quantitation of sprouts and filopodia in P6 control, Flt1iPC/+ and Flt1iPC retinas. Error bars, s.e.m. p-values, one-way ANOVA and Tukey’s multiple comparison test. NS, not statistically significant. f Confocal images of IB4 (red), Erg1 (green) and VEGFR2 (white) stained P6 retinas highlighting the accumulation of EC nuclei and enhanced VEGFR2 immunosignals (arrowheads) in Flt1iPC sprouts. Vessels are outlined by dashed lines on the right panel. Scale bar, 100 µm. g Quantitation of EC proliferation (EdU+ Erg1+) at the angiogenic front, EC density in sprouts and leading front vessel and VEGFR2 immunosignals intensity in the angiogenic front of control and Flt1iPC P6 retinas. Error bars, s.e.m. p-values, Student’s t-test. h Esm1 (white) expression (arrowheads) in the angiogenic front (IB4+, red, first two columns) and detection of desmin+ pericytes (green, third column) in P6 control and Flt1iPC retinas. Scale bar, 100 µm. i Quantitation of Esm1+ proportion relative to vascular area (IB4+) in the angiogenic front of control and Flt1iPC P6 retinas. Error bars, s.e.m. p-values, Student’s t-test. j Confocal images of P6 retinas stained for NG2 (green) and IB4 (red) showing no significant changes in pericyte coverage in the front (first two columns) or the remodeling plexus around veins (v) or arteries (a) (last two columns). Scale bar, 100 µm. k, l Quantitation of pericyte coverage k and relative gene expression by qPCR on whole lysates l in control and Flt1iPC P6 retinas. Error bars, s.e.m. p-values, Student’s t-test. NS, not statistically significant Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29146905), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse Endocan/ESM‑1 Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample:
Sample:
Perfusion fixed frozen sections of mouse embryo (13 d.p.c.)
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse Endocan/ESM‑1 (Catalog # 1999-EC)
Sample: Recombinant Mouse Endocan/ESM‑1 (Catalog # 1999-EC)
Neutralization
Measured by its ability to neutralize Endocan/ESM‑1-mediated adhesion of the Jurkat human acute T cell leukemia cell line. The Neutralization Dose (ND50) is typically 1-5 µg/mL in the presence of 25 µg/mL Recombinant Mouse Endocan/ESM‑1.
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Endocan/ESM-1
References
- Lassalle, P. et al. (1996) J. Biol. Chem. 271:20458.
- Lassalle, P. (1999) Genbank Accession #: Q9QYY7.
- Bechard, D. et al. (2001) J. Biol. Chem. 276:48341.
- Aitkenhead, M. et al. (2002) Microvasc. Res. 63:159.
- Wellner, M. et al. (2003) Horm. Metab. Res. 35:217
- Bechard, D. et al. (2000) J. Vasc. Res. 37:417.
- Tsai, J.C. et al. (2002) J. Vasc. Res. 39:148.
Alternate Names
ESM-1, ESM1, IGFBP-rp6
Gene Symbol
ESM1
UniProt
Additional Endocan/ESM-1 Products
Product Documents for Mouse Endocan/ESM‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Endocan/ESM‑1 Antibody
For research use only
Related Research Areas
Citations for Mouse Endocan/ESM‑1 Antibody
Customer Reviews for Mouse Endocan/ESM‑1 Antibody
There are currently no reviews for this product. Be the first to review Mouse Endocan/ESM‑1 Antibody and earn rewards!
Have you used Mouse Endocan/ESM‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...