Fibroblast growth factors (FGFs) comprise a family of at least eighteen structurally related proteins that are involved in a multitude of physiological and pathological cellular processes, including cell growth, differentiation, angiogenesis, wound healing and tumorgenesis. The biological activities of the FGFs are mediated by a family of type I transmembrane tyrosine kinases which undergo dimerization and autophosphorylation after ligand binding. Four distinct genes encoding closely related FGF receptors, FGF R1‑4, are known. All four genes for FGF Rs encode proteins with an N‑terminal signal peptide, three immunoglobulin (Ig)‑like domains, an acid‑box region containing a run of acidic residues between the IgI and IgII domains, a transmembrane domain and the split tyrosine‑kinase domain. Multiple forms of FGF R1‑3 are generated by alternative splicing of the mRNAs. A frequent splicing event involving FGF R1 and 2 results in receptors containing all three Ig domains, referred to as the alpha isoform, or only IgII and IgIII, referred to as the beta isoform. Only the alpha isoform has been identified for FGF R3 and FGF R4. Additional splicing events for FGF R1‑3, involving the C‑terminal half of the IgIII domain encoded by two mutually exclusive alternative exons, generate FGF receptors with alternative IgIII domains (IIIb and IIIc). A IIIa isoform which is a secreted FGF binding protein containing only the N‑terminal half of the IgIII domain plus some intron sequences has also been reported for FGF R1. Mutations in FGF R1‑3 have been found in patients with birth defects involving craniosynostosis. The complex patterns of expression of these receptors as well as the specificity of their interactions with the various FGF ligand family members are being investigated.

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Mouse
Applications
Validated:
Neutralization
Cited:
Immunohistochemistry-Frozen, Neutralization
Label
Unconjugated
Antibody Source
Monoclonal Rat IgG2A Clone # 136812
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse FGF R3
Glu21-Tyr369 (Asn310Ile, Ala354 del) (predicted)
Accession # NP_001156689
Glu21-Tyr369 (Asn310Ile, Ala354 del) (predicted)
Accession # NP_001156689
Specificity
Detects mouse FGF R3 in direct ELISAs. In direct ELISAs, 100% cross-reactivity with the (IIIb) and (IIIc) isoforms of recombinant human FGF R3 is observed. Neutralizes the bioactivity of mouse FGF R3. Functions equally as well in the neutralization of recombinant mouse (rm) FGF R3 alpha (IIIb) and rmFGF R3 alpha (IIIc).
Clonality
Monoclonal
Host
Rat
Isotype
IgG2A
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse FGFR3 Antibody
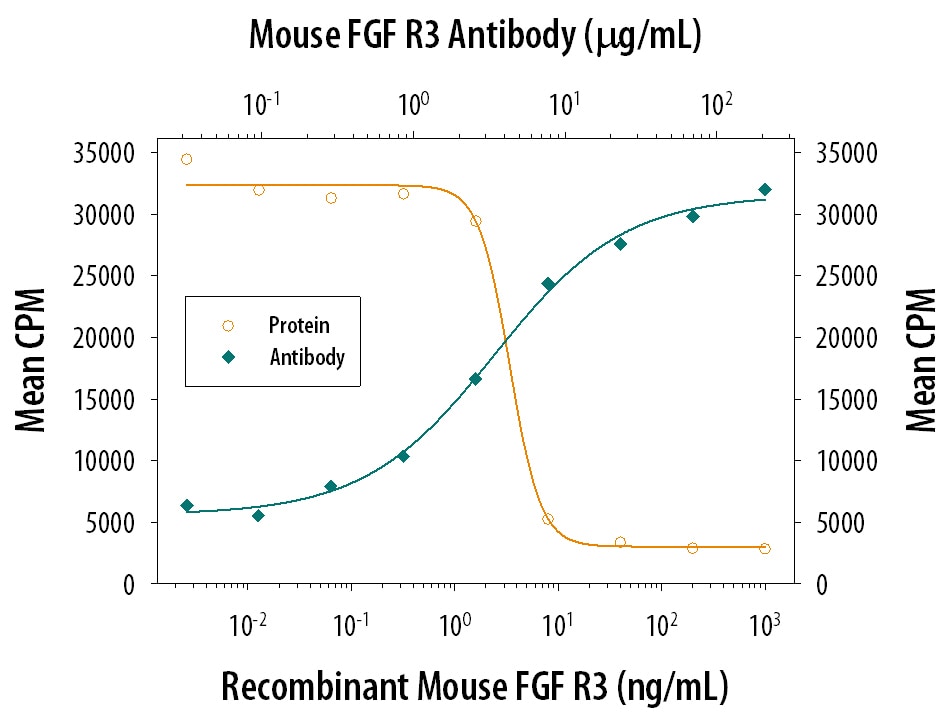
FGF R3 Inhibition of FGF acidic-dependent Cell Proliferation and Neutral-ization by Mouse FGF R3 Antibody.
Recombinant Mouse FGF R3 (IIIc) Fc Chimera (Catalog # 710-MF) inhibits Recom-binant Human FGF acidic (Catalog # 232-FA) induced proliferation in the NR6R-3T3 mouse fibroblast cell line in a dose-dependent manner (orange line). Inhibition of Recom-binant Human FGF acidic (0.3 ng/mL) activity elicited by Recom-binant Mouse FGF R3 (IIIc) Fc Chimera (12 ng/mL) is neutralized (green line) by increasing concentrations of Rat Anti-Mouse FGF R3 Mono-clonal Antibody (Catalog # MAB710). The ND50 is typically 3-12 µg/mL in the presence of heparin (10 µg/mL).Applications for Mouse FGFR3 Antibody
Application
Recommended Usage
Neutralization
Measured by its ability to neutralize FGF R3-mediated inhibition of proliferation in the NR6R‑3T3 mouse fibroblast cell line. The Neutralization Dose (ND50) is typically 3-12 µg/mL in the presence of 12 ng/mL Recombinant Mouse FGF R3 (IIIc) Fc Chimera, 0.3 ng/mL Recombinant Human FGF acidic, and 10 µg/mL heparin.
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: FGFR3
References
- Galzie, Z. et al. (1997) Biochem. Cell Biol. 75:669.
- Burke, D. et al. (1998) Trends Biochem. Sci. 23:59.
Long Name
Fibroblast Growth Factor Receptor 3
Alternate Names
CD333, CEK, FGF R3, JTK4
Gene Symbol
FGFR3
UniProt
Additional FGFR3 Products
Product Documents for Mouse FGFR3 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse FGFR3 Antibody
For research use only
Related Research Areas
Citations for Mouse FGFR3 Antibody
Customer Reviews for Mouse FGFR3 Antibody
There are currently no reviews for this product. Be the first to review Mouse FGFR3 Antibody and earn rewards!
Have you used Mouse FGFR3 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review




