Key Product Details
Assay Type
Assay Range
Sample Type
Note: Diluents for complex matrices, such as serum and plasma, should be evaluated prior to use in this DuoSet
Reactivity
Mouse G-CSF DuoSet ELISA Features
- Optimized capture and detection antibody pairings with recommended concentrations save lengthy development time
- Development protocols are provided to guide further assay optimization
- Assay can be customized to your specific needs
- Economical alternative to complete kits
Product Summary for Mouse G-CSF DuoSet ELISA
This DuoSet ELISA Development kit contains the basic components required for the development of sandwich ELISAs to measure natural and recombinant mouse G-CSF. The suggested diluent is suitable for the analysis of most cell culture supernate samples. Diluents for complex matrices, such as serum and plasma, should be evaluated prior to use in this DuoSet.
Product Specifications
Assay Format
Sample Volume Required
Detection Method
Conjugate
Specificity
Label
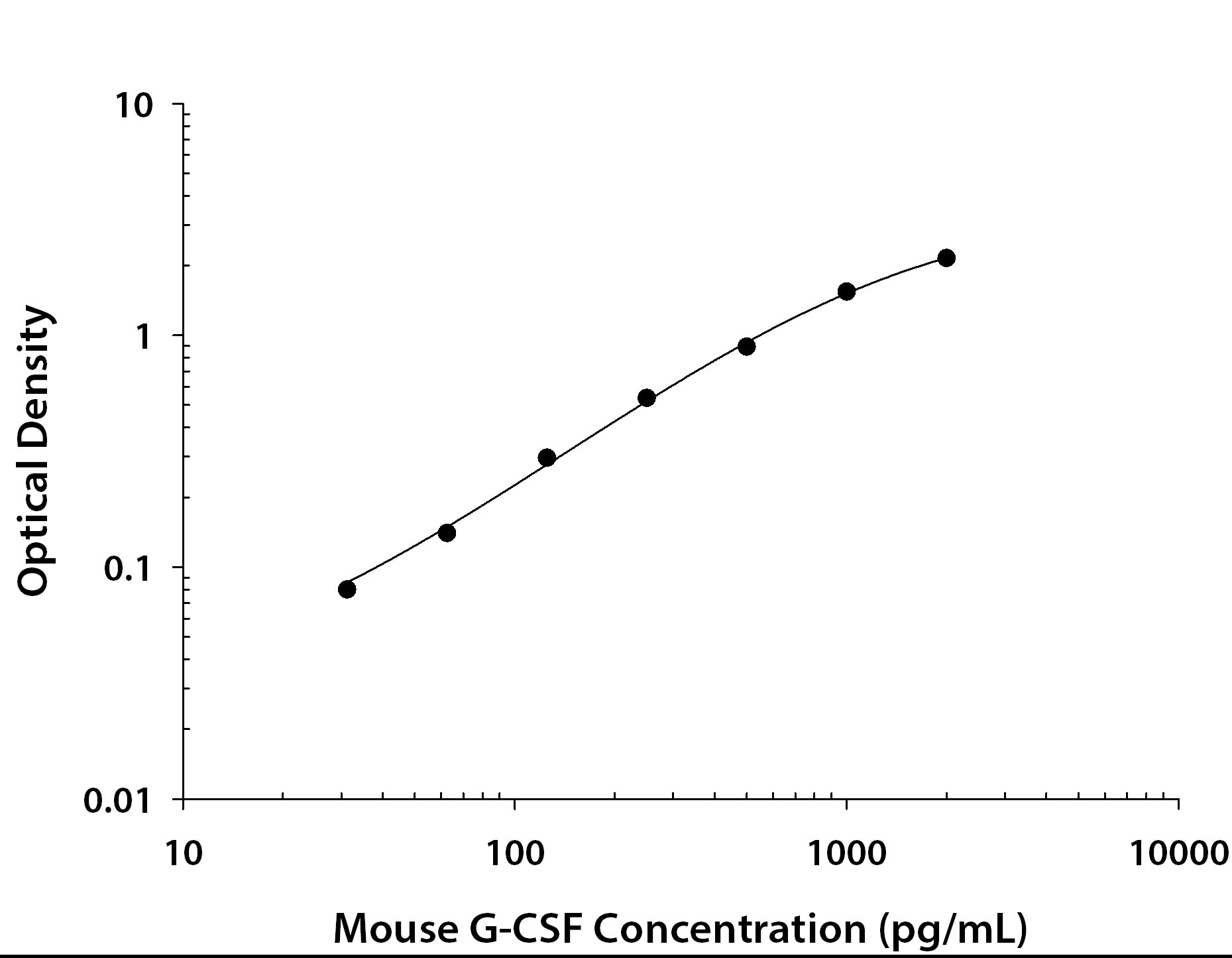
Scientific Data Images for Mouse G-CSF DuoSet ELISA
Mouse G-CSF ELISA Standard Curve
Kit Contents for Mouse G-CSF DuoSet ELISA
- Capture Antibody
- Detection Antibody
- Recombinant Standard
- Streptavidin conjugated to horseradish-peroxidase (Streptavidin-HRP)
Other Reagents Required
DuoSet Ancillary Reagent Kit 2 (5 plates): (Catalog # DY008C) containing 96 well microplates, plate sealers, substrate solution, stop solution, plate coating buffer (PBS), wash buffer, and Reagent Diluent Concentrate 2.
PBS: (Catalog # DY006), or 137 mM NaCl, 2.7 mM KCl, 8.1 mM Na2HPO4, 1.5 mM KH2PO4, pH 7.2 - 7.4, 0.2 µm filtered
Wash Buffer: (Catalog # WA126), or equivalent
Reagent Diluent*
Blocking Buffer*
Substrate Solution: ELISA TMB Substrate (Catalog # DY999B or DY999B-250)
Stop Solution: Methanesulfonic acid (Catalog # DY994B or DY994B-250)
Microplates: (Catalog # DY990), or equivalent
Plate Sealers: (Catalog # DY992), or equivalent
*For the recommended Reagent Diluent and Blocking Buffer for a specific DuoSet ELISA Development Kit, refer to the product datasheet.
Preparation and Storage
Shipping
Stability & Storage
Background: G-CSF
Long Name
Alternate Names
Gene Symbol
Additional G-CSF Products
Product Documents for Mouse G-CSF DuoSet ELISA
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse G-CSF DuoSet ELISA
For research use only
Citations for Mouse G-CSF DuoSet ELISA
Customer Reviews for Mouse G-CSF DuoSet ELISA (4)
Have you used Mouse G-CSF DuoSet ELISA?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
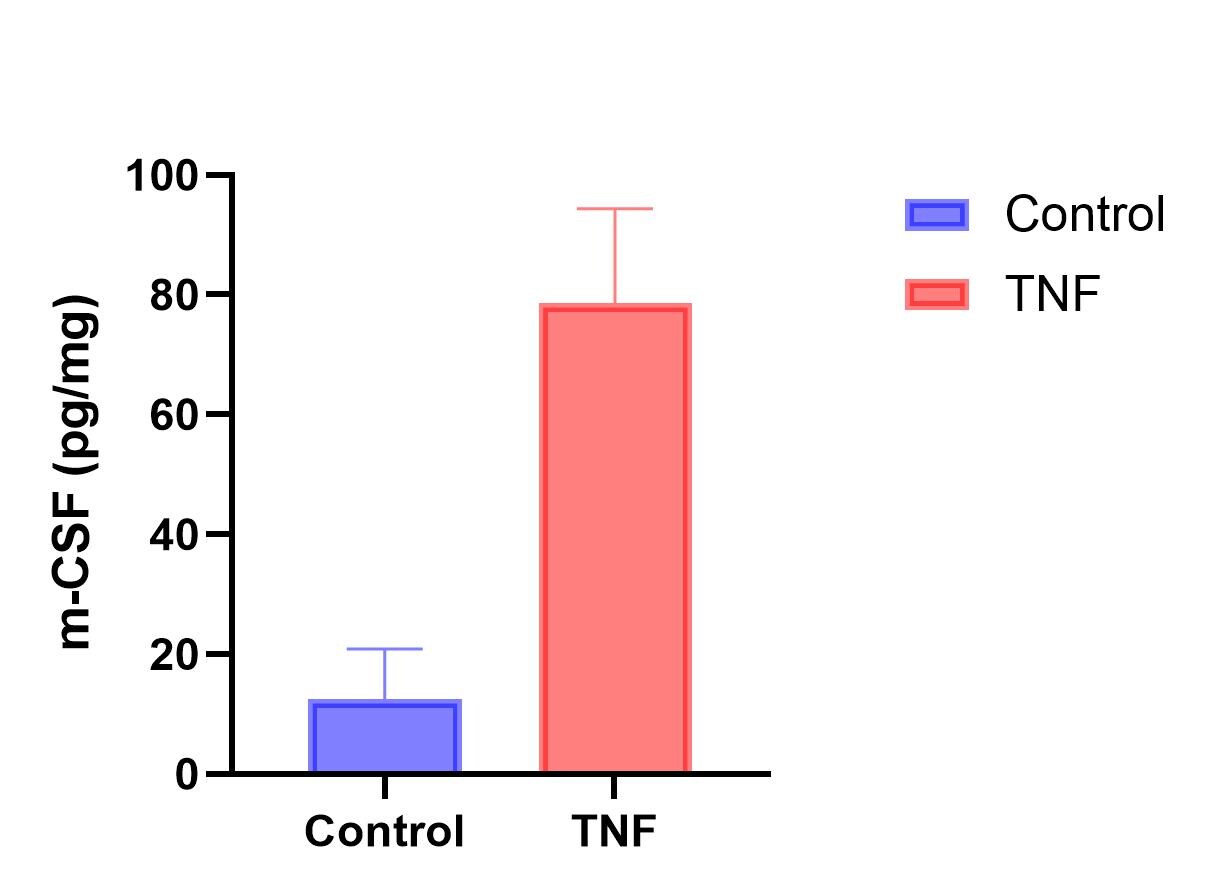
Sample Tested: Bone marrow cellsSpecies: Mouse and MouseVerified Customer | Posted 03/28/2026g-csf in bone marrow cellsTNF-induced G-CSF in primary bone marrow cells
-
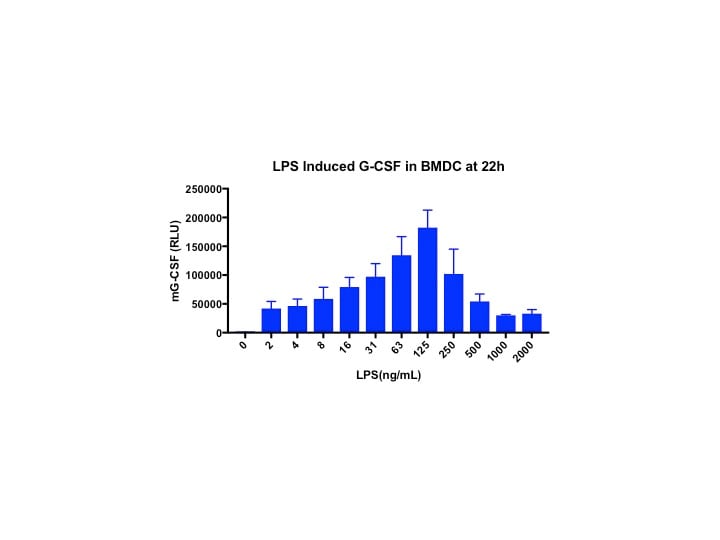
Sample Tested: BMDCVerified Customer | Posted 03/28/2018
-
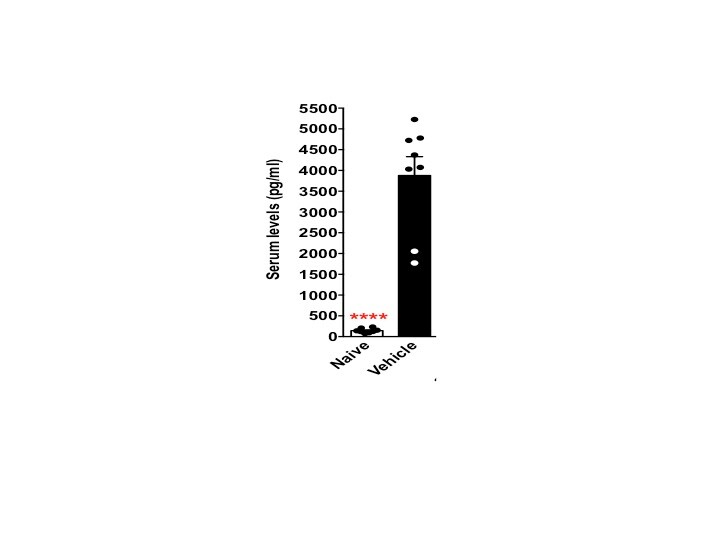
Sample Tested: mouse serumVerified Customer | Posted 08/04/2016
-
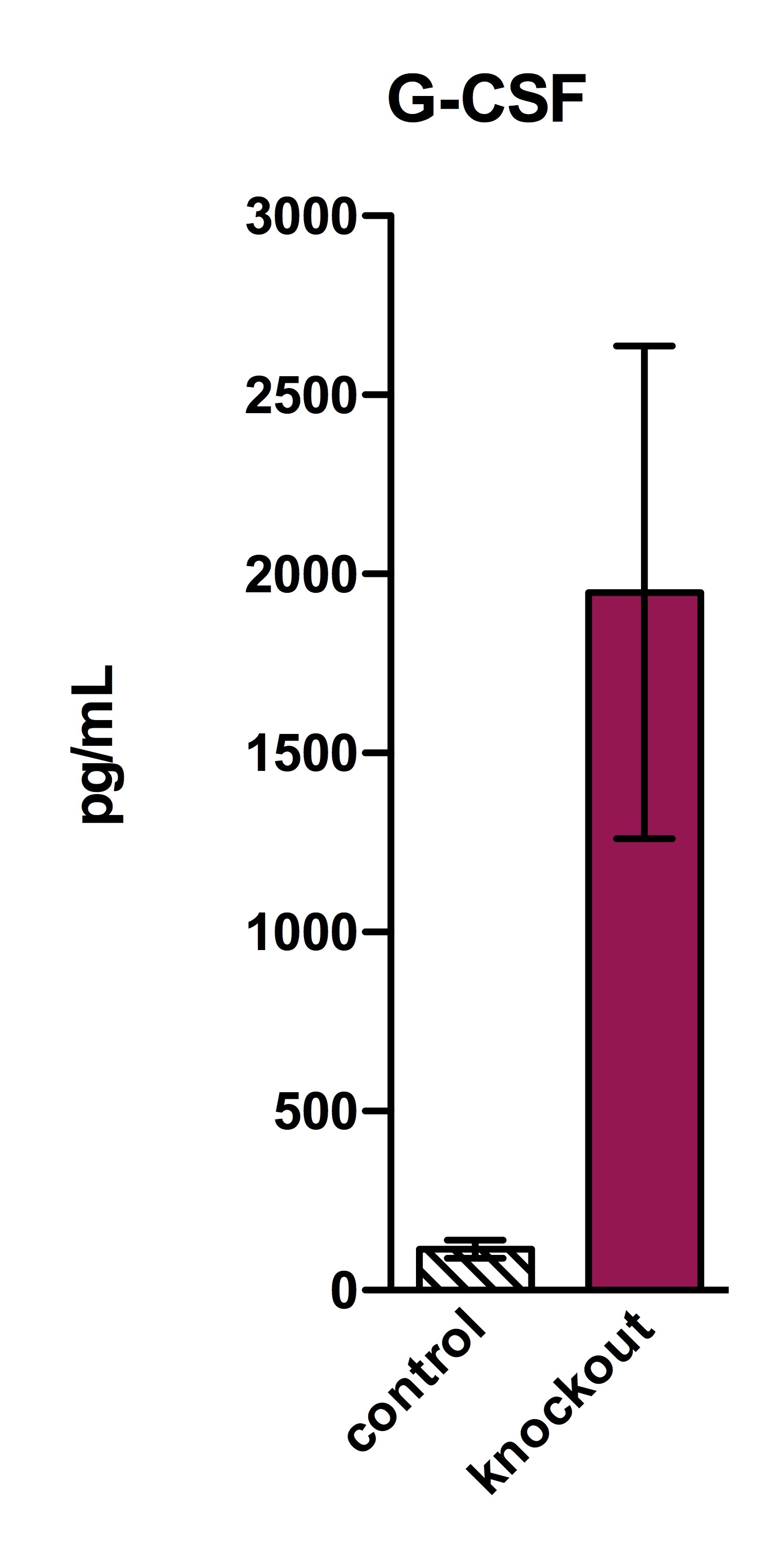
Sample Tested: murine lung homogenate supernatant, murine blood and murine serum (blood)Verified Customer | Posted 04/05/2016homogenate sup was filtered through 0.22um filter before use and then diluted 1:3 for analysis.
There are no reviews that match your criteria.
Protocols
View specific protocols for Mouse G-CSF DuoSet ELISA (DY414):
GENERAL ELISA PROTOCOL
Plate Preparation
- Dilute the Capture Antibody to the working concentration in PBS without carrier protein. Immediately coat a 96-well microplate with 100 μL per well of the diluted Capture Antibody. Seal the plate and incubate overnight at room temperature.
- Aspirate each well and wash with Wash Buffer, repeating the process two times for a total of three washes. Wash by filling each well with Wash Buffer (400 μL) using a squirt bottle, manifold dispenser, or autowasher. Complete removal of liquid at each step is essential for good performance. After the last wash, remove any remaining Wash Buffer by aspirating or by inverting the plate and blotting it against clean paper towels.
- Block plates by adding 300 μL Reagent Diluent to each well. Incubate at room temperature for a minimum of 1 hour.
- Repeat the aspiration/wash as in step 2. The plates are now ready for sample addition.
Assay Procedure
- Add 100 μL of sample or standards in Reagent Diluent, or an appropriate diluent, per well. Cover with an adhesive strip and incubate 2 hours at room temperature.
- Repeat the aspiration/wash as in step 2 of Plate Preparation.
- Add 100 μL of the Detection Antibody, diluted in Reagent Diluent, to each well. Cover with a new adhesive strip and incubate 2 hours at room temperature.
- Repeat the aspiration/wash as in step 2 of Plate Preparation.
- Add 100 μL of the working dilution of Streptavidin-HRP to each well. Cover the plate and incubate for 20 minutes at room temperature. Avoid placing the plate in direct light.
- Repeat the aspiration/wash as in step 2.
- Add 100 μL of Substrate Solution to each well. Incubate for 20 minutes at room temperature. Avoid placing the plate in direct light.
- Add 50 μL of Stop Solution to each well. Gently tap the plate to ensure thorough mixing.
- Determine the optical density of each well immediately, using a microplate reader set to 450 nm. If wavelength correction is available, set to 540 nm or 570 nm. If wavelength correction is not available, subtract readings at 540 nm or 570 nm from the readings at 450 nm. This subtraction will correct for optical imperfections in the plate. Readings made directly at 450 nm without correction may be higher and less accurate.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- Troubleshooting Guide: ELISA
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways