EGF is the prototypic member of a family of growth factors that also includes amphiregulin, betacellulin, epigen, epiregulin, HB-EGF, neuregulins-1 through -6, and TGF-alpha (1). These proteins contain EGF-like domains with three intramolecular disulfide bonds between conserved cysteines (2). EGF family members are synthesized as transmembrane preproproteins with varying numbers of EGF-like domains (3). The extracellular region of mouse pro-EGF contains eight LDL R class B repeats and nine EGF-like domains (4). Mature EGF is derived from the juxtamembrane EGF-like domain. EGF binds ErbB1 and induces the formation of homodimers or heterodimers containing ErbB2 (5). Pro-EGF is most highly expressed in the submaxillary gland and kidney (6). In the kidney, the 190 kDa preproprotein is cleaved by membrane-associated serine proteases, liberating the extracellular region which is subsequently processed into smaller fragments including the 6 kDa mature EGF (7‑10). The various cleavage products produced in the kidney also are present in urine (9‑11). In the submaxillary gland, however, nearly all EGF is processed intracellularly and stored in secretory vesicles (6, 12). The soluble precursor binds ErbB1 and induces cellular proliferation, although it is significantly less potent than mature EGF (8, 9). In human thyroid carcinoma cells, a splice variant of pro-EGF with a deletion in the cytoplasmic domain induces increased proliferative activity relative to wild-type pro-EGF (13). Within the extracellular region, mouse pro-EGF shares 79% amino acid sequence identity with rat pro-EGF and 67%‑69% with human, canine, feline, and porcine pro-EGF.

Key Product Details
Species Reactivity
Mouse
Applications
Immunohistochemistry, Western Blot
Label
Unconjugated
Antibody Source
Monoclonal Rat IgG2A Clone # 463004
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse EGF
Trp29-Arg1029
Accession # P01132
Trp29-Arg1029
Accession # P01132
Specificity
Detects mouse Pro EGF in direct ELISAs and Western blots. In direct ELISAs, no cross-reactivity with recombinant mouse (rm) Amphiregulin, rmEpigen, rmEpiregulin, rmEGF, rmMFG-E8, recombinant human (rh) EGF, rhHB-EGF, rhHRG alpha, rhHRG beta, or recombinant rat EGF is observed.
Clonality
Monoclonal
Host
Rat
Isotype
IgG2A
Scientific Data Images for Mouse Pro EGF Antibody
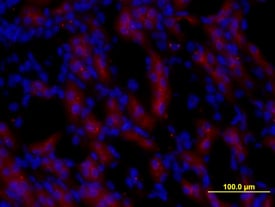
Pro EGF in Mouse Kidney.
Pro Epidermal Growth Factor (Pro EGF) was detected in perfusion fixed frozen sections of mouse kidney using Rat Anti-Mouse Pro EGF Monoclonal Antibody (Catalog # MAB5256) at 10 µg/mL for 3 hours at room temperature. Tissue was stained (red) and counterstained with DAPI (blue). View our protocol for Fluorescent IHC Staining of Frozen Tissue Sections.Applications for Mouse Pro EGF Antibody
Application
Recommended Usage
Immunohistochemistry
8-25 µg/mL
Sample: Perfusion fixed frozen sections of mouse kidney
Sample: Perfusion fixed frozen sections of mouse kidney
Western Blot
1 µg/mL
Sample: Recombinant Mouse Pro EGF aa 29-1029 (Catalog # 4095-EG) under non-reducing conditions only
Sample: Recombinant Mouse Pro EGF aa 29-1029 (Catalog # 4095-EG) under non-reducing conditions only
Reviewed Applications
Read 2 reviews rated 5 using MAB5256 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: EGF
References
- Singh, A.B. and R.C. Harris (2005) Cell. Signal. 17:1183.
- Wouters, M.A. et al. (2005) Prot. Sci. 14:1091.
- Sanderson, M.P. et al. (2006) Growth Factors 24:121.
- Gray, A. et al. (1983) Nature 303:722.
- Jorissen, R.N. et al. (2003) Exp. Cell Res. 284:31.
- Rall, L.B. et al. (1985) Nature 313:228.
- Le Gall, S.M. et al. (2004) Regul. Pept. 122:119.
- Breyer, J.A. and S. Cohen (1990) J. Biol. Chem. 265:16564.
- Parries, G. et al. (1995) J. Biol. Chem. 270:27954.
- Le Gall, S.M. et al. (2003) J. Biol. Chem. 278:45255.
- Lakshmanan, J. et al. (1990) Biochem. Biophys. Res. Commun. 173:902.
- Pasquini, F. et al. (1974) Exp. Cell Res. 86:233.
- Pyka, J. et al. (2005) Cancer Res. 65:1343.
Long Name
Epidermal Growth Factor
Alternate Names
HOMG4, URG, Urogastrone
Gene Symbol
EGF
UniProt
Additional EGF Products
Product Documents for Mouse Pro EGF Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse Pro EGF Antibody
For research use only
Related Research Areas
Citations for Mouse Pro EGF Antibody
Customer Reviews for Mouse Pro EGF Antibody (2)
5 out of 5
2 Customer Ratings
Have you used Mouse Pro EGF Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
2 of
2 reviews
Showing All
Filter By:
-
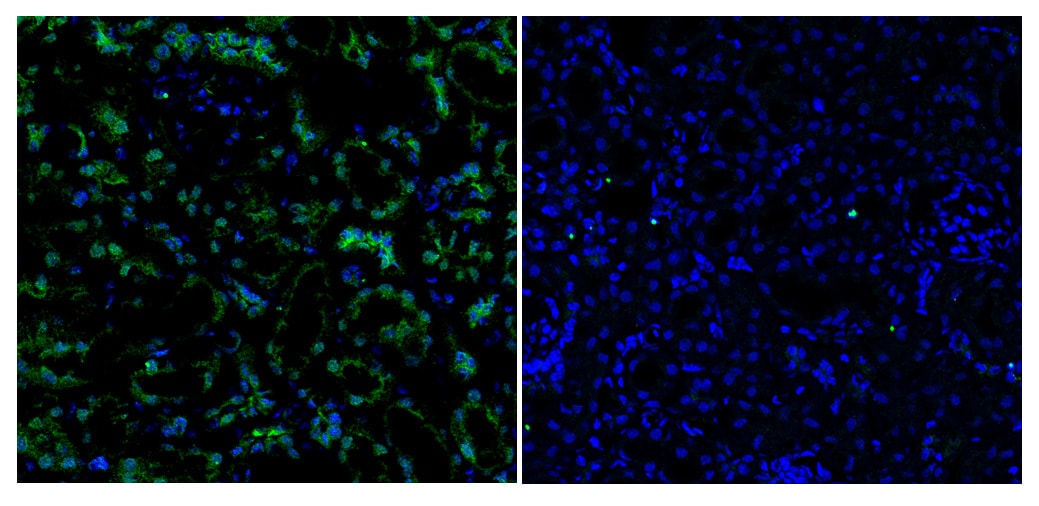
Application: ImmunohistochemistrySample Tested: Brain tissueSpecies: MouseVerified Customer | Posted 10/26/2021
-
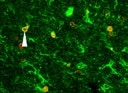
Application: ImmunocytochemistrySample Tested: Mouse kidneySpecies: MouseVerified Customer | Posted 05/17/2017Pro-EGF staining in normal C57 kidney tissue (Green). The right image is negative control with only the secondary antibody added.
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars