Vascular endothelia growth factor D (VEGF-D), also known as c-fos-induced growth factor (FIGF), is a secreted glycoprotein of the VEGF/PDGF family. VEGFs regulate angiogenesis and lymphangiogenesis during development and tumor growth, and are characterized by eight conserved cysteine residues that form a cysteine-knot structure (1‑3). VEGF-C and VEGF-D, which share 23% amino acid (aa) sequence identity, are uniquely expressed as preproproteins that contain long N- and C-terminal propeptide extensions around the VEGF homology domain (VHD) (1, 2). Proteolytic processing of either 358 aa or 326 aa splice forms of mouse VEGF-D preproprotein creates a secreted proprotein. Further processing by extracellular serine proteases, such as plasmin or furin-like proprotein convertases, forms mature VEGF-D consisting of non-covalently linked 42 kDa homodimers of the 117 aa VHD (4‑7). Mature mouse VEGF-D shares 94%, 99%, 93%, 91% and 89% aa identity with the VHD of human, rat, equine, canine and bovine VEGF-D, respectively. It is expressed in adult lung, heart, muscle, and small intestine, and is most abundantly expressed in fetal lungs and skin (1-4). Mouse and human VEGF-D are ligands for VEGF receptor 3 (VEGF-R3, also called Flt-4) that are active across species and show enhanced affinity when processed (8). Unlike human VEGF‑D, which is also a ligand for VEGF-R2 (also called Flk-1 or KDR), mouse VEGF-D does not bind to VEGF-R2 (8). VEGF-R3 is strongly expressed in lymphatic endothelial cells and is essential for regulation of the growth and differentiation of lymphatic endothelium (1, 2). While VEGF-C is the critical ligand for VEGF-R3 during embryonic lymphatic development, VEGF-D is most active in neonatal lymphatic maturation and bone growth (9‑11). Both promote tumor lymphangiogenesis (12). Consonant with their activity on VEGF receptors, binding of VEGF-C and VEGF-D to neuropilins contributes to VEGF-R3 signaling in lymphangiogenesis, while binding to integrin alpha 9 beta 1 mediates endothelial cell adhesion and migration (13, 14).

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Human, Mouse
Applications
Validated:
Immunohistochemistry, Western Blot
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse VEGF-D
Phe98-Ser206
Accession # P97946
Phe98-Ser206
Accession # P97946
Specificity
Detects mouse VEGF-D in direct ELISAs and Western blots. In direct ELISAs, less than 40% cross-reactivity with recombinant human (rh) VEGF-D is observed, and less than 1% cross-reactivity with recombinant mouse (rm) VEGF
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Mouse VEGF‑D Antibody
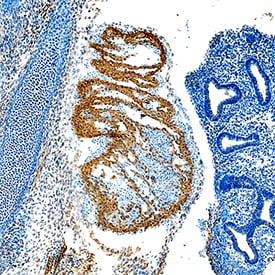
VEGF-D in Mouse Lung Tissue.
VEGF-D was detected in immersion fixed paraffin-embedded sections of mouse lung tissue using Goat Anti-Mouse VEGF-D Antigen Affinity-purified Polyclonal Antibody (Catalog # AF469) at 15 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Goat IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC004). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm in pneumocytes in lung. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Mouse VEGF-D by Immunohistochemistry
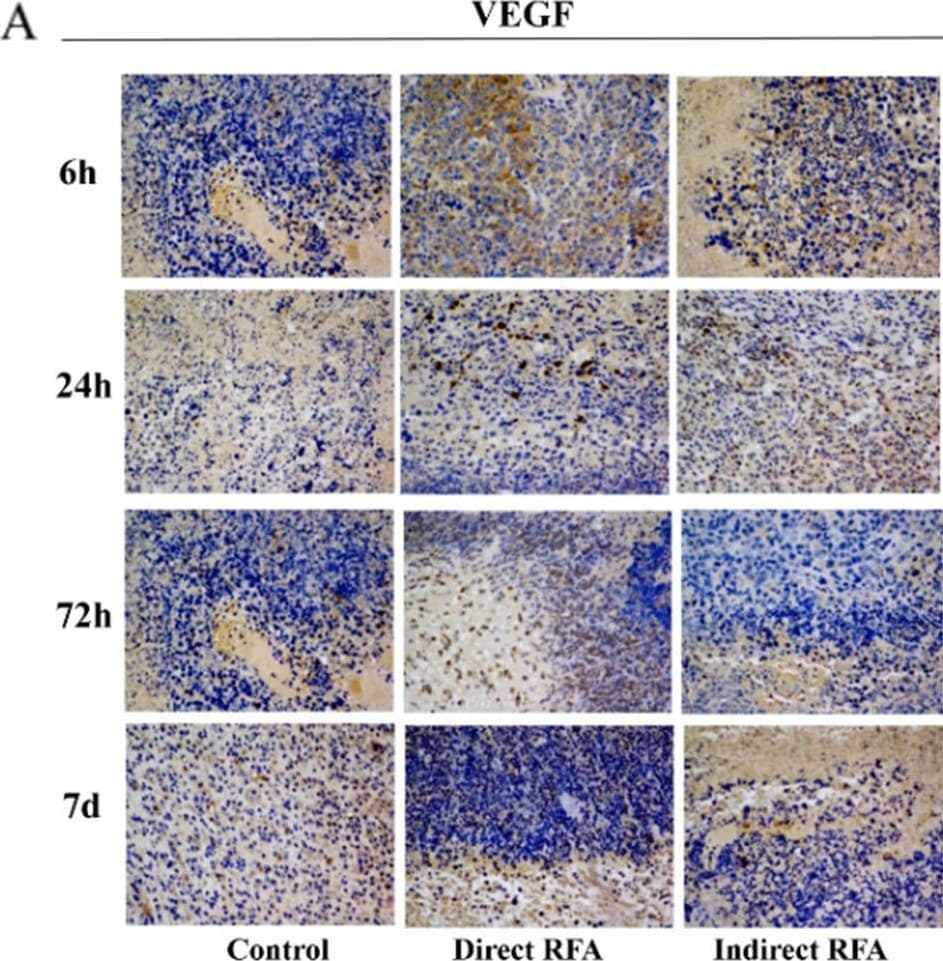
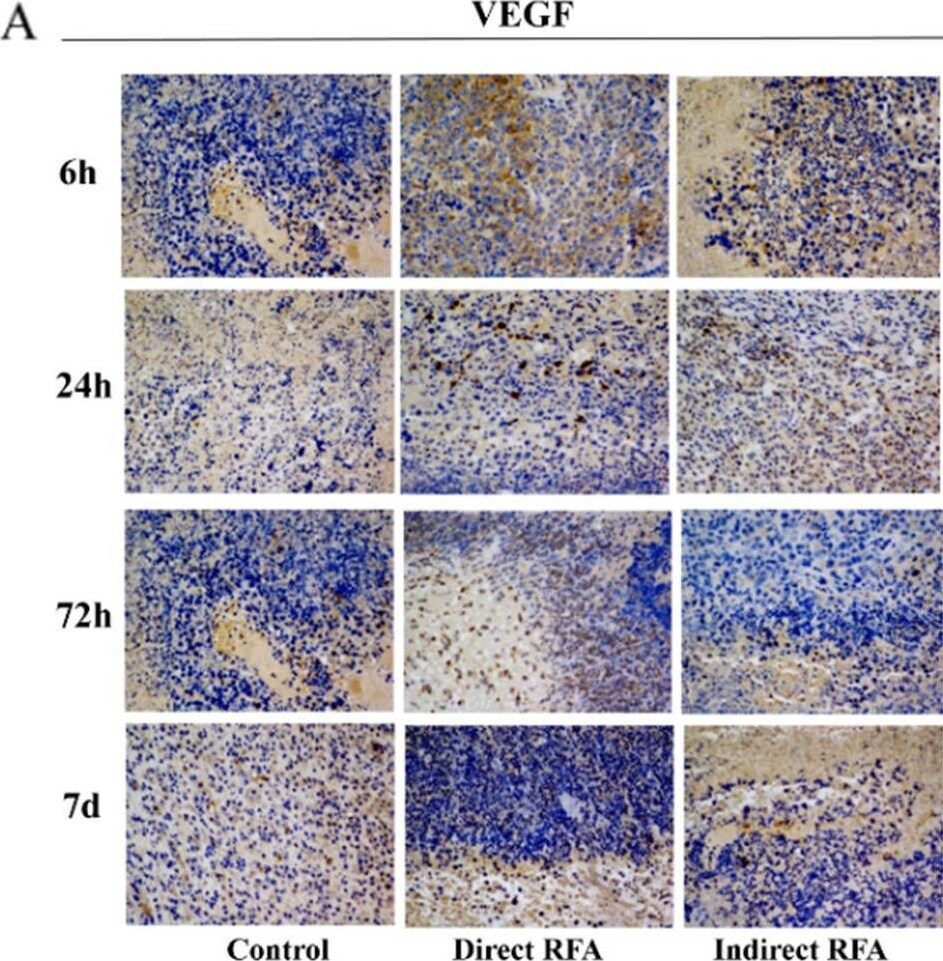
Assessment of pathologic findings obtained at 6 h, 24 h, 72 h and 7 days after treatment in different groups and the semi-quantitative analysis of VEGF (A-B) and CD31 (C-D). The expression of VEGF in the direct RFA group and indirect RFA group were always similar at all time points. The expression of CD31 (red) was increased at 24 h after RFA in both direct RFA and indirect RFA groups compared to the control group. The bar graph showed the percentage of positive cells among the direct RFA group (blue), indirect RFA group (red) and control group (grey) at different time points after RFA. The representative microphotographs were under 200 × magnification (A, C) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35710408), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse VEGF-D by Immunohistochemistry
Assessment of pathologic findings obtained at 6 h, 24 h, 72 h and 7 days after treatment in different groups and the semi-quantitative analysis of VEGF (A-B) and CD31 (C-D). The expression of VEGF in the direct RFA group and indirect RFA group were always similar at all time points. The expression of CD31 (red) was increased at 24 h after RFA in both direct RFA and indirect RFA groups compared to the control group. The bar graph showed the percentage of positive cells among the direct RFA group (blue), indirect RFA group (red) and control group (grey) at different time points after RFA. The representative microphotographs were under 200 × magnification (A, C) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35710408), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse VEGF‑D Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded mouse lung tissue
Sample: Immersion fixed paraffin-embedded mouse lung tissue
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse VEGF-D (Catalog # 469-VD)
Sample: Recombinant Mouse VEGF-D (Catalog # 469-VD)
Reviewed Applications
Read 1 review rated 3 using AF469 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: VEGF-D
References
- Roy, H. et al. (2006) FEBS Lett. 580:2879.
- Otrock, Z.H. et al. (2007) Blood Cells Mol. Dis. 38:258.
- Orlandini, M. et al. (1996) Proc. Natl. Acad. Sci. USA 93:11675.
- Stacker, S.A. et al. (1999) J. Biol. Chem. 274:32127.
- McColl, B.K. et al. (2003) J. Exp. Med. 198:863.
- McColl, B.K. et al. (2007) FASEB J. 21:1088.
- Baldwin, M.E. et al. (2001) J. Biol. Chem. 276:44307.
- Baldwin, M.E. et al. (2001) J. Biol. Chem. 276:19166.
- Baldwin, M.E. et al. (2005) Mol. Cell. Biol. 25:2441.
- Karpanen, T. et al. (2006) Am. J. Pathol. 169:708.
- Orlandini, M. et al. (2006) J. Biol. Chem. 281:17961.
- Stacker, S.A. et al. (2001) Nature Med. 7:186.
- Karpanen, T. et al. (2006) FASEB J. 20:1462.
- Vlahakis, N.E. et al. (2005) J. Biol. Chem. 280:4544.
Long Name
Vascular Endothelial Growth Factor D/cFos-induced Growth Factor
Alternate Names
FIGF, VEGFD
Gene Symbol
VEGFD
UniProt
Additional VEGF-D Products
Product Documents for Mouse VEGF‑D Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse VEGF‑D Antibody
For research use only
Citations for Mouse VEGF‑D Antibody
Customer Reviews for Mouse VEGF‑D Antibody (1)
3 out of 5
1 Customer Rating
Have you used Mouse VEGF‑D Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Western BlotSample Tested: Adult heartSpecies: MouseVerified Customer | Posted 03/13/2020Dilution used was 1:300 in 3% Milk /TBS-T solution with overnight incubation at 4 degrees. Anti-Goat HRP 1:500 was used as secondary antibody (1h at room temperature)
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...