Recombinant Human CD155/PVR Fc Chimera Protein, CF
R&D Systems | Catalog # 9174-CD
Analyzed by SEC-MALS

Loading...
Key Product Details
- R&D Systems HEK293-derived Recombinant Human CD155/PVR Fc Chimera Protein (9174-CD)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
HEK293
Accession Number
Structure / Form
Disulfide-linked homodimer
Applications
Bioactivity
Loading...
Product Specifications
Source
Human embryonic kidney cell, HEK293-derived human CD155/PVR protein
| Human CD155/PVR (Asp28-Asn343) Accession # NP_006496 |
IEGRMD | Human IgG1 (Pro100-Lys330) |
| N-terminus | C-terminus |
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Asp28
Predicted Molecular Mass
61 kDa
SDS-PAGE
81-98 kDa, reducing conditions
Activity
Measured by its binding ability in a functional ELISA.
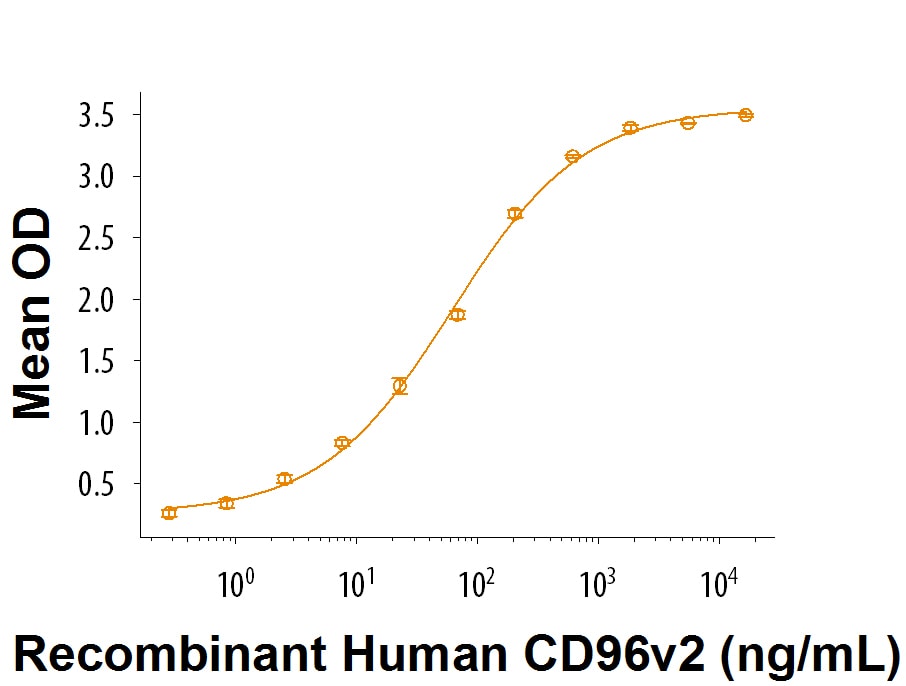
When Recombinant Human CD155/PVR Fc Chimera is immobilized at 1 μg/mL (100 μL/well), the concentration of Recombinant Human CD96 v2 Fc Chimera (Catalog # 9556-CD) that produces 50% of the optimal binding response is 30-150 ng/mL.
When Recombinant Human CD155/PVR Fc Chimera is immobilized at 1 μg/mL (100 μL/well), the concentration of Recombinant Human CD96 v2 Fc Chimera (Catalog # 9556-CD) that produces 50% of the optimal binding response is 30-150 ng/mL.
Reviewed Applications
Read 1 review rated 4 using 9174-CD in the following applications:
Scientific Data Images for Recombinant Human CD155/PVR Fc Chimera Protein, CF
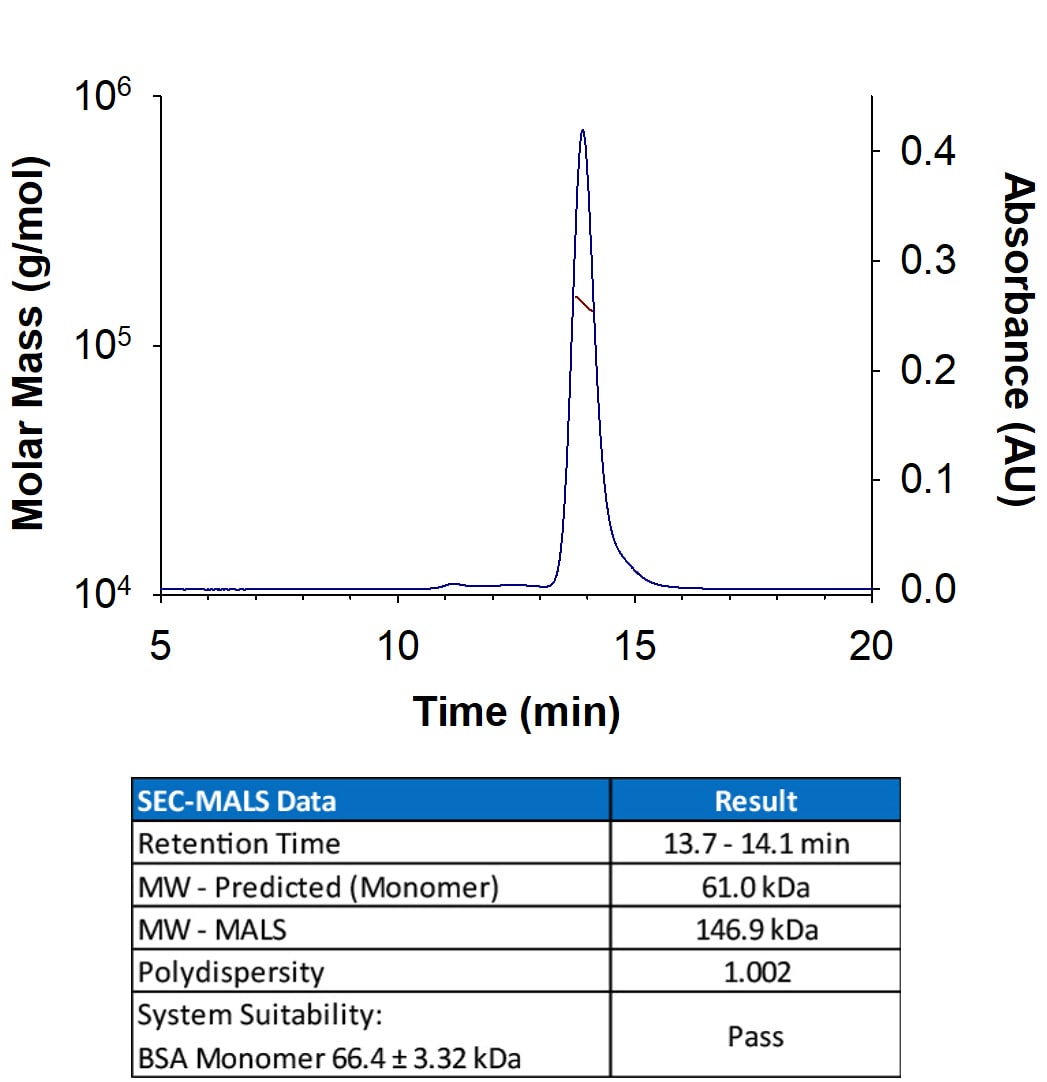
Recombinant Human CD155/PVR Fc Chimera Protein SEC-MALS.
Recombinant Human CD155/PVR Fc Chimera (Catalog # 9174-CD) has a molecular weight (MW) of 146.9 kDa as analyzed by SEC-MALS, suggesting that this protein is a homodimer. MW may differ from predicted MW due to post-translational modifications (PTMs) present (i.e. Glycosylation).Recombinant Human CD155/PVR Fc Chimera Protein Binding Activity
When Recombinant Human CD155/PVR Fc Chimera (Catalog # 9174-CD) is immobilized at 1 µg/mL, Recombinant Human CD96 v2 Fc Chimera (9556-CD) binds with an ED50 of 30-150 ng/mL.Binding of Human TIGIT to CD155/PVR by surface plasmon resonance (SPR).
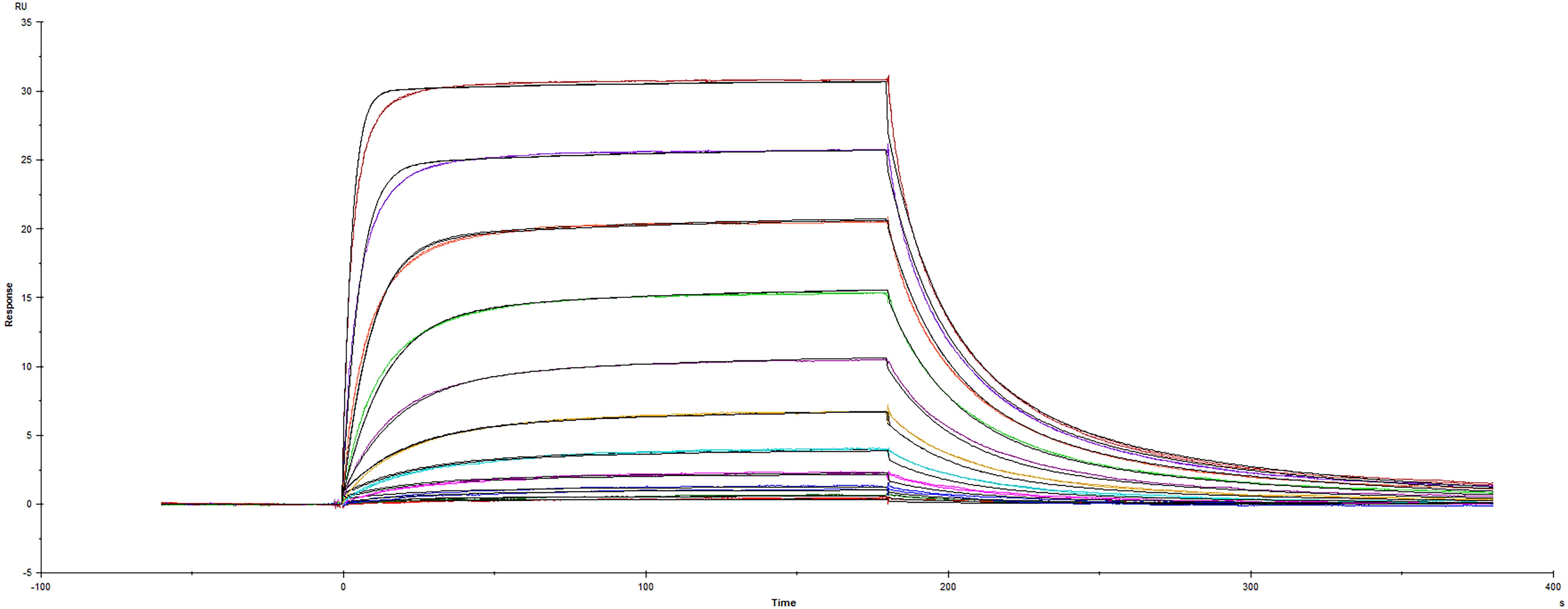
Recombinant Human CD155/PVR Fc protein (Catalog # 9174-CD) was immobilized on a Biacore Sensor Chip CM5, and binding to Recombinant Human TIGIT (A103) Fc protein (7898-TGB) was measured at a concentration range between 0.531 nM and 544 nM. The double-referenced sensorgram was fit to a 1:1 binding model to determine the binding kinetics and affinity, with an affinity constant of KD=67.7 nM.Formulation, Preparation, and Storage
9174-CD
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 500 μg/mL in PBS.
Loading...
|
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: CD155/PVR
References
- Mandai, K. et al. (2015) Curr. Top. Dev. Biol. 112:197.
- Mendelsohn, C.L. et al. (1989) Cell 56:855.
- Sato, T. et al. (2004) Genes to Cells 9:791.
- Meyer, D. et al. (2009) J. Biol. Chem. 284:2235.
- Koike, S. et al. (1990) EMBO J. 9:3217.
- Escalante, N.K. et al. (2011) Arterioscler. Thromb. Vasc. Biol. 31:1177.
- Xu, Z. and B. Jin (2010) Cell. Mol. Immunol. 7:11.
- Reymond, N. et al. (2004) J. Exp. Med. 199:1331.
- Maier, M.K. et al. (2007) Eur. J. Immunol. 37 :2214.
- Lozano, E. et al. (2013) J. Immunol. 191:3673.
- Yamashita-Kanemaru, Y. et al. (2015) J. Immunol. 194:5644.
- Mueller, S. and E. Wimmer (2003) J. Biol. Chem. 278:31251.
- Chan, C.J. et al. (2010) J. Immunol. 184:902.
- Qui, Q. et al. (2010) J. Immunol. 184:1681.
- Seth, S. et al. (2009) Eur. J. Immunol. 39:3160.
- Stanietsky, N. et al. (2009) Proc. Natl. Acad. Sci. USA 106:17858.
Long Name
Poliovirus Receptor
Alternate Names
CD155, HVED, Necl-5, PVR, PVS
Entrez Gene IDs
Gene Symbol
PVR
UniProt
Additional CD155/PVR Products
Product Documents for Recombinant Human CD155/PVR Fc Chimera Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human CD155/PVR Fc Chimera Protein, CF
For research use only
Citations for Recombinant Human CD155/PVR Fc Chimera Protein, CF
Customer Reviews for Recombinant Human CD155/PVR Fc Chimera Protein, CF (1)
4 out of 5
1 Customer Rating
Have you used Recombinant Human CD155/PVR Fc Chimera Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
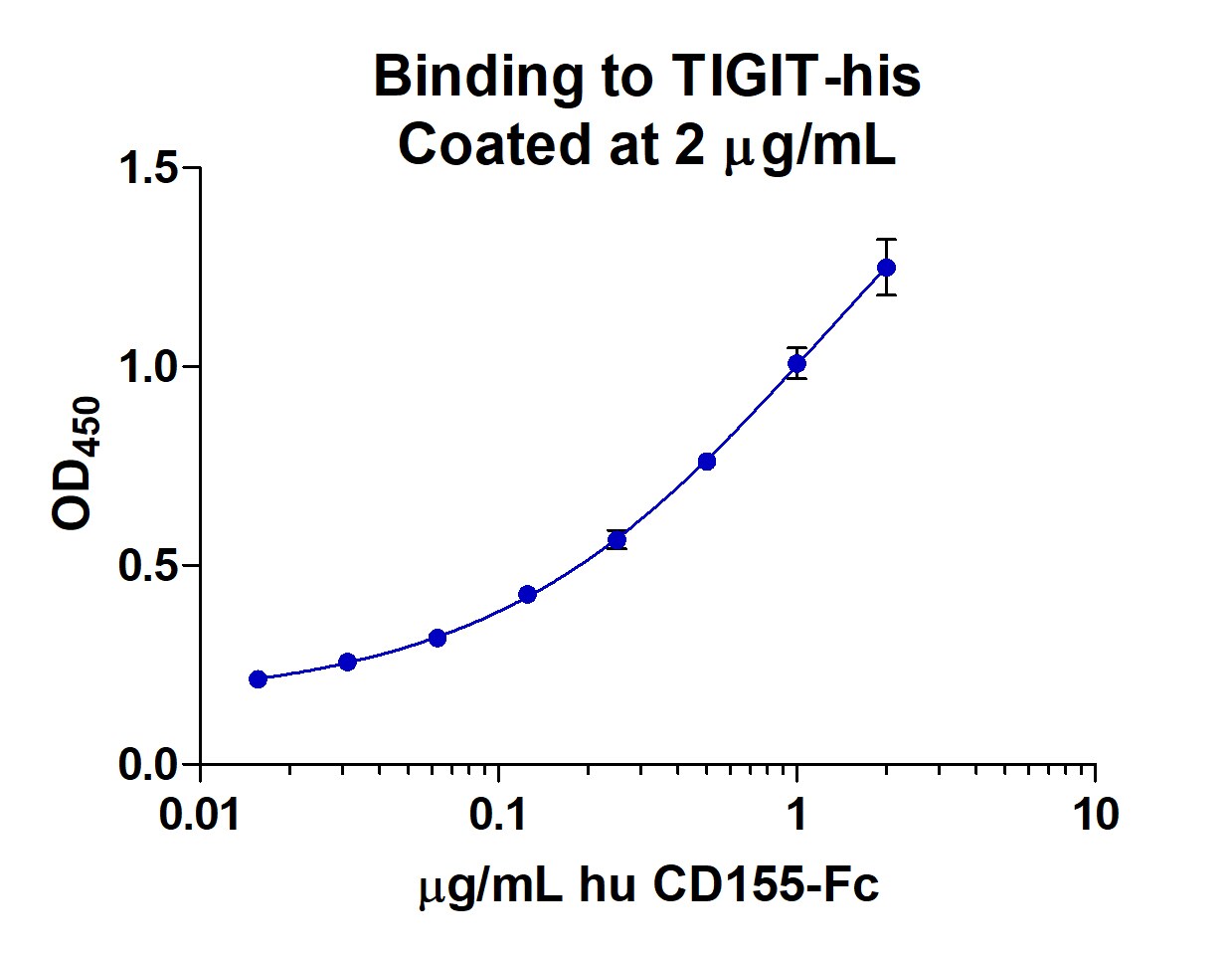
Application: Binding assay/Protein-protein interactionVerified Customer | Posted 11/09/2018ELISA binding to TIGIT
There are no reviews that match your criteria.
Loading...