Human matriptase, encoded by the ST14 (suppression of tumorogenicity 14) gene, is also known as tumor associated differentially expressed gene 15 protein/TADG‑15), epithin, and membrane‑type serine protease 1/MT‑SP1 (1). Predicted to have a significant role in tumor biology, matriptase may be a novel target for anti‑cancer therapy (2). However, expressed in most human epithelia, matriptase is also important in several physiological processes (1). For example, it activates prostasin to initiate a protease cascade that is essential for epidermal differentiation (3), and it converts a single‑chain IGFBP-rp1 into the two‑chain form (4). Matriptase is a type II transmembrane serine protease with a complex modular structure (1). The 855 amino acid (aa) sequence of human matriptase consists of a cytoplasmic tail (aa 1‑55), a transmembrane domain (aa 56‑76), and an extracellular portion (aa 77‑855). The latter contains the following domains: SEA (aa 86‑201), two CUBs (aa 214‑334 and 340‑447), four LDLRAs (aa 452‑486, 487‑523, 524‑560, and 566‑603), and a serine protease (aa 615‑855). The physiological activation of the single‑chain zymogen requires the cleavage at the SEA domain within the ER or Golgi, association with HAI-1, which facilitates the transport of the protease to the cell surface, and auto‑cleavage at QAR-V(615)VGG (1). The activated matriptase is inhibited by HAI-1, and the resulting HAI-1 complex can be shed from the cell surface (1). R&D Systems recombinant human (rh) ST14 corresponds to the catalytic domain, and is inhibited effectively by rhHAI-1 and rhHAI-2A (Catalog # http://www.rndsystems.com/product_results.aspx?k=1048-PI">1048‑PI and http://www.rndsystems.com/product_results.aspx?k=1106-PI">1106‑PI).
Recombinant Human Matriptase/ST14 Catalytic Domain, CF
R&D Systems | Catalog # 3946-SEB

Key Product Details
- R&D Systems E. coli-derived Recombinant Human Matriptase/ST14 Catalytic Domain (3946-SEB)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Applications
Product Specifications
Source
Gly596-Val855, with an N-terminal Met and 6-His tag
The protein was purified, auto-activated and further purified.
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
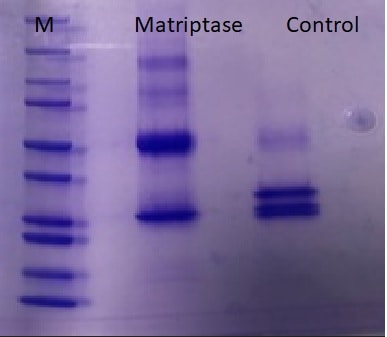
SDS-PAGE
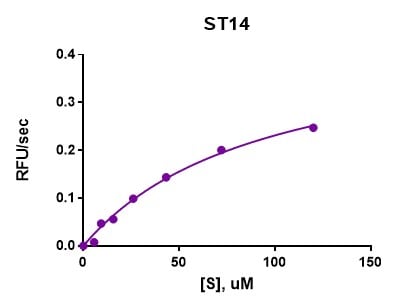
Activity
The specific activity is >10,000 pmol/min/μg, as measured under the described conditions.
Reviewed Applications
Read 4 reviews rated 5 using 3946-SEB in the following applications:
Scientific Data Images for Recombinant Human Matriptase/ST14 Catalytic Domain, CF
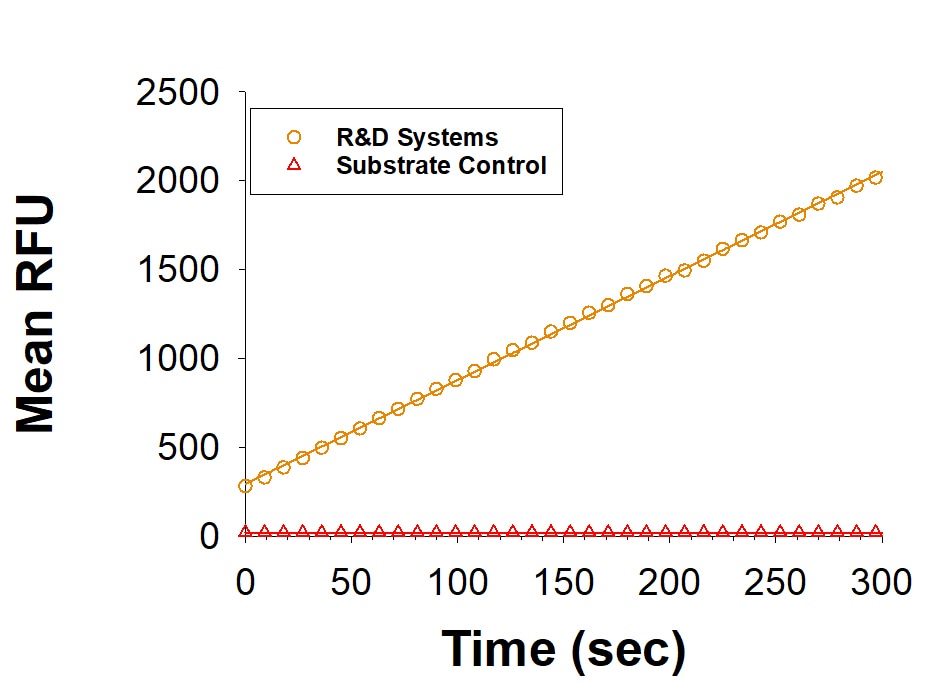
Recombinant Human Matriptase/ST14 Catalytic Domain Enzyme Activity
Recombinant Human Matriptase is measured by its ability to cleave the fluorogenic peptide substrate Boc-QAR-AMC (Catalog # ES014).Formulation, Preparation, and Storage
3946-SEB
| Formulation | Supplied as a 0.2 μm filtered solution in Tris-HCl and Glycerol. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: Matriptase/ST14
References
- List, K. et al. (2006) Mol. Med. 12:1.
- Uhland, K. (2006) Cell. Mol. Life Sci. 63:2968.
- Netzel-Arnett, S. et al. (2006) J. Biol. Chem. 281:32941.
- Ahmed, S. et al. (2006) FEBS J. 273:615.
Alternate Names
Gene Symbol
UniProt
Additional Matriptase/ST14 Products
Product Documents for Recombinant Human Matriptase/ST14 Catalytic Domain, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human Matriptase/ST14 Catalytic Domain, CF
For research use only
Related Research Areas
Citations for Recombinant Human Matriptase/ST14 Catalytic Domain, CF
Customer Reviews for Recombinant Human Matriptase/ST14 Catalytic Domain, CF (4)
Have you used Recombinant Human Matriptase/ST14 Catalytic Domain, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Enzymatic activity in vitroVerified Customer | Posted 12/11/2019Substrate fusion proteins were purified from Drosophila S2R+ cells using anti-RFP magnetic agarose Substrates were incubated with 0.1 ug matriptase at 37°C for one hour. Reaction was stopped by reducing/denaturing conditions. Samples were examined by SDS-PAGE/western blots and antibody stainings using respective antibodys.
-
Application: SDS-PAGE ControlVerified Customer | Posted 04/07/2019
-
Application: SDS-PAGE ControlVerified Customer | Posted 03/07/2019Protein sample treated with Matriptase enzyme was cleaved after 30min incubation compared with control (untreated). Protein ladder ran to compare sizes
-
Application: Enzymatic activity in vitroVerified Customer | Posted 07/31/20185nM of activated ST14 used
There are no reviews that match your criteria.
Protocols
View specific protocols for Recombinant Human Matriptase/ST14 Catalytic Domain, CF (3946-SEB):
- Assay Buffer: 50 mM Tris, 50 mM NaCl, 0.01% (v/v) Tween® 20, pH 9.0
- Recombinant Human Matriptase/ST14 Catalytic Domain (rhMatriptase) (Catalog # 3946-SEB)
- Substrate: BOC-Gln-Ala-Arg-AMC (Catalog # ES014), 10 mM stock in DMSO
- F16 Black Maxisorp Plate (Nunc, Catalog # 475515)
- Fluorescent Plate Reader (Model: SpectraMax Gemini EM by Molecular Devices) or equivalent
- Dilute rhMatriptase to 0.1 µg/mL in Assay Buffer.
- Dilute Substrate to 50 µM in Assay Buffer.
- Load 50 µL of 0.1 µg/mL rhMatriptase into a plate, and start the reaction by adding 50 µL of 50 µM substrate. Include a Substrate Blank containing 50 µL of Assay Buffer and 50 µL of Substrate.
- Read at excitation and emission wavelengths of 380 nm and 460 nm (top read), respectively, in kinetic mode for 5 minutes.
- Calculate specific activity:
Specific Activity (pmol/min/µg) = | Adjusted Vmax* (RFU/min) x Conversion Factor** (pmol/RFU) |
| amount of enzyme (µg) |
*Adjusted for Substrate Blank.
**Derived using calibration standard 7-Amino, 4-Methyl Coumarin (AMC) (Sigma, Catalog # A9891).
Per Well:
- rhMatriptase: 0.005 µg
- Substrate: 25 µM
FAQs for Recombinant Human Matriptase/ST14 Catalytic Domain, CF
-
Q: Catalog # 3946-SEB is formulated with 10% glycerol. Will the glycerol prevent it from freezing?
A: No, this protein in 10% glycerol will freeze at -20 C and -80 C.