Integrin alpha M beta 2, also called MAC‑1 or complement receptor type 3 (CR3), is one of three leukocyte beta 2 integrins. The non‑covalent heterodimer of 170 kDa alpha M/CD11b and 95 kDa beta 2/CD18 integrin subunits is expressed mainly on myeloid and natural killer cells (1‑6). The alpha M vWFA or I‑domain, which contains adhesion sites, forms the N‑terminal head region with the alpha M beta ‑propeller and the beta 2 vWFA domain. Unlike most integrins, the calf domain of alpha M is lectin‑like and binds carbohydrates (7). Each subunit has a transmembrane sequence and a short cytoplasmic tail. The 1089 amino acid (aa) mouse alpha M/CD11b ECD shares 87% aa sequence identity with rat, and 70‑74% with human, equine, bovine, feline, and canine alpha M ECD. A potential mouse alpha M isoform lacks 117 aa within the beta ‑propeller. The 679 aa mouse beta 2/CD18 ECD shares 91% aa sequence identity with rat, and 80‑82% with human, bovine, canine, and porcine beta 2 ECD. Like other integrins, alpha M beta 2 has multiple activation states (1‑3). In the presence of divalent cations and "inside‑out" signaling, alpha M beta 2 is fully active and extended. In the inactive state, the heterodimer flexes in the center at the alpha M thigh and calf domains and beta 2 I‑EGF domains, impeding access to adhesion sites. Active alpha M beta 2 binds an unusually large number of adhesion partners, including the complement opsonin fragment iC3b, coagulation proteins fibrinogen, plasminogen and factor X, extracellular matrix (ECM) proteins fibronectin, laminin and collagen, and cell surface ICAMs, myelin basic protein and DC-SIGN (3, 4, 7). alpha M beta 2 lectin-like adhesion partners include heparin, bacterial lipopolysaccharides, and GPI‑linked glycoproteins such as uPAR and Fc gamma RIIIB (3, 7). Binding of platelet JAM‑C links platelets with myeloid and dendritic cell (DC) alpha M beta 2 and recruits these cells to inflamed or injured endothelium, while neutrophil alpha M beta 2 adheres to RAGE on inflamed endothelium; both are atherogenic events (3, 8, 9). However, activation of alpha M beta 2 inhibits alternative activation of macrophages and atherosclerotic foam cell formation (3, 10). alpha M beta 2 can either suppress or allow constitutive neutrophil apoptosis, depending on its ligand and activation state (3, 11, 12). Deletion of mouse alpha M causes defects in neutrophil adhesion and degranulation, while mutations of human or mouse beta 2 cause leukocyte adhesion deficiency (LAD‑1) and susceptibility to bacterial infections (3, 12, 13).
Recombinant Mouse Integrin alpha M beta 2 Protein, CF
R&D Systems | Catalog # 7959-AM


Key Product Details
- R&D Systems CHO-derived Recombinant Mouse Integrin alpha M beta 2 Protein (7959-AM)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Structure / Form
Applications
Product Specifications
Source
| Mouse Integrin alpha M (Phe17-Asn1105) Accession # NP_032427 |
His-Pro | GGGSGGGS | Acidic Tail | 6-His tag |
| Mouse Integrin beta 2 (Gln24-Asn702) Accession # P11835 |
His-Pro | GGGSGGGS | Basic Tail | |
| N-terminus | C-terminus | |||
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The ED50 for this effect is 0.4-1.6 μg/mL.
Formulation, Preparation, and Storage
7959-AM
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 400 μg/mL in PBS.
Loading...
|
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: Integrin alpha M beta 2
References
- Takada, Y. et al. (2007) Genome Biol. 8:215.
- Luo, B-H. et al. (2007) Annu. Rev. Immunol. 25:619.
- Tan, S.M. (2012) Biosci. Rep. 32:241.
- Pytela, R. (1988) EMBO J. 7:1371.
- Kishimoto, T. K. et al. (1987) Cell 48:681.
- Muto, S. et al. (1993) J. Clin. Immunol. 13:175.
- Xia, Y. et al. (2002) J. Immunol. 169:6417.
- Santoso, S. et al. (2002) J. Exp. Med. 196:679.
- Langer, H.F. et al. (2007) Arterioscler. Thromb. Vasc. Biol. 27:1463.
- Yakubenko, V.P. et al. (2011) Circ. Res. 108:544.
- Pluskota, E. et al. (2008) J. Immunol. 181:3609.
- Coxon, A. et al. (1996) Immunity 5:653.
- Lu, H. et al. (1997) J. Clin. Invest. 99:1340.
Entrez Gene IDs
Gene Symbol
Additional Integrin alpha M beta 2 Products
Product Documents for Recombinant Mouse Integrin alpha M beta 2 Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Mouse Integrin alpha M beta 2 Protein, CF
For research use only
Citations for Recombinant Mouse Integrin alpha M beta 2 Protein, CF
Customer Reviews for Recombinant Mouse Integrin alpha M beta 2 Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Mouse Integrin alpha M beta 2 Protein, CF and earn rewards!
Have you used Recombinant Mouse Integrin alpha M beta 2 Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
FAQs for Recombinant Mouse Integrin alpha M beta 2 Protein, CF
-
Q: What is the amino acid sequence of the acidic and basic tails?
A: Acidic and basic tails are added to the protein to help facilitate optimal activity. While we generally include sequence information on the product datasheet, the sequences of these tails are considered confidential information.
Associated Pathways
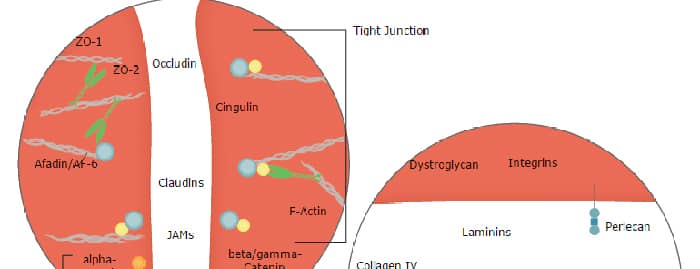
 Blood-Brain Barrier Pathway: Anatomy
Blood-Brain Barrier Pathway: Anatomy