Vascular endothelial growth factor (VEGF or VEGF‑A), also known as vascular permeability factor (VPF), is a potent mediator of both angiogenesis and vasculogenesis in the fetus and adult (1-3). It is a member of the PDGF family that is characterized by a cystine knot structure formed by eight conserved cysteine residues (4). Alternately spliced isoforms of 121, 145, 165, 183, 189, and 206 amino acids (aa) have been identified in humans, with 120, 164 and 188 aa isoforms found in mouse (2, 4). Isoforms other than VEGF120 and VEGF121 contain basic heparin-binding regions and are not freely diffusible (4). Mouse VEGF164 shares 97% aa sequence identity with corresponding regions of rat, 89% with human and porcine, 88% with bovine, and 90% with feline, equine and canine VEGF, respectively. VEGF binds the type I transmembrane receptor tyrosine kinases VEGF R1 (also called Flt-1) and VEGF R2 (Flk-1/KDR) on endothelial cells (4). Although affinity is highest for binding to VEGF R1, VEGF R2 appears to be the primary mediator of VEGF angiogenic activity (3, 4). Human VEGF165 binds the semaphorin receptor, Neuropilin-1 and promotes complex formation with VEGF R2 (5). VEGF is required during embryogenesis to regulate the proliferation, migration, and survival of endothelial cells (3, 4). In adults, VEGF functions mainly in wound healing and the female reproductive cycle (3). Pathologically, it is involved in tumor angiogenesis and vascular leakage (6, 7). Circulating VEGF levels correlate with disease activity in autoimmune diseases such as rheumatoid arthritis, multiple sclerosis and systemic lupus erythematosus (8). VEGF is induced by hypoxia and cytokines such as IL-1, IL-6, IL-8, oncostatin M and TNF-alpha (3, 4, 9).
Recombinant Mouse VEGF 164 Protein
R&D Systems | Catalog # 493-MV


Key Product Details
- R&D Systems Sf 21 (baculovirus)-derived Recombinant Mouse VEGF 164 Protein (493-MV)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Structure / Form
Applications
Product Specifications
Source
Ala27-Arg190
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The ED50 for this effect is 0.8-4 ng/mL.
Reviewed Applications
Read 8 reviews rated 4.9 using 493-MV in the following applications:
Formulation, Preparation, and Storage
Carrier Free
What does CF mean?CF stands for Carrier Free (CF). We typically add Bovine Serum Albumin (BSA) as a carrier protein to our recombinant proteins. Adding a carrier protein enhances protein stability, increases shelf-life, and allows the recombinant protein to be stored at a more dilute concentration. The carrier free version does not contain BSA.
What formulation is right for me?In general, we advise purchasing the recombinant protein with BSA for use in cell or tissue culture, or as an ELISA standard. In contrast, the carrier free protein is recommended for applications, in which the presence of BSA could interfere.
Carrier: 493-MV
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein. |
| Reconstitution | Reconstitute at 50 μg/mL in sterile PBS containing at least 0.1% human or bovine serum albumin. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Carrier Free: 493-MV/CF
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 50 µg/mL in sterile PBS. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: VEGF
References
- Breier, G. et al. (1992) Development 114:521.
- Shima, D.T. et al. (1996) J. Biol. Chem. 271:3877.
- Byrne, A.M. et al. (2005) J. Cell. Mol. Med. 9:777.
- Robinson, C.J. and S.E. Stringer (2001) J. Cell. Sci. 114:853.
- Pan, Q. et al. (2007) J. Biol. Chem. 282:24049.
- Weis, S.M. and D.A. Cheresh (2005) Nature 437:497.
- Thurston, G. (2002) J. Anat. 200:575.
- Carvalho, J.F. et al. (2007) J. Clin. Immunol. 27:246.
- Angelo, L.S. and R. Kurzrock (2007) Clin. Cancer Res. 13:2825.
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
UniProt
Additional VEGF Products
Product Documents for Recombinant Mouse VEGF 164 Protein
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Mouse VEGF 164 Protein
For research use only
Citations for Recombinant Mouse VEGF 164 Protein
Customer Reviews for Recombinant Mouse VEGF 164 Protein (8)
Have you used Recombinant Mouse VEGF 164 Protein?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: In vitro bioactivity in cell cultureVerified Customer | Posted 11/11/2024
-
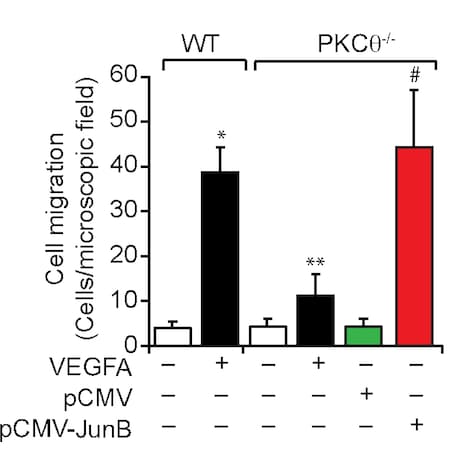
Application: Cell migration/motilityVerified Customer | Posted 04/06/2021
-
Application: Cell AdhesionVerified Customer | Posted 10/17/2019
-
Application: Cell ProliferationVerified Customer | Posted 05/03/2018
-
Application: SDS-PAGE ControlVerified Customer | Posted 04/26/2018
-
Application: Cell ProliferationVerified Customer | Posted 04/22/2018
-
Verified Customer | Posted 04/17/2018
-
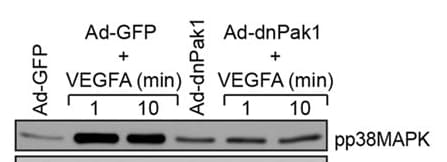
Application: VEGFA effect on p38MAPK activation in mouse retinal epithelial cellsVerified Customer | Posted 06/13/2017
There are no reviews that match your criteria.
Associated Pathways
 Embryonic and Induced Pluripotent Stem Cell Differentiation Pathways & Lineage-specific Markers
Embryonic and Induced Pluripotent Stem Cell Differentiation Pathways & Lineage-specific Markers
 Neural Stem Cell Differentiation Pathways & Lineage-specific Markers
Neural Stem Cell Differentiation Pathways & Lineage-specific Markers
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways