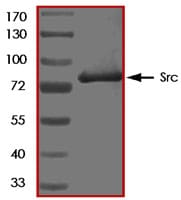
The non-receptor tyrosine kinase Src was originally identified as a transforming protein of the Rous sarcoma virus that had enzymatic ability to phosphorylate tyrosine in protein substrates (1). The proto-oncogene c-Src is the cellular homologue of viral Src (v-Src) and the founding member of the Src family kinases. c-Src is over-expressed and activated in a large number of human maligancies and has been linked to the development of cancer and progression to distant metastases (2). In addition to increasing cell proliferation, a key role of c-Src in cancer seems to be the ability to promote invasion and motility, functions that might contribute to tumor progression.
Although v-Src and c-Src share 88% amino acid identity, v-Src, unlike c-Src, is constitutively active mainly because it lacks a crucial c-terminal negative-regulatory region (3). As a result, v-Src is missing a tyrosine residue (Y530 in human c-Src) that upon phosphorylation contributes to c-Src assuming an inactive conformation.