VEGF Antibody (VG1) - BSA Free
Novus Biologicals | Catalog # NB100-664

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Specificity
Clonality
Host
Isotype
Scientific Data Images for VEGF Antibody (VG1) - BSA Free
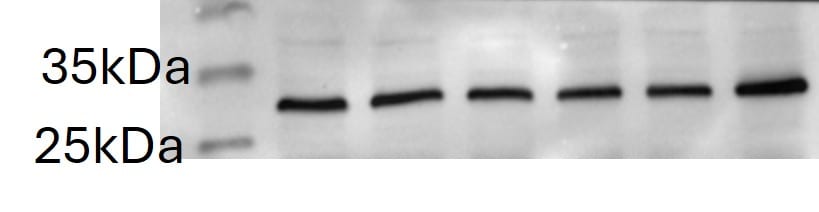
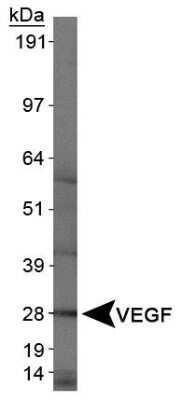
Western Blot Analysis of VEGF in Human Kidney
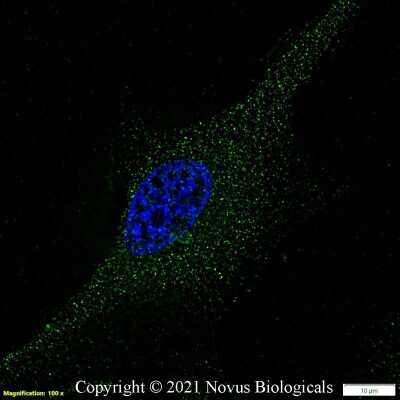
Analysis of VEGF in human kidney protein using NB100-664.Immunocytochemistry/Immunofluorescence Staining of VEGF in U87 Cells
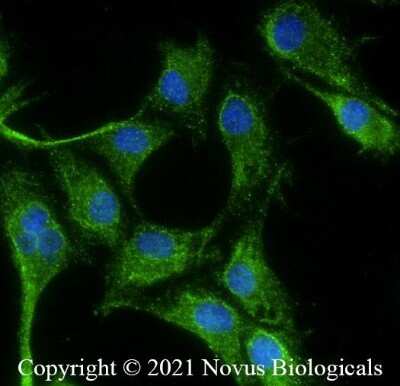
U87 cells were fixed in 4% paraformaldehyde for 10 minutes and permeabilized in 0.05% Triton X-100 in PBS for 5 minutes. The cells were incubated with anti- NB100-664 at 1 ug/ml overnight at 4C and detected with an anti-mouse Dylight 488 (Green) at a 1:1000 dilution for 60 minutes. Nuclei were counterstained with DAPI (Blue). Cells were imaged using a 40X objective.Immunohistochemical Analysis of VEGF in Peritoneal Tissue Sections
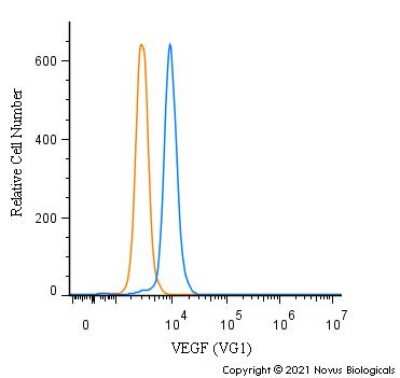
VEGF-Antibody-VG1-BSA-Free-Immunohistochemistry-NB100-664-img0015.jpgFlow Cytometry of U-937 Cells Stained with VEGF Antibody
An intracellular stain was performed on U-937 cells with NB100-664 (blue) and a matched isotype control (orange). Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 1.0 ug/mL for 30 minutes at room temperature, followed by Mouse IgG (H+L) Cross-Adsorbed Secondary Antibody, Dylight 550 (35503, Thermo Fisher).Western Blot Detection of VEGF in Transfected ADMSCs
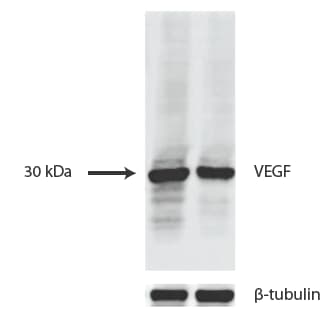
VEGF-Antibody-VG1-BSA-Free-Western-Blot-NB100-664-img0019.jpgWestern Blot Analysis of VEGF in Transfected Cell Lines
HUVEC and 293T cells transfected with a plasmid expressing human VEGF165 at 1:1000. Image provided by verified customer review.Immunocytochemistry/Immunofluorescence Staining of VEGF in U87 Cells
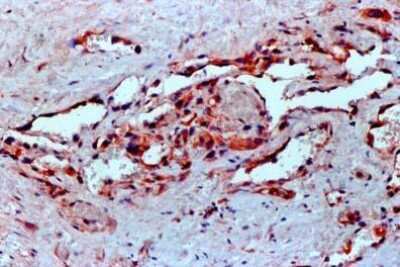
U-87 cells were fixed in 4% paraformaldehyde for 10 minutes and permeabilized in 0.05% Triton X-100 in PBS for 5 minutes. The cells were incubated with anti- NB100-664 at 2 ug/ml overnight at 4C and detected with an anti-mouse Dylight 488 (Green) at a 1:1000 dilution for 60 minutes. Nuclei were counterstained with DAPI (Blue). Cells were imaged using a 100X objective and digitally deconvolved.Immunohistochemical Analysis of VEGF in Paraffin Embedded Human Angiosarcoma Tissue
FFPE human angiosarcoma tissue section using VEGF antibody (clone VG1). The endothelial cells of the blood vessels and most of the cancer cells showed strong positivity for VEGF protein.Flow Cytometry of HUVEC Cells Stained with PerCP Conjugated VEGF Antibody
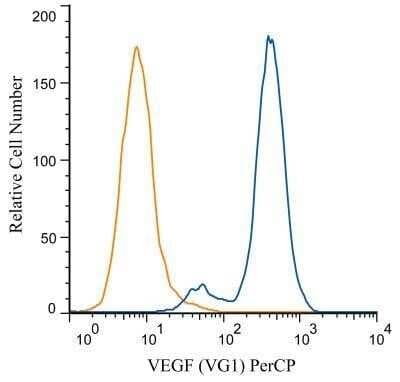
Analysis of PerCP conjugate of NB100-664. An intracellular stain was performed on HUVEC cells with VEGF (VG1) antibody NB100-664PCP (blue) and a matched isotype control NBP2-27287PCP (orange). Cells were fixed with 4% PFA and then permeablized with 0.1% saponin. Cells were incubated in an antibody dilution of 10 ug/mL for 30 minutes at room temperature. Both antibodies were conjugated to PerCP.Flow Cytometry of U-937 Cells Stained with FITC Conjugated VEGF Antibody
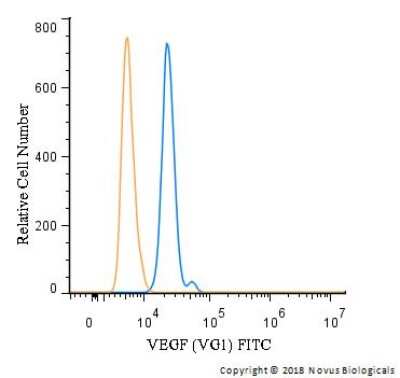
An intracellular stain was performed on U-937 cells with NB100-664F (blue) and a matched isotype control (orange). Cells were fixed with 4% PFA and then permeablized with 0.1% saponin. Cells were incubated in an antibody dilution of 10 ug/mL for 30 minutes at room temperature. Both antibodies were conjugated to FITC.Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] -
Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - Regorafenib reduced TNBC cell migration & targeted p-STAT3/VEGF-A signaling.(A) Chemical structure of regorafenib. (B,C), Transwell assay (B) & wound-healing assay (C) were performed in TNBC cell lines after regorafenib treatment for 24 h. *p < 0.05, **p < 0.01. (D) dose-dependent effects of regorafenib on p-STAT3 & VEGF-A proteins were analyzed by western blot. (E) Dose-dependent effects of regorafenib on VEGF-A mRNA were analyzed by qPCR. TNBC cells were exposed to the indicated doses for 24 hours. *p < 0.05, **p < 0.01. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27364975), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] -
Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - SC-78, a derivative of regorafenib, exhibited more potent anti-migratory effects than regorafenib.(A) Chemical structure of SC-78. (B) The protein levels of p-VEGFR2, VEGFR2, p-PDGFR, & PDGFR in EAhy926 treated with various doses of regorafenib or SC-78 were determined by western blot. (C,D), The effect of SC-78 on migration of human TNBC cells was analyzed by Transwell migration assay (C) & wound-healing assay (D). *p < 0.05, **p < 0.01. (E) Human TNBC cells were treated with SC-78 dose dependently for 24 h, & the cell lysates were subjected to western blot assay. (F) MDA-MB-231 cells were transfected with vector control or VEGF-A overexpression plasmid (left), STAT3 overexpression plasmid (middle), scramble or SHP-1 siRNA (right), respectively. After 48 h transfection, cells were treated with SC-78 (2.5 μM) for 24 h & subjected to western blot assay or cells analyzed by Transwell migration assay. *p < 0.05, **p < 0.01. (G) SHP-1 activity assay was performed in MDA-MB-231 & MDA-MB-468 cells treated with SC-78 dose dependently. *p < 0.05, **p < 0.01. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27364975), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] -
Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - Anti-tumor activity of regorafenib & SC-78 in a murine TNBC metastasis model.(A) Nude mice were subcutaneously injected with MDA-MB-468 cells (2 × 106). Mice were treated with vehicle, bevacizumab (125 μg/100 μl, twice a week, IP), regorafenib, or SC-78 (10 mg/kg, everyday, oral) for 43 days (n = 6), & the tumor size (left, bottom) was measured. In vivo protein levels were analyzed (Middle, top). Middle, bottom, the in vivo SHP-1 activity. Right, Representative images of IHC staining (100 × ). *p < 0.05, **p < 0.01. (B) Luciferase-expressing MDA-MB-231 (1 × 106) cells were injected orthotopically into the mammary fat pad of the mice. After two weeks, mice received regorafenib & SC-78, or vehicle orally at 10 mg/kg/every day (n = 5). Tumor growth was monitored by IVIS imaging system at the indicated times. Left, top, visualized by IVIS analysis. Left, bottom, quantification analysis from the IVIS total flux. Right, Representative images of IHC staining (100×). *p < 0.05, **p < 0.01. (C) In vivo bioluminescence images of nude mice injected i.v. with MDA-MB-231/Luc2 cells (1 × 106). After bioluminescence was observed, mice received vehicle or SC-78 orally at 10 mg/kg/every day. left, visualized by IVIS analysis. Right, Kaplan–Meier plot showing animal survival after treatment with vehicle or SC-78 (n = 8). The survival endpoint was set at 64 days after drug administration. (D) Schematic displays the drug mechanism of regorafenib & SC-78 on VEGF-A autocrine & paracrine inhibition & cell migration. SC-78 suppressed cancer metastasis dominantly through SHP-1 dependent-STAT3 dephosphorylation. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27364975), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] -
Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - Regorafenib transcriptionally inhibited VEGF-A expression through decreasing binding of STAT3 on promoter of VEGF-A.(A) Top, MDA-MB-231 cells co-transfected w/ a Renilla control vector & plasmids containing firefly luciferase gene driven by wild-type or STAT3 binding site-mutated VEGF-A promoter. After transfection for 48 h, cells treated w/ regorafenib for 24 h. Promoter activity analyzed by luciferase assay after regorafenib treatment. Bottom, MDA-MB-231 cells transfected w/ vector-control or STAT3-overexpression plasmid for 48 h. After, cells further co-transfected w/ Renilla & wild-type VEGF-A promoter & detect promoter activity as mentioned above. *p < 0.05, **p < 0.01. (B) After regorafenib treatment for 24 h, STAT3 binding site fragment detected by PCR in ChIP samples precipitated w/ STAT3 & rabbit IgG control antibodies in MDA-MB-231 cells. (C,D), MDA-MB-231 cells transfected, respectively, w/ control vector or VEGF-A overexpression plasmid (C) or STAT3 overexpression plasmid (D) for 48 h. After transfection, cells treated w/wo regorafenib for 24 h & subjected to WB assay or seeded to Transwell to analyze migration ability. *p < 0.05, **p < 0.01. (E) Cells treated w/ regorafenib at indicated dosages for 24 h & cell lysates analyzed by SHP-1 phosphatase activity assay. *p < 0.05, **p < 0.01. (F) MDA-MB-231 cells pretreated w/ pan-phosphatase inhibitor (left), or specific SHP-1 inhibitor (Middle) for 1 h before regorafenib treatment. Right, MDA-MB-231 cells transfected, respectively, w/ control siRNA or SHP-1 siRNA for 48 h. After transfection, cells treated w/wo regorafenib (10 μM) for 24 h. The protein levels analyzed by WB assay or cells seeded to Transwell to analyze migration ability. *p < 0.05, **p < 0.01. Image collected & cropped by CiteAb from following publication (https://pubmed.ncbi.nlm.nih.gov/27364975), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] -
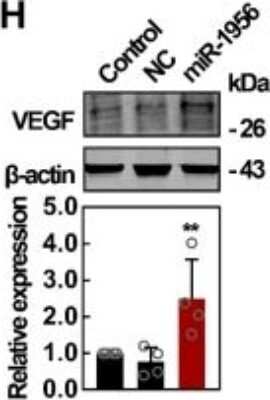
Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - Notch-1 is identified as an intermediate molecule in the miR-1956-VEGF axis. (A) Notch-1 is a target gene for miR-1956. The sequence of miR-1956, the potential binding site at the 3' UTR of Notch-1 mRNA, & the nucleotides mutated in the Notch-1-3' UTR mutant are shown. HEK293 cells were transiently cotransfected with miR-1956 mimic or NC using luciferase reporter vectors. The luciferase activity was normalized to the activity of Renilla luciferase. Data are shown as mean ± SD from three independent experiments. *, significantly different from NC; **, p < 0.01. After transfection with 50 nmol/L of miR-1956 mimic for 3 days, the mRNA level of Notch-1 was determined using qRT-PCR (B), & the protein expression of Notch-1 was measured using western blotting (C). For B, data are shown as mean ± SD from three independent experiments. *, significantly different from control; **, p < 0.01. For C, representative images of two independent experiments are shown. Data are shown as mean ± SD. *, significantly different from control; **, p < 0.01. ADMSCs were transfected with 2.5 μg Notch-1 plasmid & 50 nmol/L miR-1956 mimic or 100 nmol/L miR-1956 inhibitor for 3 days, the intracellular expression of Notch-1 (D) & VEGF (E) was detected by western blotting. For D & E, representative images of four independent experiments are shown. Data are shown as mean ± SD. *, significantly different from the control group transfected with control plasmid & NC mimic or inhibitor; *, p < 0.01; **, p < 0.01. #, significantly different from the group transfected with Notch-1 plasmid & NC mimic or inhibitor; #, p < 0.05. One-way ANOVA followed by Tukey's post-test was performed (A-E). Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31938051), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] -
Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - SHP-1 is negatively associated with p-STAT3/VEGF-A signaling & metastasis in TNBC cells & clinical samples.(A) Protein expression pattern of SHP-1, p-STAT3, & VEGF-A in nine TNBC cell lines were analyzed by western blot. (B) The migration abilities of nine TNBC cell lines were analyzed by Transwell assay. DAPI stains the nuclei. BT-20 cells were used as a normalization control. (C) The correlation (linear regression model) of VEGF-A (top), p-STAT3 (middle), & SHP-1 (bottom) & migration ability in nine TNBC cell lines. (D) TNBC cells from representative two patients with SHP-1, p-STAT3, & VEGF-A staining. (200×) (E) Kaplan-Meier graph was prepared to compare DMFS (left) & DFS (right) in patients with high VEGF-A (H score > 160) or low VEGF-A (H score < = 160) levels for the indicated time of follow up. Chi-square test indicated a significant difference between VEGF-A high (N = 21) & low (N = 76) patients. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/27364975), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] -
Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - Regorafenib transcriptionally inhibited VEGF-A expression through decreasing binding of STAT3 on promoter of VEGF-A.(A) Top, MDA-MB-231 cells co-transfected w/ a Renilla control vector & plasmids containing firefly luciferase gene driven by wild-type or STAT3 binding site-mutated VEGF-A promoter. After transfection for 48 h, cells treated w/ regorafenib for 24 h. Promoter activity analyzed by luciferase assay after regorafenib treatment. Bottom, MDA-MB-231 cells transfected w/ vector-control or STAT3-overexpression plasmid for 48 h. After, cells further co-transfected w/ Renilla & wild-type VEGF-A promoter & detect promoter activity as mentioned above. *p < 0.05, **p < 0.01. (B) After regorafenib treatment for 24 h, STAT3 binding site fragment detected by PCR in ChIP samples precipitated w/ STAT3 & rabbit IgG control antibodies in MDA-MB-231 cells. (C,D), MDA-MB-231 cells transfected, respectively, w/ control vector or VEGF-A overexpression plasmid (C) or STAT3 overexpression plasmid (D) for 48 h. After transfection, cells treated w/wo regorafenib for 24 h & subjected to WB assay or seeded to Transwell to analyze migration ability. *p < 0.05, **p < 0.01. (E) Cells treated w/ regorafenib at indicated dosages for 24 h & cell lysates analyzed by SHP-1 phosphatase activity assay. *p < 0.05, **p < 0.01. (F) MDA-MB-231 cells pretreated w/ pan-phosphatase inhibitor (left), or specific SHP-1 inhibitor (Middle) for 1 h before regorafenib treatment. Right, MDA-MB-231 cells transfected, respectively, w/ control siRNA or SHP-1 siRNA for 48 h. After transfection, cells treated w/wo regorafenib (10 μM) for 24 h. The protein levels analyzed by WB assay or cells seeded to Transwell to analyze migration ability. *p < 0.05, **p < 0.01. Image collected & cropped by CiteAb from following publication (https://pubmed.ncbi.nlm.nih.gov/27364975), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: Mouse Monoclonal VEGF Antibody (VG1) [NB100-664]
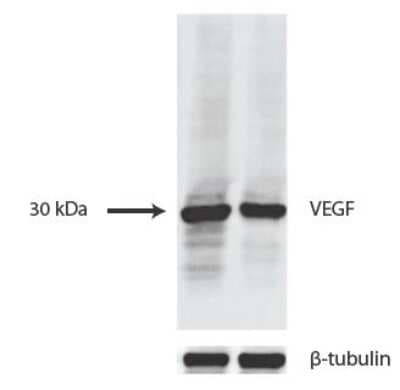
VEGF expression in mouse liver tissues. Dilution: 2 ug/ml in 1X TBST. Image from a verified customer review.Immunohistochemical Analysis of VEGF in Paraffin Embedded Human Liver
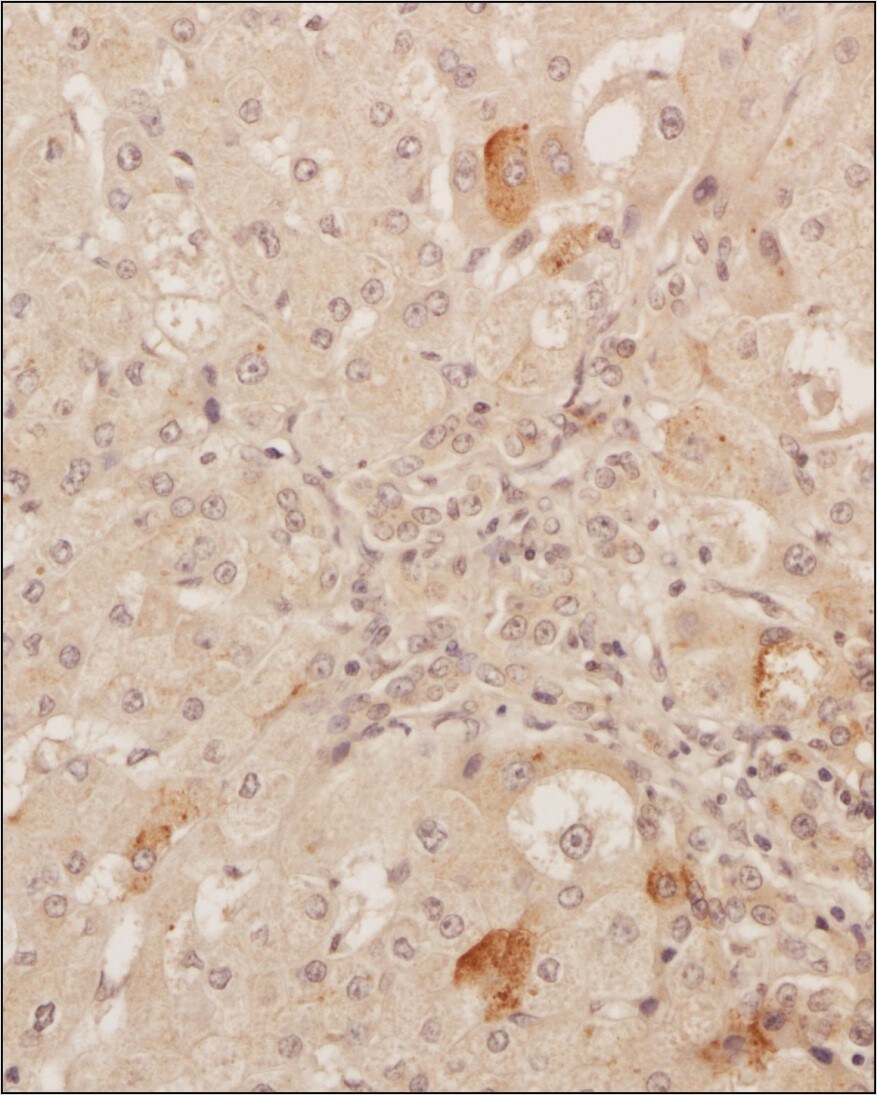
Analysis of a FFPE tissue section of human liver using 1:200 dilution of VEGF antibody [VG1]. The staining was developed using HRP labeled anti-rabbit secondary antibody and DAB reagent, and nuclei of cells were counter-stained with hematoxylin.Immunohistochemical Analysis of VEGF in Paraffin Embedded Mouse Liver
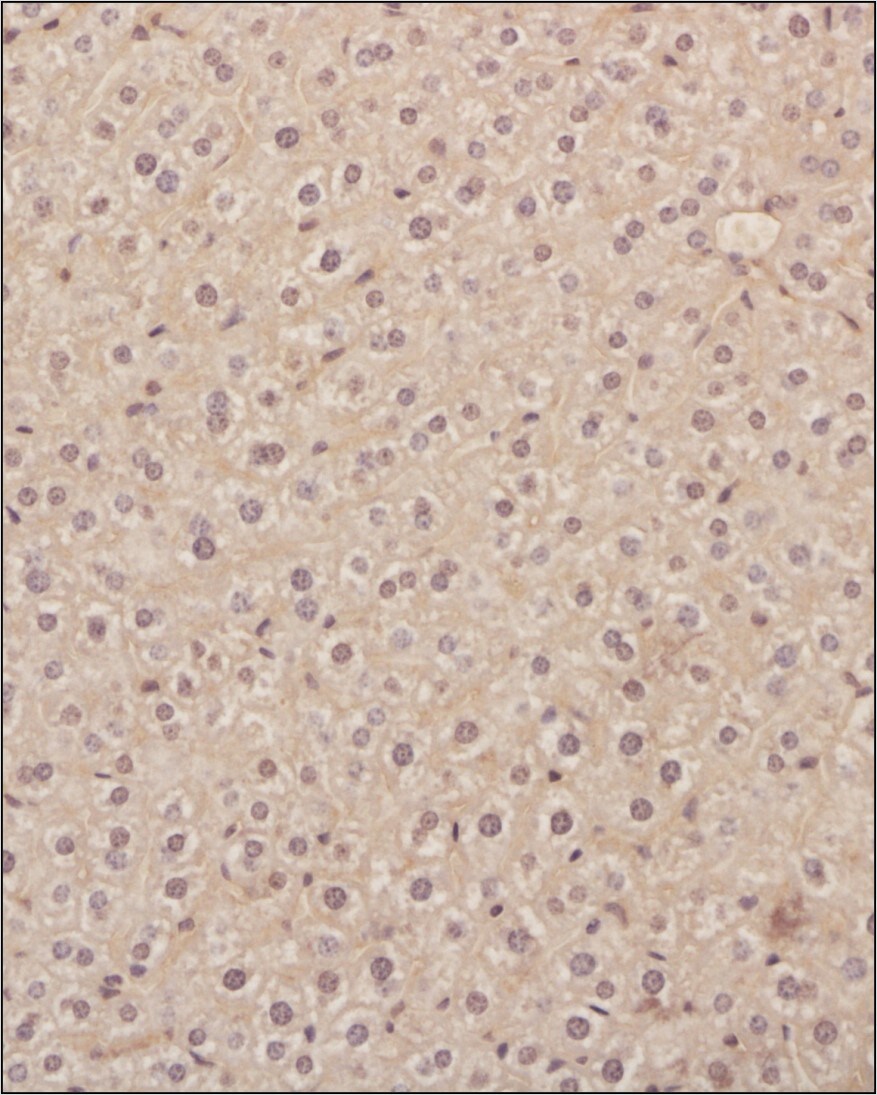
Analysis of a FFPE tissue section of mouse liver using 1:200 dilution of VEGF antibody [VG1]. The staining was developed using HRP labeled anti-rabbit secondary antibody and DAB reagent, and nuclei of cells were counter-stained with hematoxylin.Immunohistochemistry: VEGF Antibody (VG1) - BSA Free [NB100-664] -
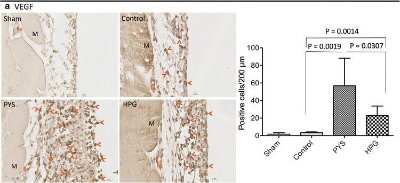
HPG induces less VEGF production, less myofibroblast differentiation and lower macrophage activation. The expression of VEGF, alpha -SMA and MAC387 in the peritoneal tissue sections was examined using a routine immunohistochemical method. Data were a typical microscopic view of the peritoneal tissue sections in each group. a VEGF was detected using mouse monoclonal anti-VEGF antibody from Novus. Left graph a typical microscopic view, Dark brown stain VEGF-expressing cells (pointed by red arrows), Bv blood vessels, M muscle, black small bar 10 um. Right graph VEGF-expressing cells per 200 μm PM length in cross sections. Data were presented as mean +/- SD (n = 6) and were analyzed using t test. b alpha -SMA (a myofibroblast marker) was detected using mouse monoclonal anti-alpha -SMA antibody from Sigma-Aldrich. Left graph a typical microscopic view, Dark brown stain alpha -SMA-expressing cells or myofibroblasts (pointed by red arrows), Bv blood vessels, M muscle, black small bar 10 um. Right graph alpha -SMA-expressing cells per 200 μm PM length in cross sections. Data were presented as mean +/- SD (n = 6) and were analyzed using t test. c Macrophages were detected using mouse monoclonal anti-MAC387 antibody from Santa Cruz Biotech. Left graph a typical microscopic view, Dark brown stain MAC387-expressing cells or macrophages (pointed by red arrows), M muscle, black small bar 10 um. Right graph MAC387-expressing cells per 200 μm PM length in cross sections. Data were presented as mean +/- SD (n = 6) and were analyzed using t test Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/27964722), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: VEGF Antibody (VG1) - BSA Free [NB100-664] -
Comparison of Col I and VEGF expression. (A) Representative images of Col I and VEGF immunohistochemical staining (30× magnification). Col I was stained with a reddish color. The black arrows indicate the location of VEGF staining. (B) Quantification of Col I and VEGF expression by immunohistochemical staining (mean +/- SD). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37151660), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for VEGF Antibody (VG1) - BSA Free
Flow (Intracellular)
Flow Cytometry
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Frozen
Immunohistochemistry-Paraffin
Western Blot
See Simple Western Antibody Database for Simple Western validation: tested in mouse aortas and HUVEC lysate; separated by charge; detects a band at 30 kDa
Reviewed Applications
Read 2 reviews rated 5 using NB100-664 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: VEGF
References
1. Melincovici CS, Bosca AB, susman S, et al. Vascular endothelial growth factor (VEGF) - key factor in normal and pathological angiogenesis. Rom J Morphol Embryol. 2018;59(2):455-467.
2. Shaik F, Cuthbert GA, Homer-Vanniasinkam S, Muench SP, Ponnambalam S, Harrison MA. Structural Basis for Vascular Endothelial Growth Factor Receptor Activation and Implications for Disease Therapy. Biomolecules. 2020;10(12):1673. https://doi.org/10.3390/biom10121673
3. Apte RS, Chen DS, Ferrara N. VEGF in Signaling and Disease: Beyond Discovery and Development. Cell. 2019;176(6):1248-1264. https://doi.org/10.1016/j.cell.2019.01.021
4. Matsumoto K, Ema M. Roles of VEGF-A signalling in development, regeneration, and tumours. J Biochem. 2014;156(1):1-10. https://doi.org/10.1093/jb/mvu031
5. Itatani Y, Kawada K, Yamamoto T, Sakai Y. Resistance to Anti-Angiogenic Therapy in Cancer-Alterations to Anti-VEGF Pathway. Int J Mol Sci. 2018;19(4):1232. Published 2018 Apr 18. doi:10.3390/ijms19041232
6. Uniprot (P15692)
7. Hamilton JL, Nagao M, Levine BR, Chen D, Olsen BR, Im HJ. Targeting VEGF and Its Receptors for the Treatment of Osteoarthritis and Associated Pain. J Bone Miner Res. 2016;31(5):911-924. https://doi.org/10.1002/jbmr.2828
Long Name
Alternate Names
Gene Symbol
UniProt
Additional VEGF Products
Product Documents for VEGF Antibody (VG1) - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for VEGF Antibody (VG1) - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for VEGF Antibody (VG1) - BSA Free
Customer Reviews for VEGF Antibody (VG1) - BSA Free (2)
Have you used VEGF Antibody (VG1) - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Western BlotSample Tested: LiverSpecies: MouseVerified Customer | Posted 10/21/2025VEGF expression in liver tissuesDilution: 2 ug/ml in 1X TBST
-
Application: Western BlotSample Tested: HUVEC and 293T cellsSpecies: HumanVerified Customer | Posted 02/28/2014Western blot analysis of VEGF expression in HUVEC cells (A), and 293T cells transfected with a plasmid expressing human VEGF165
There are no reviews that match your criteria.
Protocols
View specific protocols for VEGF Antibody (VG1) - BSA Free (NB100-664):
Sample Preparation.
1. Grow cells to 60-85% confluency. Flow cytometry requires between 2 x 105 and 1 x 106 cells for optimal performance.
2. If cells are adherent, harvest gently by washing once with staining buffer and then scraping. Avoid using trypsin as this can disrupt certain epitopes of interest. If enzymatic harvest is required, use Accutase, Collagenase, or TrypLE Express for a less damaging option.
3. Reserve 100 uL for counting, then transfer cell volume into a 50 mL conical tube and centrifuge for 8 minutes at 400 RCF.
a. Count cells using a hemocytometer and a 1:1 trypan blue exclusion stain to determine cell viability before starting the flow protocol. If cells appear blue, do not proceed.
4. Re-suspend cells to a concentration of 1 x 106 cells/mL in staining buffer (NBP2-26247).
5. Aliquot out 1 mL samples in accordance with your experimental samples.
Tip: When cell surface and intracellular staining are required in the same sample, it is advisable that the cell surface staining be performed first since the fixation and permeablization steps might reduce the availability of surface antigens.
Intracellular Staining.
Tip: When performing intracellular staining, it is important to use appropriate fixation and permeabilization reagents based upon the target and its subcellular location. Generally, our Intracellular Flow Assay Kit (NBP2-29450) is a good place to start as it contains an optimized combination of reagents for intracellular staining as well as an inhibitor of intracellular protein transport (necessary if staining secreted proteins). Certain targets may require more gentle or transient permeabilization protocols such as the commonly employed methanol or saponin-based methods.
Protocol for Cytoplasmic Targets:
Optional: Perform cell surface staining as described in the previous section.
1. Fix the cells by adding 100 uL fixation solution (such as 4% PFA) to each sample for 10-15 minutes.
2. Permeabilize cells by adding 100 uL of a permeabization buffer to every 1 x 106 cells present in the sample. Mix well and incubate at room temperature for 15 minutes.
a. For cytoplasmic targets, use a gentle permeabilization solution such as 1X PBS + 0.5% Saponin or 1X PBS + 0.5% Tween-20.
b. To maintain the permeabilized state throughout your experiment, use staining buffer + 0.1% of the permeabilization reagent (i.e. 0.1% Tween-20 or 0.1% Saponin).
3. Following the 15 minute incubation, add 2 mL of the staining buffer + 0.1% permeabilizer to each sample.
4. Centrifuge for 5 minutes at 400 RCF.
5. Discard supernatant and re-suspend in 1 mL of staining buffer + 0.1% permeabilizer.
6. Stain each sample at 1 uL/ 1 x 106 cells of primary antibody or 1-3 uL/ 1 x 106 cells for directly conjugated antibodies. Mix well and incubate at room temperature for 30 minutes- 1 hour. Gently mix samples every 10-15 minutes.
7. Following the primary/conjugate incubation, add 2 mL/sample of staining buffer +0.1% permeabilizer and centrifuge for 5 minutes at 400 RCF.
8. Remove supernatant and re-suspend each sample in 2 mL staining buffer + 0.1% permeabilizer, repeat wash for 5 minutes at 400 RCF.
9. If using a directly conjugated antibody, after the second wash, re-suspend cell pellet to a final volume of 500 uL per sample and proceed with flow analysis.
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and wash the cells briefly in PBS. Add 10% formalin to the dish and fix at room temperature for 10 minutes.
2. Remove the formalin and wash the cells in PBS.
3. Permeablize the cells with 0.1% Triton X100 or other suitable detergent for 10 min.
4. Remove the permeablization buffer and wash three times for 10 minutes each in PBS. Be sure to not let the specimen dry out.
5. To block nonspecific antibody binding, incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
6. Add primary antibody at appropriate dilution and incubate overnight at 4C.
7. Remove primary antibody and replace with PBS. Wash three times for 10 minutes each.
8. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
9. Remove secondary antibody and replace with PBS. Wash three times for 10 minutes each.
10. Counter stain DNA with DAPi if required.
Antigen Unmasking:
Bring slides to a boil in 10 mM sodium citrate buffer (pH 6.0) then maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench-top for 30 minutes (keep slides in the sodium citrate buffer at all times).
Staining:
1. Wash sections in deionized water three times for 5 minutes each.
2. Wash sections in PBS for 5 minutes.
3. Block each section with 100-400 ul blocking solution (1% BSA in PBS) for 1 hour at room temperature.
4. Remove blocking solution and add 100-400 ul diluted primary antibody. Incubate overnight at 4 C.
5. Remove antibody solution and wash sections in wash buffer three times for 5 minutes each.
6. Add 100-400 ul HRP polymer conjugated secondary antibody. Incubate 30 minutes at room temperature.
7. Wash sections three times in wash buffer for 5 minutes each.
8. Add 100-400 ul DAB substrate to each section and monitor staining closely.
9. As soon as the sections develop, immerse slides in deionized water.
10. Counterstain sections in hematoxylin.
11. Wash sections in deionized water two times for 5 minutes each.
12. Dehydrate sections.
13. Mount coverslips.
1. Perform SDS-PAGE on samples to be analyzed, loading 10-25 ug of total protein per lane.
2. Transfer proteins to PVDF membrane according to the instructions provided by the manufacturer of the membrane and transfer apparatus.
3. Stain the membrane with Ponceau S (or similar product) to assess transfer success, and mark molecular weight standards where appropriate.
4. Rinse the blot TBS -0.05% Tween 20 (TBST).
5. Block the membrane in 5% Non-fat milk in TBST (blocking buffer) for at least 1 hour.
6. Wash the membrane in TBST three times for 10 minutes each.
7. Dilute primary antibody in blocking buffer and incubate overnight at 4C with gentle rocking.
8. Wash the membrane in TBST three times for 10 minutes each.
9. Incubate the membrane in diluted HRP conjugated secondary antibody in blocking buffer (as per manufacturer's instructions) for 1 hour at room temperature.
10. Wash the blot in TBST three times for 10 minutes each (this step can be repeated as required to reduce background).
11. Apply the detection reagent of choice in accordance with the manufacturer's instructions.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- R&D Systems Quality Control Western Blot Protocol
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: ELISA
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for VEGF Antibody (VG1) - BSA Free
-
Q: I am interested in buying an antibody to VEGF to detect VEGF in human umbilical vein endothelial cells for western blotting and was wondering if you might have any recommendations? I was thinking about NB100-664. Do you have a recommended amount to use for western blotting? Could I ask you what concentration the antibody is at since it says 0.1mg so what total volume do you send? Also is this the most commonly purchase one for western blotting do you know?
A: We do not have a recommended dilution noted for this specific antibody, but our general recommendation for Western blot is 1:1000-1:2000.This is a good starting point, but we always recommend optimization by the end user, as the optimal dilution is to some extent dependent on the particular sample/assay. The concentration for this product is stated as 1 mg/ml, so 0.1 mg would be supplied in 0.1 ml.
-
Q: I am looking for a VEGF-A antibody for the IHC staining of mouse colon. Do you have any publications or application references for this product?
A: NB100-664 is one of our best sellers with great customer feedback and citations in at least 18 peer reviewed publications from highly esteemed researchers. As far as mouse tissues are concerned, I would recommend that you go through the following paper: Milanesi A, et al. Beta-Cell regeneration mediated by human bone marrow mesenchymal stem cells. PLoS One. 2012;7(8):e42177. doi: 10.1371/journal.pone.0042177. Epub 2012 Aug 7. PMID:22879915.
-
Q: I would be very grateful if you could let me know a recommended dilution for the VEGF antibody antibody NB100-664
A: The recommended dilution for IHC is 1:20-1:50 in an ABC method. If instead you are testing on cell culture, I would recommend a starting optimization point of 1:10 -1:2000.
-
Q: I would like to know the aproximate dilution of the VEGF Antibody (NB100-664) to use it in a Western blot assay.
A: In regards to your inquiry about our VEGF antibody (NB100-664), I am showing from the lab data that they have tested dilutions in the range of 1:500-1:1000 for this antibody. The optimal dilution should depend on your samples and the level of expression of VEGF, but a good starting point would be 1:1000 and then optimize from there.
-
Q: What is the heparin binding activity of FGF and VEGF?
A: These proteins are not assayed for their ability to bind Heparin. More information about the FGF family of growth factors is available in this review article: Basilico, C. (1992) Adv. Can. Res. 59:115.
-
Q: What is the minimum amount of protein necessary for the detection of VEGF by Western blot using VEGF Antibody (NB100-664)?
A: I would suggest at minimum 10ug of protein per well but if you have enough sample you might want to run multiple wells with increasing amounts of protein to optimize the conditions in your hands. You could start with 5ug and do a couple of wells with 10ug, 20ug and 40ug. Also try using different dilutions of primary, start with 1:500-1:1000 and work your way up or down depending on what your blot looks like.
-
Q: Why is the molecular weight of VEGF different from the similar antibody, for some companies the the molecular weight is 40KD)?
A: I can't comment on another company's antibody because I don't have any information about their products. I can tell you that VEGF is expressed in a variety of isoforms and is subject to various post-translational modifications that influence its apparent molecular weight in an SDS-PAGE gel compared to the theoretical molecular weight.
-
Q: I am interested in buying an antibody to VEGF to detect VEGF in human umbilical vein endothelial cells for western blotting and was wondering if you might have any recommendations? I was thinking about NB100-664. Do you have a recommended amount to use for western blotting? Could I ask you what concentration the antibody is at since it says 0.1mg so what total volume do you send? Also is this the most commonly purchase one for western blotting do you know?
A: We do not have a recommended dilution noted for this specific antibody, but our general recommendation for Western blot is 1:1000-1:2000.This is a good starting point, but we always recommend optimization by the end user, as the optimal dilution is to some extent dependent on the particular sample/assay. The concentration for this product is stated as 1 mg/ml, so 0.1 mg would be supplied in 0.1 ml.
-
Q: I am looking for a VEGF-A antibody for the IHC staining of mouse colon. Do you have any publications or application references for this product?
A: NB100-664 is one of our best sellers with great customer feedback and citations in at least 18 peer reviewed publications from highly esteemed researchers. As far as mouse tissues are concerned, I would recommend that you go through the following paper: Milanesi A, et al. Beta-Cell regeneration mediated by human bone marrow mesenchymal stem cells. PLoS One. 2012;7(8):e42177. doi: 10.1371/journal.pone.0042177. Epub 2012 Aug 7. PMID:22879915.
-
Q: I would be very grateful if you could let me know a recommended dilution for the VEGF antibody antibody NB100-664
A: The recommended dilution for IHC is 1:20-1:50 in an ABC method. If instead you are testing on cell culture, I would recommend a starting optimization point of 1:10 -1:2000.
-
Q: I would like to know the aproximate dilution of the VEGF Antibody (NB100-664) to use it in a Western blot assay.
A: In regards to your inquiry about our VEGF antibody (NB100-664), I am showing from the lab data that they have tested dilutions in the range of 1:500-1:1000 for this antibody. The optimal dilution should depend on your samples and the level of expression of VEGF, but a good starting point would be 1:1000 and then optimize from there.
-
Q: What is the heparin binding activity of FGF and VEGF?
A: These proteins are not assayed for their ability to bind Heparin. More information about the FGF family of growth factors is available in this review article: Basilico, C. (1992) Adv. Can. Res. 59:115.
-
Q: What is the minimum amount of protein necessary for the detection of VEGF by Western blot using VEGF Antibody (NB100-664)?
A: I would suggest at minimum 10ug of protein per well but if you have enough sample you might want to run multiple wells with increasing amounts of protein to optimize the conditions in your hands. You could start with 5ug and do a couple of wells with 10ug, 20ug and 40ug. Also try using different dilutions of primary, start with 1:500-1:1000 and work your way up or down depending on what your blot looks like.
-
Q: Why is the molecular weight of VEGF different from the similar antibody, for some companies the the molecular weight is 40KD)?
A: I can't comment on another company's antibody because I don't have any information about their products. I can tell you that VEGF is expressed in a variety of isoforms and is subject to various post-translational modifications that influence its apparent molecular weight in an SDS-PAGE gel compared to the theoretical molecular weight.
-
Q: I am interested in buying an antibody to VEGF to detect VEGF in human umbilical vein endothelial cells for western blotting and was wondering if you might have any recommendations? I was thinking about NB100-664. Do you have a recommended amount to use for western blotting? Could I ask you what concentration the antibody is at since it says 0.1mg so what total volume do you send? Also is this the most commonly purchase one for western blotting do you know?
A: We do not have a recommended dilution noted for this specific antibody, but our general recommendation for Western blot is 1:1000-1:2000.This is a good starting point, but we always recommend optimization by the end user, as the optimal dilution is to some extent dependent on the particular sample/assay. The concentration for this product is stated as 1 mg/ml, so 0.1 mg would be supplied in 0.1 ml.
-
Q: I am looking for a VEGF-A antibody for the IHC staining of mouse colon. Do you have any publications or application references for this product?
A: NB100-664 is one of our best sellers with great customer feedback and citations in at least 18 peer reviewed publications from highly esteemed researchers. As far as mouse tissues are concerned, I would recommend that you go through the following paper: Milanesi A, et al. Beta-Cell regeneration mediated by human bone marrow mesenchymal stem cells. PLoS One. 2012;7(8):e42177. doi: 10.1371/journal.pone.0042177. Epub 2012 Aug 7. PMID:22879915.
-
Q: I would be very grateful if you could let me know a recommended dilution for the VEGF antibody antibody NB100-664
A: The recommended dilution for IHC is 1:20-1:50 in an ABC method. If instead you are testing on cell culture, I would recommend a starting optimization point of 1:10 -1:2000.
-
Q: I would like to know the aproximate dilution of the VEGF Antibody (NB100-664) to use it in a Western blot assay.
A: In regards to your inquiry about our VEGF antibody (NB100-664), I am showing from the lab data that they have tested dilutions in the range of 1:500-1:1000 for this antibody. The optimal dilution should depend on your samples and the level of expression of VEGF, but a good starting point would be 1:1000 and then optimize from there.
-
Q: What is the heparin binding activity of FGF and VEGF?
A: These proteins are not assayed for their ability to bind Heparin. More information about the FGF family of growth factors is available in this review article: Basilico, C. (1992) Adv. Can. Res. 59:115.
-
Q: What is the minimum amount of protein necessary for the detection of VEGF by Western blot using VEGF Antibody (NB100-664)?
A: I would suggest at minimum 10ug of protein per well but if you have enough sample you might want to run multiple wells with increasing amounts of protein to optimize the conditions in your hands. You could start with 5ug and do a couple of wells with 10ug, 20ug and 40ug. Also try using different dilutions of primary, start with 1:500-1:1000 and work your way up or down depending on what your blot looks like.
-
Q: Why is the molecular weight of VEGF different from the similar antibody, for some companies the the molecular weight is 40KD)?
A: I can't comment on another company's antibody because I don't have any information about their products. I can tell you that VEGF is expressed in a variety of isoforms and is subject to various post-translational modifications that influence its apparent molecular weight in an SDS-PAGE gel compared to the theoretical molecular weight.
-
Q: I am interested in buying an antibody to VEGF to detect VEGF in human umbilical vein endothelial cells for western blotting and was wondering if you might have any recommendations? I was thinking about NB100-664. Do you have a recommended amount to use for western blotting? Could I ask you what concentration the antibody is at since it says 0.1mg so what total volume do you send? Also is this the most commonly purchase one for western blotting do you know?
A: We do not have a recommended dilution noted for this specific antibody, but our general recommendation for Western blot is 1:1000-1:2000.This is a good starting point, but we always recommend optimization by the end user, as the optimal dilution is to some extent dependent on the particular sample/assay. The concentration for this product is stated as 1 mg/ml, so 0.1 mg would be supplied in 0.1 ml.
-
Q: I am looking for a VEGF-A antibody for the IHC staining of mouse colon. Do you have any publications or application references for this product?
A: NB100-664 is one of our best sellers with great customer feedback and citations in at least 18 peer reviewed publications from highly esteemed researchers. As far as mouse tissues are concerned, I would recommend that you go through the following paper: Milanesi A, et al. Beta-Cell regeneration mediated by human bone marrow mesenchymal stem cells. PLoS One. 2012;7(8):e42177. doi: 10.1371/journal.pone.0042177. Epub 2012 Aug 7. PMID:22879915.
-
Q: I would be very grateful if you could let me know a recommended dilution for the VEGF antibody antibody NB100-664
A: The recommended dilution for IHC is 1:20-1:50 in an ABC method. If instead you are testing on cell culture, I would recommend a starting optimization point of 1:10 -1:2000.
-
Q: I would like to know the aproximate dilution of the VEGF Antibody (NB100-664) to use it in a Western blot assay.
A: In regards to your inquiry about our VEGF antibody (NB100-664), I am showing from the lab data that they have tested dilutions in the range of 1:500-1:1000 for this antibody. The optimal dilution should depend on your samples and the level of expression of VEGF, but a good starting point would be 1:1000 and then optimize from there.
-
Q: What is the heparin binding activity of FGF and VEGF?
A: These proteins are not assayed for their ability to bind Heparin. More information about the FGF family of growth factors is available in this review article: Basilico, C. (1992) Adv. Can. Res. 59:115.
-
Q: What is the minimum amount of protein necessary for the detection of VEGF by Western blot using VEGF Antibody (NB100-664)?
A: I would suggest at minimum 10ug of protein per well but if you have enough sample you might want to run multiple wells with increasing amounts of protein to optimize the conditions in your hands. You could start with 5ug and do a couple of wells with 10ug, 20ug and 40ug. Also try using different dilutions of primary, start with 1:500-1:1000 and work your way up or down depending on what your blot looks like.
-
Q: Why is the molecular weight of VEGF different from the similar antibody, for some companies the the molecular weight is 40KD)?
A: I can't comment on another company's antibody because I don't have any information about their products. I can tell you that VEGF is expressed in a variety of isoforms and is subject to various post-translational modifications that influence its apparent molecular weight in an SDS-PAGE gel compared to the theoretical molecular weight.
-
Q: I am interested in buying an antibody to VEGF to detect VEGF in human umbilical vein endothelial cells for western blotting and was wondering if you might have any recommendations? I was thinking about NB100-664. Do you have a recommended amount to use for western blotting? Could I ask you what concentration the antibody is at since it says 0.1mg so what total volume do you send? Also is this the most commonly purchase one for western blotting do you know?
A: We do not have a recommended dilution noted for this specific antibody, but our general recommendation for Western blot is 1:1000-1:2000.This is a good starting point, but we always recommend optimization by the end user, as the optimal dilution is to some extent dependent on the particular sample/assay. The concentration for this product is stated as 1 mg/ml, so 0.1 mg would be supplied in 0.1 ml.
-
Q: I am looking for a VEGF-A antibody for the IHC staining of mouse colon. Do you have any publications or application references for this product?
A: NB100-664 is one of our best sellers with great customer feedback and citations in at least 18 peer reviewed publications from highly esteemed researchers. As far as mouse tissues are concerned, I would recommend that you go through the following paper: Milanesi A, et al. Beta-Cell regeneration mediated by human bone marrow mesenchymal stem cells. PLoS One. 2012;7(8):e42177. doi: 10.1371/journal.pone.0042177. Epub 2012 Aug 7. PMID:22879915.
-
Q: I would be very grateful if you could let me know a recommended dilution for the VEGF antibody antibody NB100-664
A: The recommended dilution for IHC is 1:20-1:50 in an ABC method. If instead you are testing on cell culture, I would recommend a starting optimization point of 1:10 -1:2000.
-
Q: I would like to know the aproximate dilution of the VEGF Antibody (NB100-664) to use it in a Western blot assay.
A: In regards to your inquiry about our VEGF antibody (NB100-664), I am showing from the lab data that they have tested dilutions in the range of 1:500-1:1000 for this antibody. The optimal dilution should depend on your samples and the level of expression of VEGF, but a good starting point would be 1:1000 and then optimize from there.
-
Q: What is the heparin binding activity of FGF and VEGF?
A: These proteins are not assayed for their ability to bind Heparin. More information about the FGF family of growth factors is available in this review article: Basilico, C. (1992) Adv. Can. Res. 59:115.
-
Q: What is the minimum amount of protein necessary for the detection of VEGF by Western blot using VEGF Antibody (NB100-664)?
A: I would suggest at minimum 10ug of protein per well but if you have enough sample you might want to run multiple wells with increasing amounts of protein to optimize the conditions in your hands. You could start with 5ug and do a couple of wells with 10ug, 20ug and 40ug. Also try using different dilutions of primary, start with 1:500-1:1000 and work your way up or down depending on what your blot looks like.
-
Q: Why is the molecular weight of VEGF different from the similar antibody, for some companies the the molecular weight is 40KD)?
A: I can't comment on another company's antibody because I don't have any information about their products. I can tell you that VEGF is expressed in a variety of isoforms and is subject to various post-translational modifications that influence its apparent molecular weight in an SDS-PAGE gel compared to the theoretical molecular weight.
-
Q: I am interested in buying an antibody to VEGF to detect VEGF in human umbilical vein endothelial cells for western blotting and was wondering if you might have any recommendations? I was thinking about NB100-664. Do you have a recommended amount to use for western blotting? Could I ask you what concentration the antibody is at since it says 0.1mg so what total volume do you send? Also is this the most commonly purchase one for western blotting do you know?
A: We do not have a recommended dilution noted for this specific antibody, but our general recommendation for Western blot is 1:1000-1:2000.This is a good starting point, but we always recommend optimization by the end user, as the optimal dilution is to some extent dependent on the particular sample/assay. The concentration for this product is stated as 1 mg/ml, so 0.1 mg would be supplied in 0.1 ml.
-
Q: I am looking for a VEGF-A antibody for the IHC staining of mouse colon. Do you have any publications or application references for this product?
A: NB100-664 is one of our best sellers with great customer feedback and citations in at least 18 peer reviewed publications from highly esteemed researchers. As far as mouse tissues are concerned, I would recommend that you go through the following paper: Milanesi A, et al. Beta-Cell regeneration mediated by human bone marrow mesenchymal stem cells. PLoS One. 2012;7(8):e42177. doi: 10.1371/journal.pone.0042177. Epub 2012 Aug 7. PMID:22879915.
-
Q: I would be very grateful if you could let me know a recommended dilution for the VEGF antibody antibody NB100-664
A: The recommended dilution for IHC is 1:20-1:50 in an ABC method. If instead you are testing on cell culture, I would recommend a starting optimization point of 1:10 -1:2000.
-
Q: I would like to know the aproximate dilution of the VEGF Antibody (NB100-664) to use it in a Western blot assay.
A: In regards to your inquiry about our VEGF antibody (NB100-664), I am showing from the lab data that they have tested dilutions in the range of 1:500-1:1000 for this antibody. The optimal dilution should depend on your samples and the level of expression of VEGF, but a good starting point would be 1:1000 and then optimize from there.
-
Q: What is the heparin binding activity of FGF and VEGF?
A: These proteins are not assayed for their ability to bind Heparin. More information about the FGF family of growth factors is available in this review article: Basilico, C. (1992) Adv. Can. Res. 59:115.
-
Q: What is the minimum amount of protein necessary for the detection of VEGF by Western blot using VEGF Antibody (NB100-664)?
A: I would suggest at minimum 10ug of protein per well but if you have enough sample you might want to run multiple wells with increasing amounts of protein to optimize the conditions in your hands. You could start with 5ug and do a couple of wells with 10ug, 20ug and 40ug. Also try using different dilutions of primary, start with 1:500-1:1000 and work your way up or down depending on what your blot looks like.
-
Q: Why is the molecular weight of VEGF different from the similar antibody, for some companies the the molecular weight is 40KD)?
A: I can't comment on another company's antibody because I don't have any information about their products. I can tell you that VEGF is expressed in a variety of isoforms and is subject to various post-translational modifications that influence its apparent molecular weight in an SDS-PAGE gel compared to the theoretical molecular weight.
-
Q: I am interested in buying an antibody to VEGF to detect VEGF in human umbilical vein endothelial cells for western blotting and was wondering if you might have any recommendations? I was thinking about NB100-664. Do you have a recommended amount to use for western blotting? Could I ask you what concentration the antibody is at since it says 0.1mg so what total volume do you send? Also is this the most commonly purchase one for western blotting do you know?
A: We do not have a recommended dilution noted for this specific antibody, but our general recommendation for Western blot is 1:1000-1:2000.This is a good starting point, but we always recommend optimization by the end user, as the optimal dilution is to some extent dependent on the particular sample/assay. The concentration for this product is stated as 1 mg/ml, so 0.1 mg would be supplied in 0.1 ml.
-
Q: I am looking for a VEGF-A antibody for the IHC staining of mouse colon. Do you have any publications or application references for this product?
A: NB100-664 is one of our best sellers with great customer feedback and citations in at least 18 peer reviewed publications from highly esteemed researchers. As far as mouse tissues are concerned, I would recommend that you go through the following paper: Milanesi A, et al. Beta-Cell regeneration mediated by human bone marrow mesenchymal stem cells. PLoS One. 2012;7(8):e42177. doi: 10.1371/journal.pone.0042177. Epub 2012 Aug 7. PMID:22879915.
-
Q: I would be very grateful if you could let me know a recommended dilution for the VEGF antibody antibody NB100-664
A: The recommended dilution for IHC is 1:20-1:50 in an ABC method. If instead you are testing on cell culture, I would recommend a starting optimization point of 1:10 -1:2000.
-
Q: I would like to know the aproximate dilution of the VEGF Antibody (NB100-664) to use it in a Western blot assay.
A: In regards to your inquiry about our VEGF antibody (NB100-664), I am showing from the lab data that they have tested dilutions in the range of 1:500-1:1000 for this antibody. The optimal dilution should depend on your samples and the level of expression of VEGF, but a good starting point would be 1:1000 and then optimize from there.
-
Q: What is the heparin binding activity of FGF and VEGF?
A: These proteins are not assayed for their ability to bind Heparin. More information about the FGF family of growth factors is available in this review article: Basilico, C. (1992) Adv. Can. Res. 59:115.
-
Q: What is the minimum amount of protein necessary for the detection of VEGF by Western blot using VEGF Antibody (NB100-664)?
A: I would suggest at minimum 10ug of protein per well but if you have enough sample you might want to run multiple wells with increasing amounts of protein to optimize the conditions in your hands. You could start with 5ug and do a couple of wells with 10ug, 20ug and 40ug. Also try using different dilutions of primary, start with 1:500-1:1000 and work your way up or down depending on what your blot looks like.
-
Q: Why is the molecular weight of VEGF different from the similar antibody, for some companies the the molecular weight is 40KD)?
A: I can't comment on another company's antibody because I don't have any information about their products. I can tell you that VEGF is expressed in a variety of isoforms and is subject to various post-translational modifications that influence its apparent molecular weight in an SDS-PAGE gel compared to the theoretical molecular weight.
Associated Pathways
 Embryonic and Induced Pluripotent Stem Cell Differentiation Pathways & Lineage-specific Markers
Embryonic and Induced Pluripotent Stem Cell Differentiation Pathways & Lineage-specific Markers
 Neural Stem Cell Differentiation Pathways & Lineage-specific Markers
Neural Stem Cell Differentiation Pathways & Lineage-specific Markers
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways


![Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - VEGF Antibody (VG1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-664_mouse-monoclonal-vegf-antibody-vg1-310202415382436.jpg)
![Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - VEGF Antibody (VG1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-664_mouse-monoclonal-vegf-antibody-vg1-310202415535199.jpg)
![Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - VEGF Antibody (VG1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-664_mouse-monoclonal-vegf-antibody-vg1-310202415523928.jpg)
![Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - VEGF Antibody (VG1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-664_mouse-monoclonal-vegf-antibody-vg1-31020241555228.jpg)
![Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - VEGF Antibody (VG1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-664_mouse-monoclonal-vegf-antibody-vg1-310202415535177.jpg)
![Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - VEGF Antibody (VG1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-664_mouse-monoclonal-vegf-antibody-vg1-3102024155374.jpg)
![Western Blot: VEGF Antibody (VG1) - BSA Free [NB100-664] - VEGF Antibody (VG1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-664_mouse-monoclonal-vegf-antibody-vg1-31020241682159.jpg)
![Western Blot: Mouse Monoclonal VEGF Antibody (VG1) [NB100-664] VEGF Antibody (VG1) - BSA Free](https://resources.rndsystems.com/images/products/antibody/nb100-664_mouse-monoclonal-vegf-antibody-vg1-western-blot-3112025142219.jpg)
![Immunohistochemistry: VEGF Antibody (VG1) - BSA Free [NB100-664] - VEGF Antibody (VG1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-664_mouse-monoclonal-vegf-antibody-vg1-immunohistochemistry-13220261814152.jpg)
![Immunohistochemistry: VEGF Antibody (VG1) - BSA Free [NB100-664] - VEGF Antibody (VG1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-664_mouse-monoclonal-vegf-antibody-vg1-immunohistochemistry-132202619283423.jpg)