Interleukin 1
Overview
Interleukin 1 (IL-1) is a general name for two distinct proteins, IL-1 alpha and IL-1 beta, that are considered the first of a small (but possibly growing) family of regulatory and inflammatory cytokines.1 Along with IL-1 receptor antagonist (IL-1ra)2 and IL-18,3 these molecules play important roles in the up- and down-regulation of acute inflammation.2-4 In the immune system, the production of IL-1 is typically induced, generally resulting in inflammation. In fact, IL-1 alpha are generally thought of as prototypical pro-inflammatory cytokines.4-8 The effects of IL-1, however, are not limited to inflammation, as IL-1 has also been associated with bone formation and remodeling,9, 10 insulin secretion,11, 12 appetite regulation,13, 14 fever induction,15-17 neuronal phenotype development,18, 19 and IGF/GH physiology.20, 21 IL-1 has also been known by a number of alternative names, including lymphocyte activating factor, endogenous pyrogen, catabolin, hemopoietin-1, melanoma growth inhibition factor, and osteoclast activating factor. The properties and biological activities of IL-1 have been extensively reviewed.4, 5, 8, 22, 23
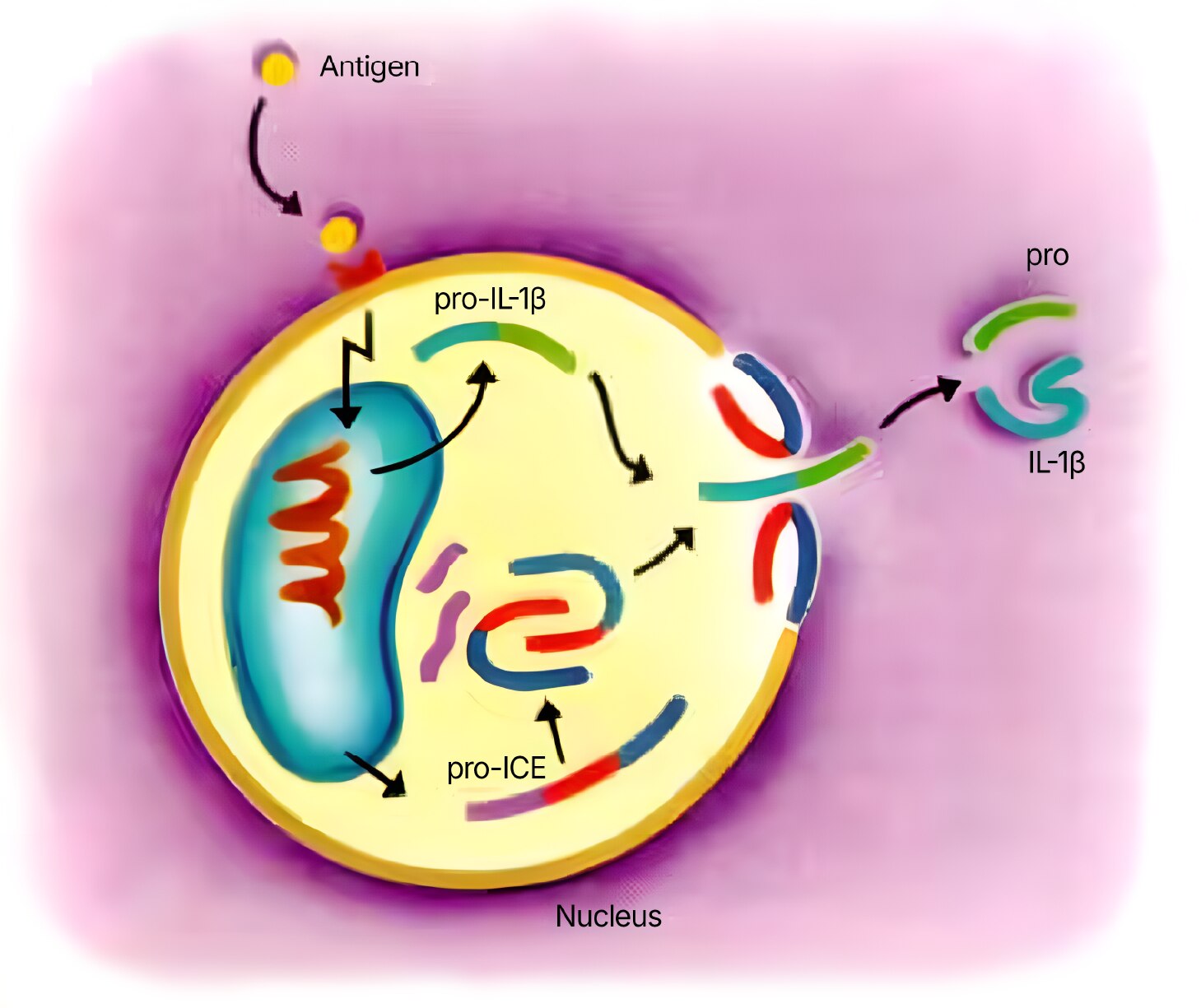
Figure 1: Pro-IL-1beta/ICE interaction
Structural Information
Human IL-1 alpha and IL-1 beta are synthesized as 31-33 kDa, variably glycosylated pro-cytokines that share 25% amino acid (aa) identity across their entire precursor structure, and 22% aa identity over their mature segments.24
IL-1 beta: The IL-1 beta precursor is 269 aa long and is cleaved into a 116 aa pro-segment, and a 153 aa, 17 kDa mature segment.24,25 Cleavage of IL-1 beta is complex, involving multiple enzymes and giving rise to multiple products. IL-1 beta cleavage is suggested to occur through the action of 45 kDa cysteine protease named IL-1 beta-converting enzyme (ICE).25 This enzyme is apparently cell-surface associated and under the control of cell membrane inhibitors. 26,27 After cleavage of the pro-form, the 17 kDa mature segment is released, along with 14 kDa (89 aa) and 17.5 kDa (116 aa) pro-region fragments.28 Although the pro-forms have no known biological activity, there may be a regulatory interaction between the pro- and mature segments both before and after cleavage.29 Neither IL-1 beta nor IL-1 alpha have classic signal sequences. However, in the case of IL-1 beta, it appears that ABC (ATP binding cassette) transporters are crucial to secretion.30 Alternative cleavage mechanisms for IL-1 beta also exist, some involving extracellular enzymes for secreted precursor9 and other involving novel pathways.31
IL-1a: IL-1 alpha differs markedly from IL-1 beta not only in primary structure, but in the importance of its pro-segment. Like IL-1 beta, IL-1 alpha can be synthesized as a 33 kDa, 271 aa pro-cytokine that is enzymatically cleaved (by calpain) into a bio-active 17 kDa, 159 aa mature segment and a 112 aa pro-sequence.24,42,43 Unlike IL-1 beta, IL-1 alpha also appears to have bioactivity while in the form of an intact pro-cytokine or precursor.44 This occurs both intracellularly and on the cell surface. Intracellularly, pro-IL-1 alpha appears to act through its nuclear localization signal that is found between residues #79-85 of its pro-region.45,46 In endothelial cells, the translocation of IL-1 alpha results in both cell migration and PAI-1 gene activation.45,47 On the cell surface, IL-1 alpha is found as a "membrane-bound" molecule, anchored to the cell membrane by either a myristoylated lysine in the pro-region, or (in the mouse) via a mannose glycosylation residue that attaches to membrane-associated lectin.49 There is a 78% aa identity between mouse and human mature IL-1 beta.60 Full length IL-1 alpha shows 54% aa identity and mature (159 aa) IL-1 alpha shows IL-1 alpha shows 58% aa identity between mouse and human.24 Both human and rat IL-1 are active on mouse cells.24,61
Receptors
IL-1 alpha and IL-1 beta exert their effects by binding to specific receptors. Two distinct IL-1 receptor binding proteins, plus a non-binding signaling accessory protein have been identified.62-66 Each have three extracellular immunoglobulin-like (Ig-like) domains, qualifying them for membership in the type IV cytokine receptor family.67 The two receptor binding proteins are termed type I IL-1 receptor (IL-1 RI) and type II IL-1 receptor (IL-1 RII) respectively. Human IL-1 RI is a 552 aa, 80 kDa transmembrane glycoprotein62 that has been isolated from astrocytes,68 chondrocytes,69 endothelium,69 fibroblasts,62, 69 keratinocytes,70 neurons,68, 71 oocytes,72 pancreatic beta-cells,73 smooth muscle cells69 and T cells.62,74 By contrast, human IL-1 RII is a 385 aa, 60-68 kDa transmembrane glyco-protein64,76 that is found on B cells,69 keratinocytes,70 monocytes,69 neutrophils76 and T-cells.74 The two types of receptors show a modest 28% aa identity in their extracellular regions (approximately 320 aa in length)62,64 but differ markedly in their cytoplasmic regions. IL-1 RI has a 213 aa cytoplasmic region, while IL-1 RII has only a short 29 aa cytoplasmic tail. This difference is reflected in the purported function of each receptor. Depending on species and cell type, the two types of receptors show differential binding characteristics for IL-1 alpha and IL-1 beta. Typically, IL-1 alpha is reported to bind preferentially to IL-1 RI (KdIL-1 alpha = 50 pm; KdIL-1 beta = 1 nM), while IL-1 beta binds preferentially to IL-1 RII (KdIL-1 alpha = 1.5 µM; KdIL-1 beta = 2 nM).77-80
Until recently, IL-1 RI was considered a signal transducing receptor, while IL-1 RII was believed to be a decoy, or activity down-modulating dummy receptor.81-83 It is now known that IL-1 signaling is not generated by the IL-1 RI molecule, but by an IL-1 RI accessory protein (IL-1 RAcP) that only interacts with IL-1 RI.65, 66 Human IL-1 RAcP is a 550 aa, 66 kDa transmembrane glycoprotein that can be assumed to occur on all cells that express functional type I IL-1 Rs.65, 84 Like IL-1 RI, it contains three Ig-like domains in its extracellular region and has 25% aa identity with each of the two binding receptors.66 Unlike IL-1 RI, it displays no affinity for either IL-1 alpha, IL-1 beta, or IL-1ra,66, 85 and its expression appears to be relatively stable, unaffected by cytokines which up- and down-modulate IL-1 RI expression.86 It appears that IL-1 first binds to IL-1 RI with a Kd of approximately 1 nM. This complex then recruits IL-1 RAcP (bound to cytosolic serine/threonine kinases) to create a 200 kDa complex with a Kd of 250 pM , which results in IL-1/IL-1 R complex internalization and downstream activation.65, 66, 85, 87
Soluble forms of IL-1 RI and IL-1 RII have been reported in sera88, 89 and a soluble form of IL-1 RAcP is possible via alternate splicing.66 The soluble form of IL-1 RI is 60 kDa and preferentially binds IL-1ra (Kd = 70 pM).88 Proteolytic cleavage of IL-1 RII produces two sizes of the molecule, 47 kDa79, 90 and 57 kDa.75 IL-1 RII demonstrates high-affinity binding for both IL-1 alpha (Kd = 2 µM) and IL-1 beta (Kd = 2 - 3 nM).75, 79 Once solubilized, IL-1 RII differs markedly from its membrane form in that 1) it shows 2000-fold less affinity for IL-1ra, making IL-1ra more available to antagonize IL-1 activity; and 2) it binds the 31 kDa IL-1 beta precursor (either bound or unbound) is inefficient in activating IL-1 RI.44 Thus, IL-1 RII, either as a membrane-bound or soluble molecule, seems to downregulate the overall activity of IL-1.
Function
IL-1 is expressed by many cells and has multiple functions including local inflammation. Cells known to express IL-1a include astrocytes,32 fibroblasts,50 hepatocytes,51 keratinocytes,52 type II Greater alveolar cells,53 brown fat adipocytes,54 thymic myoid cells,55 T cells and eosinophils,56 dendritic cells,57 macrophages,49 monocytes58 and oligodendrocytes.32 There is one report of an IL-1 beta to an IL-1 beta include astrocytes,32 adrenal cortical cells,33 NK cells,34 macrophages and monocytes,35 endothelial cells,36 keratinocytes,31 megakaryocytes and platelets,37, 38 neurons,39 neutrophils,40 oligodendroglia,32 osteoblasts,10 Schwann cells,41 trophoblasts,34 and T cells plus fibroblasts.6
Following bacterial or immunoglobulin ligation of monocyte/macrophage CD14 (the LPS receptor)92 or CD64 (the IgG receptor),93 IL-1 can be released into a local environment. Within this environment, IL-1 impacts a number of cells. First, capillary endothelial cells are induced to do two things; one, secrete chemokines such as MCP-1,94 and two, upregulate the expression of vascular adhesion molecules such as E-Selectin, ICAM-1 and VCAM-1.95 MCP-1 provides a stimulus for chemotaxis and activates mononuclear cell integrins,96 thus facilitating mononuclear infiltration into an area of early inflammation. IL-1 also induces expression of itself in newly arriving monocytes, thus reinforcing the overall process.97 In terms of other pro-inflammatory molecules, IL-1 apparently is needed for the efficient production of IFN-gamma. On resident NK cells, IL-1 apparently works in conjunction with macrophage-derived IL-12 to induce IFN-gamma induced activation of macrophages.99 Finally, IL-1 also induces the expression of MMPs from resident fibroblasts. This can have at least two effects: first, extracellular matrix degradation can facilitate monocyte migration, and second, MMPs are known to degrade IL-1 beta, thus down-modulating the local inflammatory response initiated by IL-1.
References
- Bazan, J.F. et al. (1996) Nature 379:591.
- Arend, W.P. et al. (1998) Annu. Rev. Immunol. 16:27.
- Kohno, K. & M. Kurimoto (1998) Clin. Immunol. Immunopathol. 86:11.
- Dayer-J.M. (1997) Immunologist 56:192.
- Dinarello, C.A. (1994) Eur. Cytokine Netw. 5:517.
- Sporri, B. et al. (1996) Cytokine 8:631.
- Hu, J. & I.A. Cotgreave (1997) J. Clin. Invest. 99:2312.
- Dinarello, C.A. (1996) Blood 87:2095.
- Kusano, K. et al. (1998) Endocrinology 139:1338.
- Dodds, R.A. et al. (1994) J. Histochem. Cytochem. 42:733.
- Mandrup-Poulsen, T. (1996) Diabetologia 39:1005.
- Ling, Z. et al. (1998) Endocrinology 139:1540.
- Plata-Salaman, C.R. & S.E. Ilyin (1997) J. Neurosci. Res. 49:541.
- Plata-Salaman, C.R. (1994) Physiol. Behav. 55:727.
- Hansen, M.K. et al. (1998) J. Neurosci. 18:2247.
- Goehler, L.E. et al. (1997) Brain Res. Bull. 43:357.
- Zheng, H. et al. (1995) Immunity 3:9.
- Ling, Z.D. et al. (1998) Exp. Neurol. 149:411.
- Shadiack, A.M. et al. (1993) J. Neurosci. 13:2601.
- Fan, J. et al. (1996) Am. J. Physiol. 270:R621.
- Delhanty, P.J.D. (1998) Biochem. Biophys. Res. Commun. 243:269.
- Dinarello, C.A. (1991) Blood 77: 1627.
- Dinarello, C.A. (1992) Chem. Immunol. 51:1.
- March, C.J. et al. (1985) Nature 315:641.
- Thornberry, N.A. et al. (1992) Nature 356:768.
- Singer, I.I. et al. (1995) J. Exp. Med. 182:1447.
- Schonbeck, U. et al. (1997) J. Exp. Med. 185:1287.
- Higgins, G.C. et al. (1994) J. Exp. Med. 180:607.
- Kobayashi, Y. et al. (1997) J. Biochem. 121:896.
- Hamon, Y. et al. (1997) Blood 90:2911.
- Nylander Lundquist, E. & T. Egelrud (1997) Eur. J. Immunol. 27:2165.
- da Cunha, A. et al. (1993) J. Neuroimmunol. 42:71.
- Gonzales-Hernandez, J.A. et al. (1995) Clin. Exp. Immunol. 99:137.
- Jokhi, P.P. et al. (1997) Cytokine 9:126.
- Wewers, M.D. et al. (1997) J. Immunol. 159:5964.
- Warner, S.J.C. et al. (1987) J. Immunol. 139:1911.
- Jiang, S. et al. (1994) Blood 84:4151.
- Loppnow, H. et al. (1998) Blood 91:134.
- Freidin, M. et al. (1992) Proc. Natl. Acad. Sci. USA 89:10440.
- Malyak, M. et al. (1994) J. Clin. Immunol. 14:20.
- Skundric, D.S. et al. (1997) J. Neuroimmunol. 74:9.
- Kobayashi, Y. et al. (1990) Proc. Natl. Acad. Sci. USA 87:5548.
- Watanabe, N. & Y. Kobayashi (1994) Cytokine 6:597.
- Mosley, B. et al. (1987) J. Biol. Chem. 262:2941.
- Maier, J.A.M. et al. (1994) Mol. Cell. Biol. 14:1845.
- Wessendorf, J.H.M. et al. (1993) 268:22100.
- McMahon, G.A. et al. (1997) J. Biol. Chem. 272:28202.
- Stevenson, F.T. et al. (1993) Proc. Natl. Acad. Sci. USA 90:7245.
- Brody, D.T. & S.K. Durum (1989) J. Immunol. 143:1183.
- Fini, M.E. et al. (1994) J. Biol. Chem. 269:11291.
- Tsukui, T. et al. (1994) Exp. Cell Res. 210:172.
- Yu, H-S. et al. (1996) J. Invest. Dermatol. 107:593.
- Kumar, N.M. et al. (1996) J. Cell. Physiol. 169:186.
- Burysek, L. & J. Houstek (1996) Cytokine 8:460.
- Iwakami, N. et al. (1996) Immunology 87:108.
- Krzesicki, R.F. et al. (1997) Am. J. Resp. Cell Mol. Biol. 16:693.
- Ghanekar, S. et al. (1996) J. Immunol. 157:4028.
- Mallardo, M. et al. (1994) J. Biol. Chem. 269:14899.
- Beuscher, H.U. et al. (1992) J. Exp. Med. 175:1793.
- Gray, P.W. et al. (1986) J. Immunol. 137:3644.
- Bergsteinsdottir, K. et al. (1991) J. Neuroimmunol. 34:15.
- Sims, J.E. et al. (1989) Proc. Natl. Acad. Sci. USA 86:8946.
- Sims, J.E. et al. (1988) Science 241:585.
- McMahan, C.J. et al. (1991) EMBO J. 10:2821.
- Huang, J. et al. (1997) Proc. Natl. Acad. Sci. USA 94:12829.
- Greenfeder, S.A. et al. (1995) J. Biol. Chem. 270:13575.
- Miyajima, A. et al. (1992) Annu. Rev. Immunol. 10:295.
- Yabuuchi, K. et al. (1994) Mol. Brain Res. 27:27.
- Colotta, F. et al. (1994) Immunol. Today 15:562.
- Groves, R.W. et al. (1995) J. Immunol. 154:4065.
- Cunningham, E.T. et al. (1992) J. Neurosci. 12:1101.
- Deyerle, K.L. et al. (1992) J. Immunol. 149:1657.
- Jafarian-Tehrani, M. et al. (1995) Endocrinology 136:609.
- McKean, D.J. et al. (1993) J. Immunol. 151:3500.
- Liu, C. et al. (1996) J. Biol. Chem. 271:20965.
- Colotta, F. et al. (1995) J. Exp. Med. 181:2181.
- Dower, S.K. et al. (1992) Chem. Immunol. 51:33.
- Jobling, S.A. et al. (1988) J. Biol. Chem. 263:16372.
- Symons, J.A. et al. (1991) J. Exp. Med. 174:1251.
- Chizzonite, R. et al. (1989) Proc. Natl. Acad. Sci. USA 86:8029.
- Sims, J.E. et al. (1994) Clin. Immunol. Immunopathol. 72:9.
- Sims, J.E. et al. (1993) Proc. Natl. Acad. Sci. USA 90:6155.
- Colotta, F. & A. Mantovani (1994) Trends Pharmacol. Sci. 14:138.
- Wesche, H. et al. (1996) FEBS Lett. 391:104.
- Korherr, C. et al. (1997) Eur. J. Immunol. 27:262.
- Yoon, D-Y. & C.A. Dinarello (1998) J. Immunol. 160:3170.
- Volpe, F. et al. (1997) FEBS Lett. 419:41.
- Svenson, M. et al. (1993) Cytokine 5:427.
- Giri, J.G. et al. (1994) J. Immunol. 153:5802.
- Colotta, F. et al. (1993) Science 261:472.
- Symons, J.A. et al. (1995) Proc. Natl. Acad. Sci. USA 92:1714.
- Nockher, W.A. & J.E. Scherberich (1997) J. Immunol. 158:1345.
- van de Winkel J.G.J. & P.J.A. Capel (1993) Immunol. Today 14:215.
- J. Immunol. 144:3034
- Kupper, T.S. & R.W. Groves (1995) J. Invest. Dermatol. 105:62S.
- Rollins, B.J. (1997) Blood 90:909.
- Dinarello, C.A. et al. (1987) J. Immunol. 139:1902.
- Hunter, C.A. et al. (1995) J. Immunol. 155:4347.
- Billiau, A. (1996) Adv. Immunol. 62:61.
- Ito, A. et al. (1996) J. Biol. Chem. 271:14657
