Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
112 results for "S5a/Angiocidin" in Products
S5a/Angiocidin Products
S5a/Angiocidin, also known as Anti-secretory Factor (ASF), is classified under the gene PSMD4, but is often referred to by a different name depending on the context in which it is described. S5a and ASF have identical 377 amino acid (aa) sequences, while Angiocidin is described as having an additional Gly255Glu256Arg257 sequence in its C-Terminus. The human protein shares 96% and 99% aa sequence identity with its mouse and rat orthologs, respectively. Structurally, it contains an N-terminal von ...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB, ICC/IF, Mycoplasma |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, ICC/IF |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #5G1X6 |
| Applications: | WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB, ICC/IF |
Loading...
| Reactivity: | Human, Bovine |
| Details: | Mouse IgG Polyclonal |
| Applications: | WB, IP |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Monoclonal Clone #SR1890 |
| Applications: | WB, Flow, IP |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2b Kappa Monoclonal Clone #CPTC-PSMD4-3 |
| Applications: | WB, MA |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2b Kappa Monoclonal Clone #CPTC-PSMD4-3 |
| Applications: | WB, MA |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2a Kappa Monoclonal Clone #3C9 |
| Applications: | WB, ELISA |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...


![Western Blot: S5a/Angiocidin Antibody [NBP2-19952] Western Blot: S5a/Angiocidin Antibody [NBP2-19952]](https://resources.rndsystems.com/images/products/S5a-Angiocidin-Antibody-Western-Blot-NBP2-19952-img0004.jpg)
![Western Blot: S5a/Angiocidin Antibody [NBP2-37888] Western Blot: S5a/Angiocidin Antibody [NBP2-37888]](https://resources.rndsystems.com/images/products/Proteasome-19S-S5A-Antibody-Western-Blot-NBP2-37888-img0006.jpg)
![Immunocytochemistry/ Immunofluorescence: S5a/Angiocidin Antibody [NBP1-90821] Immunocytochemistry/ Immunofluorescence: S5a/Angiocidin Antibody [NBP1-90821]](https://resources.rndsystems.com/images/products/Proteasome-19S-S5A-Antibody-Immunocytochemistry-Immunofluorescence-NBP1-90821-img0007.jpg)

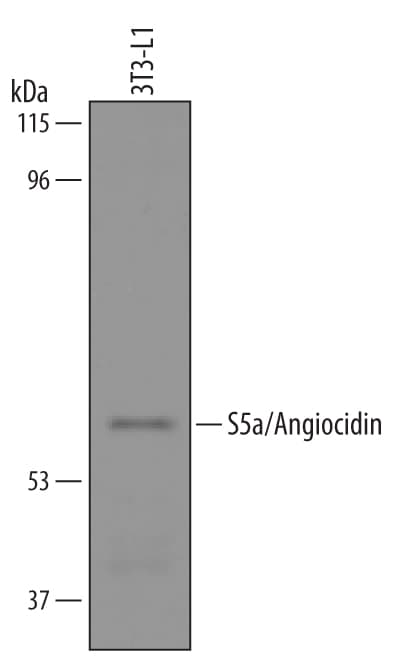
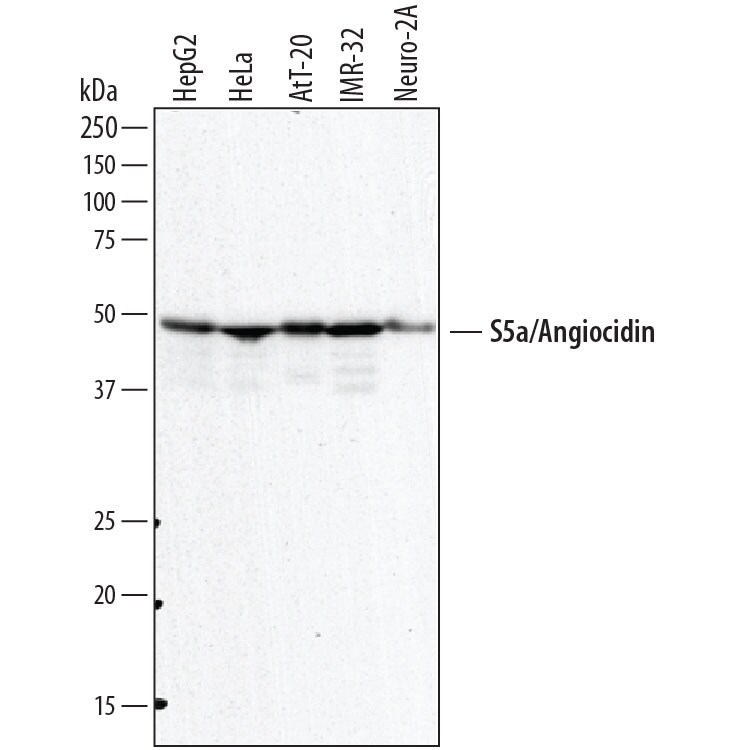
![Western Blot: S5a/Angiocidin Antibody (5G1X6) [NBP3-16240] Western Blot: S5a/Angiocidin Antibody (5G1X6) [NBP3-16240]](https://resources.rndsystems.com/images/products/S5a-Angiocidin-Antibody-5G1X6-Western-Blot-NBP3-16240-img0002.jpg)
![Western Blot: S5a/Angiocidin Antibody [NBP2-19951] Western Blot: S5a/Angiocidin Antibody [NBP2-19951]](https://resources.rndsystems.com/images/products/Proteasome-19S-S5A-Antibody-Western-Blot-NBP2-19951-img0001.jpg)
![Western Blot: S5a/Angiocidin Antibody [H00005710-B01P] Western Blot: S5a/Angiocidin Antibody [H00005710-B01P]](https://resources.rndsystems.com/images/products/Proteasome-19S-S5A-Antibody-Western-Blot-H00005710-B01P-img0001.jpg)
![Western Blot: S5a/Angiocidin Antibody (SR1890) [NBP3-21962] -](https://resources.rndsystems.com/images/products/nbp3-21962_rabbit-s5a-angiocidin-mab-sr1890-57202316112116.jpg)
![Western Blot: S5a/Angiocidin Antibody (CPTC-PSMD4-3) [NBP3-07250] Western Blot: S5a/Angiocidin Antibody (CPTC-PSMD4-3) [NBP3-07250]](https://resources.rndsystems.com/images/products/S5a-Angiocidin-Antibody-CPTC-PSMD4-3-Western-Blot-NBP3-07250-img0002.jpg)
![Western Blot: S5a/Angiocidin Antibody (CPTC-PSMD4-3)Azide and BSA Free [NBP3-08271] Western Blot: S5a/Angiocidin Antibody (CPTC-PSMD4-3)Azide and BSA Free [NBP3-08271]](https://resources.rndsystems.com/images/products/S5a-Angiocidin-Antibody-CPTC-PSMD4-3-Western-Blot-NBP3-08271-img0001.jpg)
![Western Blot: S5a/Angiocidin Antibody (3C9) [H00005710-M01] Western Blot: S5a/Angiocidin Antibody (3C9) [H00005710-M01]](https://resources.rndsystems.com/images/products/S5a-Angiocidin-Antibody-3C9-Western-Blot-H00005710-M01-img0002.jpg)