Interleukin 17 (IL-17; also IL-17A and CTLA-8) is a 17 kDa member of the IL-17 family of cytokines (1). Members of this family demonstrate a structural motif termed a cysteine knot which characterize a large superfamily of growth factors. Although most cysteine knot superfamily members use three intrachain disulfide bonds to create a knot, IL-17 family molecules generate the same structural form with only two disulfide links (2-4). Based on the amino acid (aa) sequence alignment with human IL-17, canine IL-17 is 130 aa in length. It is secreted as a 35 kDa disulfide-linked homodimer and as a 40 kDa disulfide-linked heterodimer with IL-17F (5). Canine IL‑17 is 81% identical on the aa level to human IL-17. IL-23 drives Th17 lymphocytes to produce IL-17 (6-8). IL-17’s production has also been demonstrated in gamma delta T cells (9), CD8+ memory T cells (10-11), eosinophils (12), neutrophils (10), and monocytes (13). Studies have identified that the widely expressed receptors IL‑17RA and IL-17RC form a heterodimer for the binding of IL-17 (6, 14-15). The predominant function of IL-17 is thought to be as a proinflammatory mediator through a variety of mechanisms (16). Locally, IL‑17 stimulates production of IL-6, prostaglandin E and nitric oxide (16-19), and synergy with other inflammatory cytokines such as TNF-alpha, IL‑1 beta and IFN -gamma leads to up-regulation of gene expression and progression and amplification of local inflammation (16, 20-22). IL‑17 also mediates chemotaxis of neutrophils and monocytes to sites of inflammation through the chemoattractant mediators IL-8, GRO‑ alpha, and MCP-1 (16, 22-25) while augmenting production of hematopoietic growth factors, such as G-CSF and GM-CSF (16, 26, 27), which promote the growth and maturation of the recruited myeloid cells. In addition, IL-17 serves as a bridge between innate and adaptive immune responses by enhancing the induction of co-stimulatory molecules such as ICAM-1 and other cytokines (16, 22, 28), thereby supporting T cell activation. IL-17 expression has been associated with many inflammatory diseases, such as rheumatoid arthritis, multiple sclerosis, asthma, systemic lupus erythematosus and allograft rejection (15).

Key Product Details
Species Reactivity
Canine
Applications
Neutralization
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2A Clone # 665909
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant canine IL-17
Gly26-Ala155
Accession # NP_001159350
Gly26-Ala155
Accession # NP_001159350
Specificity
Detects canine IL-17 in direct ELISAs. In direct ELISAs, approximately 25%
cross-reactivity with recombinant human (rh) IL-17A is observed and no
cross-reactivity with rhIL-17F or recombinant mouse IL-17A is observed.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2A
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Canine IL‑17 Antibody
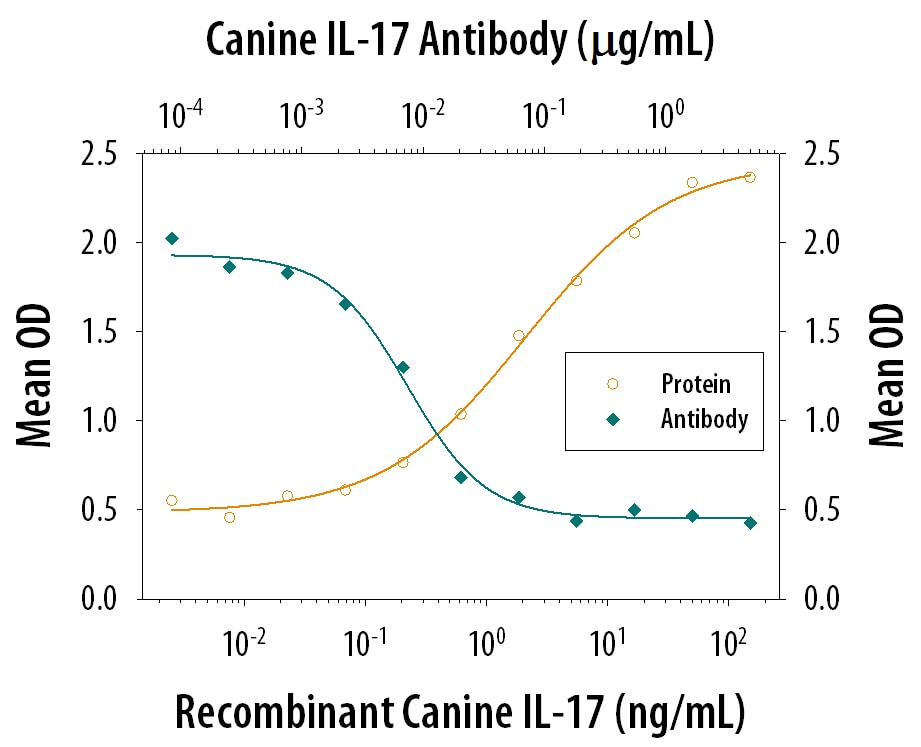
IL-6 Secretion Induced by IL‑17 and Neutralization by Canine IL‑17 Antibody.
Recombinant Canine IL-17 (Catalog # 5848-CL) induces IL-6 secretion in the NIH-3T3 mouse embryonic fibroblast cell line in a dose-dependent manner (orange line). IL-6 Secretion elicited by Recombinant Canine IL-17 (5 ng/mL) is neutralized (green line) by increasing concentrations of Mouse Anti-Canine IL-17 Monoclonal Antibody (Catalog # MAB5848). The ND50 is typically 5-30 ng/mL.Applications for Canine IL‑17 Antibody
Application
Recommended Usage
Neutralization
Measured by its ability to neutralize IL‑17-induced IL-6 secretion in the NIH‑3T3 mouse embryonic fibroblast cell line. Yao, Z. et al. (1995) Immunity 3:811.The Neutralization Dose (ND50) is typically 5-30 ng/mL in the presence of 5 ng/mL Recombinant Canine IL‑17.
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Sterile PBS to a final concentration of 0.5 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-17/IL-17A
References
- Gaffen, S.L. et al. (2006) Vitam. Horm. 74:255.
- Kawaguchi, M. et al. (2004) J. Allergy Clin. Immunol. 114:1265.
- Kolls, J.K. and A. Linden (2004) Immunity 21:467.
- Moseley, T.A. et al. (2003) Cytokine Growth Factor Rev. 14:155.
- Wright, J.F. et al. (2007) J. Biol. Chem. 282:13447.
- Cheung, P.F.Y. et al. (2008) J. Immunol. 180:5625.
- Steinman, L. (2007) Nat. Med. 13:139.
- Hunter, C.A. (2005) Nat. Rev. Immunol. 5:521.
- Lockhart, E. et al. (2006) J. Immunol. 177:4662.
- Ferretti, S. et al. (2003) J. Immunol. 170:2106.
- Shin, H.C. et al. (1999) Cytokine 11:257.
- Molet, S. et al. (2001) J. Allergy Clin. Immunol. 108:430.
- Zhou, Q. et al. (2005) Infect. Immun. 73:935.
- Kuestner, R.E. et al. (2007) J. Immunol. 179:5462.
- Chang, S.H. and C. Dong (2007) Cell Res. 17:435.
- Afzali, B. et al. (2007) Clin. Exp. Immunol. 148:32.
- Fossiez, F. et al. (1996) J. Exp. Med. 183:2593.
- Yao, Z. et al. (1995) Immunity 3:811.
- Attur, M.G. et al. (1997) Arthritis Rheum. 40:1050.
- Ruddy, M.J. et al. (2004) J. Biol. Chem. 279:2559.
- Albanesi, C. et al. (1999) J. Immunol. 162:494.
- Witowski, J. et al. (2000) J. Immunol. 165:5814.
- Miyamoto, M. et al. (2003) J. Immunol. 170:4665.
- Ye, P. et al. (2001) Am. J. Respir. Cell Mol. Biol. 25:335.
- Laan, M. et al. (2001) Br. J. Pharmacol. 133:200.
- Starnes, T. et al. (2002) J. Immunol. 169:642.
- Jones, C.E. et al. (2002) Am. J. Respir. Cell Mol. Biol. 26:748.
- Yao, Z. et al. (1995) J. Immunol. 155:5483.
Long Name
Interleukin 17
Alternate Names
CTLA-8, CTLA8, IL-17A, IL17, IL17A
Entrez Gene IDs
Gene Symbol
IL17A
UniProt
Additional IL-17/IL-17A Products
Product Documents for Canine IL‑17 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Canine IL‑17 Antibody
For research use only
Customer Reviews for Canine IL‑17 Antibody
There are currently no reviews for this product. Be the first to review Canine IL‑17 Antibody and earn rewards!
Have you used Canine IL‑17 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Loading...
Associated Pathways
 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways
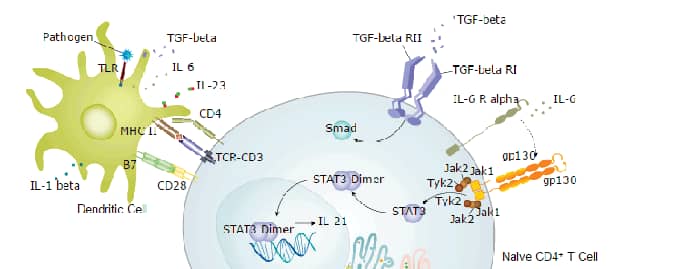
 Th17 Differentiation Pathway
Th17 Differentiation Pathway