CDO (CAM-related/down‑regulated by oncogenes, also CDON; pronounced “kid-oh”) is a 190 kDa member of the Immunoglubulin (Ig) superfamily, Ig/Fibronectin (FN) type III repeat family of cell surface proteins (1). Human CDO is a type I transmembrane (TM) glycoprotein. It is synthesized as a 1287 amino acid (aa) precursor that contains a 25 aa signal sequence, a 938 aa extracellular domain (ECD), a 21 aa TM segment and a 303 aa cytoplasmic region (1, 2). The ECD contains five C2‑type Ig-like domains, followed by three FN type III repeats. The first FN repeat (aa 577‑673) is known to bind numerous cadherins, while the third (or juxtramembrane) FN type III repeat (aa 826‑923) binds SHH (3, 4). The intracellular region is believed to signal through various bHLH transcription factors (2). One alternate splice form is reported that shows a deletion of aa 1212‑1234 in the cytoplasmic tail. The ECD of human CDO is 85% aa identical to mouse CDO ECD. CDO is found on muscle precursor and neural progenitor cells of the embryo (5, 6). It likely promotes muscle differentiation, and contributes to axon guidance and neuronal patterning (2, 7, 8, 9). These effects may be mediated through two different receptor complexes. On muscle precursors, CDO apparently acts as both a coordinating and signaling subunit. Here, it integrates N- and M-cadherin, neogenin, netrin-3 and BOC into a cis-oriented receptor complex (2). While this complex has no identified ligand, intercellular cadherin interactions or netrin, may be enough to trigger CDO/cadherin/neogenin signaling. On axons, CDO may participate in a poorly‑defined receptor complex minimally composed of CDO, BOC and Gas1 that binds SHH, and interacts with PTCH1 (7, 8, 10).


Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Asp26-Pro943 (Leu669Ile)
Accession # NP_058648
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human CDO Antibody
Detection of CDO in C2C12 Mouse Cell Line by Flow Cytometry.
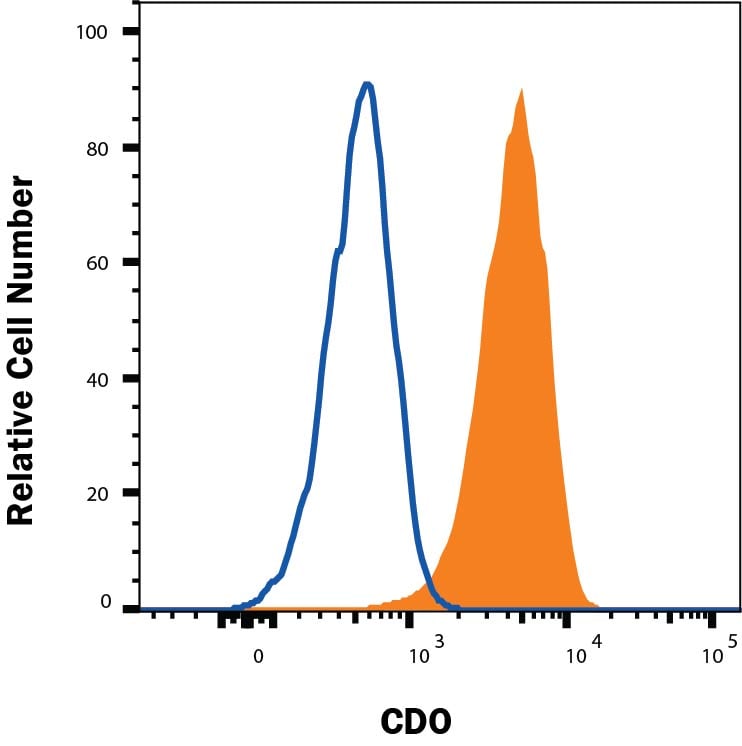
C2C12 mouse myoblast cell line was stained with Sheep Anti-Human CDO Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4384, filled histogram) or control antibody (5-001-A, open histogram), followed by NorthernLights™ 557-conjugated Anti-Sheep IgG Secondary Antibody (NL010).CDO in C2C12 Mouse Cell Line.
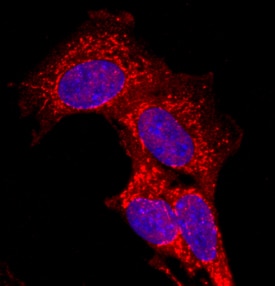
CDO was detected in immersion fixed C2C12 mouse myoblast cell line using Sheep Anti-Human CDO Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4384) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Sheep IgG Secondary Antibody (red; NL010) and counterstained with DAPI (blue). Specific staining was localized to cell membranes. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of CDO in Jurkat cells by Flow Cytometry.
Jurkat cells were stained with Sheep Anti-Human CDO Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4384, filled histogram) or isotype control antibody (Catalog # 5-001-A, open histogram), followed by Phycoerythrin-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # F0126). View our protocol for Staining Membrane-associated Proteins.Applications for Human CDO Antibody
CyTOF-ready
Flow Cytometry
Sample: C2C12 mouse myoblast and Jurkat cells
Immunocytochemistry
Sample: Immersion fixed C2C12 mouse myoblast cell line
Western Blot
Sample: Recombinant Human CDO (Catalog # 4384-CD)
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CDO
References
- Kang, J.S. et al. (1997) J. Cell Biol. 138:203.
- Krauss, R.S. et al. (2005) J. Cell Sci. 118:2355.
- Yao, S. et al. (2006) Cell 125:343.
- Kang, J-S. et al. (2003) Proc. Natl. Acad. Sci. USA 100:3989.
- Kang, J-S. et al. (2002) EMBO J. 21:114.
- Zhang, W. et al. (2006) Mol. Cell. Biol. 26:3764.
- Okada, A. et al. (2006) Nature 444:369.
- Allen, B.L. et al. (2007) Genes Dev. 21:1244.
- Kang, J-S. et al. (2004) J. Cell. Biol. 167:493.
- Tenzen, T. et al. (2006) Dev. Cell 10:647.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional CDO Products
Product Documents for Human CDO Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human CDO Antibody
For research use only
Related Research Areas
Citations for Human CDO Antibody
Customer Reviews for Human CDO Antibody
There are currently no reviews for this product. Be the first to review Human CDO Antibody and earn rewards!
Have you used Human CDO Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars