Human FGF basic/FGF2/bFGF Antibody Summary
Applications
Please Note: Optimal dilutions should be determined by each laboratory for each application. General Protocols are available in the Technical Information section on our website.
Scientific Data
 View Larger
View Larger
FGF basic/FGF2/bFGF in Human Placenta. FGF basic/FGF2/bFGF was detected in immersion fixed paraffin-embedded sections of human placenta using Goat Anti-Human FGF basic/FGF2/bFGF Antigen Affinity-purified Polyclonal Antibody (Catalog # AF-233-NA) at 10 µg/mL overnight at 4 °C. Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to trophoblast cells in chorionic villi. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.
 View Larger
View Larger
Cell Proliferation Induced by FGF basic/FGF2/bFGF and Neutralization by Human FGF basic/FGF2/bFGF Antibody. Bovine FGF basic/FGF2/bFGF (Catalog # 133-FB) stimulates proliferation in the NR6R-3T3 mouse fibroblast cell line in a dose-dependent manner (orange line). Proliferation elicited by Bovine FGF basic/FGF2/bFGF (0.5 ng/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Human FGF basic/FGF2/bFGF Antigen Affinity-purified Polyclonal Antibody (Catalog # AF-233-NA). The ND50 is typically 0.08-0.4 µg/mL.
 View Larger
View Larger
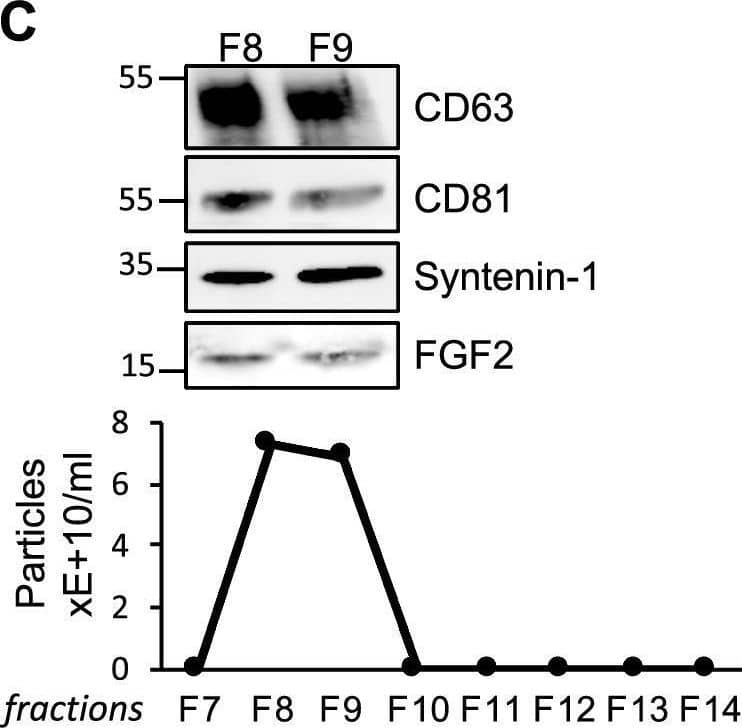
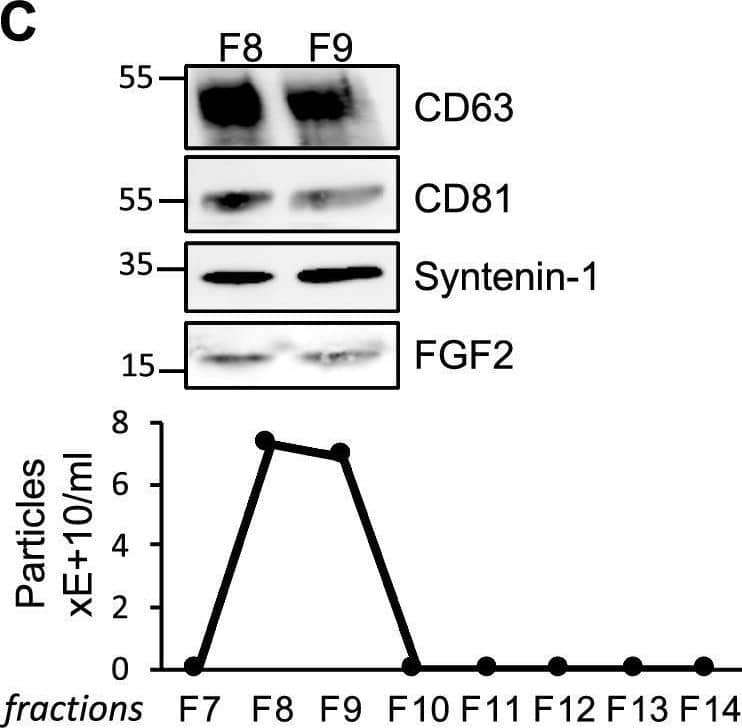
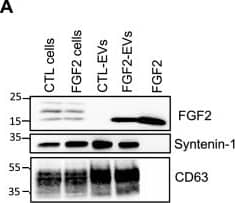
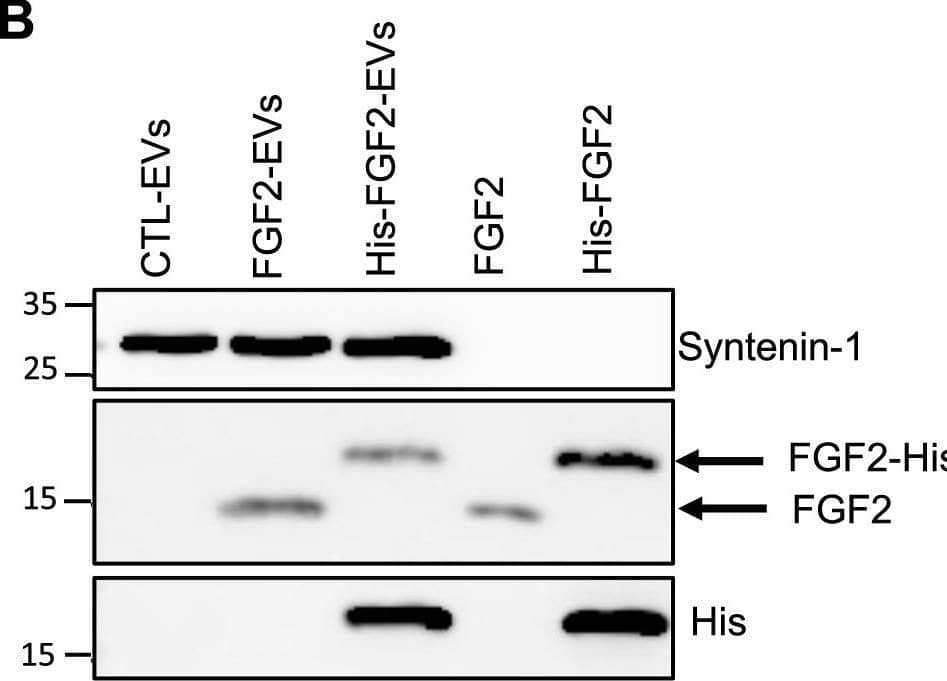
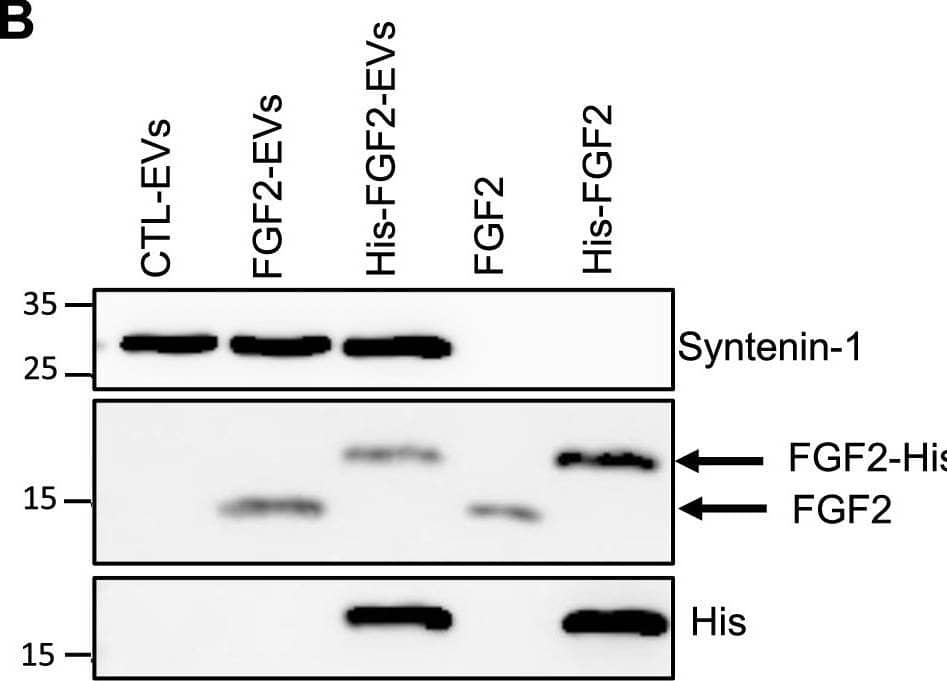
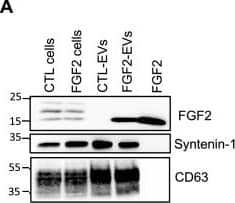
Detection of FGF basic/FGF2/bFGF by Western Blot FGF2 is found at the outer surface of EVs secreted by dermal fibroblasts cultured in FGF2-containing medium. (A) FGF2, syntenin-1 and CD63 expression in DF cells and EV lysate (20 μg) was examined by western blot. Three isoforms of endogenous FGF2 were present in DF cells while FGF2-EVs contained the low molecular weight FGF2 isoform corresponding to recombinant FGF2. Original blots are shown in Suppl. Information. (B) Cells were cultured in the presence of His-tagged FGF2; western blot with 15 μg lysate and using anti-His antibody revealed the presence of His-FGF2 in secreted EVs. A shift in FGF2 size was also observed. Syntenin-1 was used as EV marker. Original blots are shown in Suppl. Information. (C) FGF2-EVs were loaded on a SEC column and collected fractions 8 and 9 (3E + 9p) were analyzed by western blot using antibodies for FGF2 and the EV markers CD63, CD81 and syntenin-1. Original blots are shown in Suppl. Information. For quantification, fractions were also analyzed by NTA. (D) FGF2 detection in FGF2-EVs by ELISA assay. Intact CTL- and FGF2-EVs (8E + 8p) were directly placed on FGF2 ELISA wells for external surface detection. FGF2-EVs were also treated with 0.1% triton and analyzed to detect internal FGF2. No FGF2 signal was detected in CTL-EVs. (E) FGF2 expression on FGF2-EVs was detected by flow cytometry. EVs were coupled to latex beads and labeled with FGF2 antibody before analysis by cytometry. Beads without EVs (beads) and beads labelled with goat IgG antibody (IgG) serve as negative controls. A representative plot is shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36550142), licensed under a CC-BY license. Not internally tested by R&D Systems.
 View Larger
View Larger
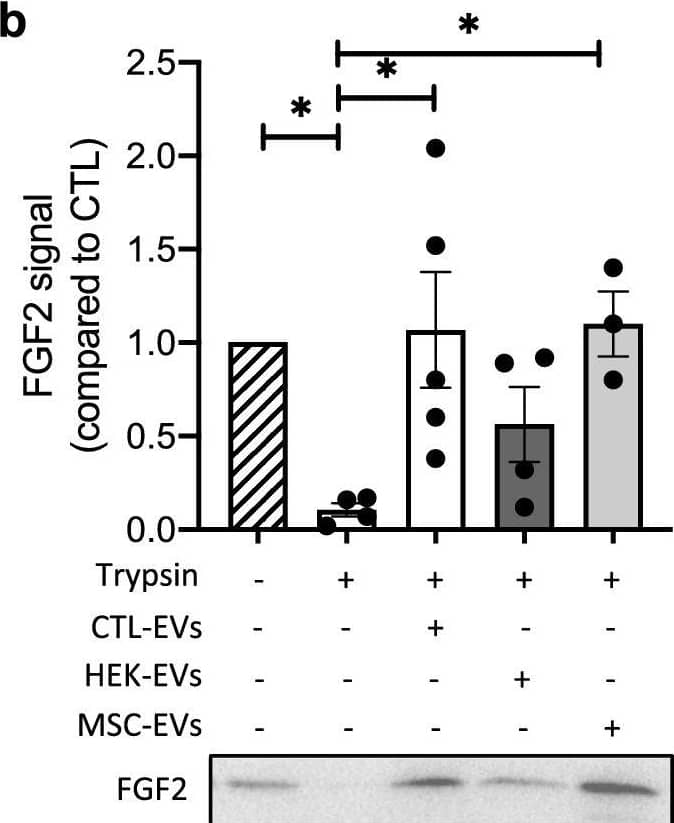
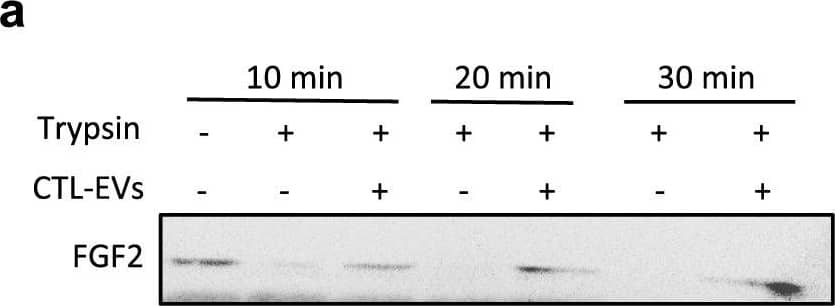
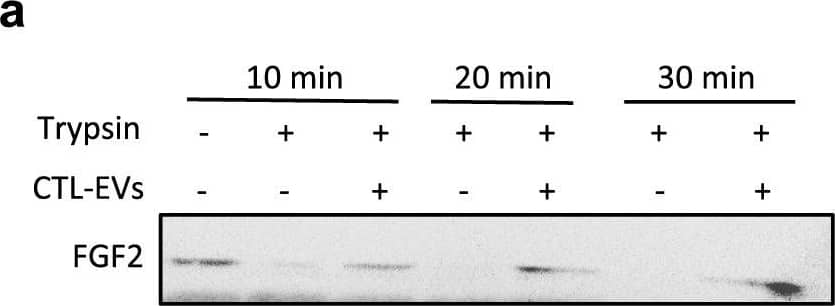
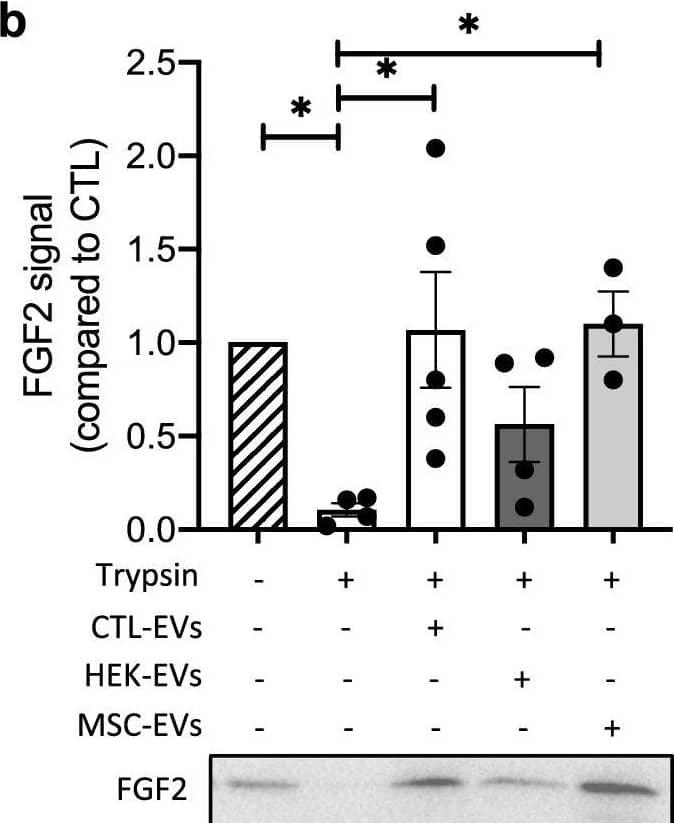
Detection of FGF basic/FGF2/bFGF by Western Blot EV-bound FGF2 is protected from degradation. (A) Effect of EV binding on FGF2 thermal stability. FGF2 (10 ng/ml), alone or after incubation with EVs (8E + 8p) from different cell types for 1 h on ice, was placed at 37 °C for 24 h to challenge FGF2 thermal stability. Residual FGF2 activity was determined by (a) proliferation assay by BrDU incorporation and (b) ELISA assay to measure ESM-1 secretion. EVs from DF, MSC, HEK cells were tested. Results are mean ± SE of four independent experiments. Statistical significance was determined by one-way ANOVA (Dunnett’s multiple test), *< 0.05, **< 0.01, ***< 0.001, ****< 0.0001. (B) Effect of EV binding on FGF2 degradation by trypsin. FGF2 (10 ng), alone or after incubation with EVs from DF, MSC or HEK cells (8E + 8p) for 1 h on ice, was held at 37 °C with trypsin/EDTA (0.05%, diluted 1:2 v/v in 25 μl) for different time periods before loading on western blot to assess residual FGF2 protein. (a) Representative blot with CTL-EVs (b) Representative blot and quantification of FGF2 signal obtained after 10 min incubation with EVs from DF, MSC or HEK cells. Results are mean ± SE of 3–5 independent experiments. Statistical significance was determined by one-way ANOVA (Dunnett’s multiple test), *< 0.05. Original blots are shown in Suppl. Information. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36550142), licensed under a CC-BY license. Not internally tested by R&D Systems.
 View Larger
View Larger
Detection of FGF basic/FGF2/bFGF by Western Blot EV-bound FGF2 is protected from degradation. (A) Effect of EV binding on FGF2 thermal stability. FGF2 (10 ng/ml), alone or after incubation with EVs (8E + 8p) from different cell types for 1 h on ice, was placed at 37 °C for 24 h to challenge FGF2 thermal stability. Residual FGF2 activity was determined by (a) proliferation assay by BrDU incorporation and (b) ELISA assay to measure ESM-1 secretion. EVs from DF, MSC, HEK cells were tested. Results are mean ± SE of four independent experiments. Statistical significance was determined by one-way ANOVA (Dunnett’s multiple test), *< 0.05, **< 0.01, ***< 0.001, ****< 0.0001. (B) Effect of EV binding on FGF2 degradation by trypsin. FGF2 (10 ng), alone or after incubation with EVs from DF, MSC or HEK cells (8E + 8p) for 1 h on ice, was held at 37 °C with trypsin/EDTA (0.05%, diluted 1:2 v/v in 25 μl) for different time periods before loading on western blot to assess residual FGF2 protein. (a) Representative blot with CTL-EVs (b) Representative blot and quantification of FGF2 signal obtained after 10 min incubation with EVs from DF, MSC or HEK cells. Results are mean ± SE of 3–5 independent experiments. Statistical significance was determined by one-way ANOVA (Dunnett’s multiple test), *< 0.05. Original blots are shown in Suppl. Information. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36550142), licensed under a CC-BY license. Not internally tested by R&D Systems.
 View Larger
View Larger
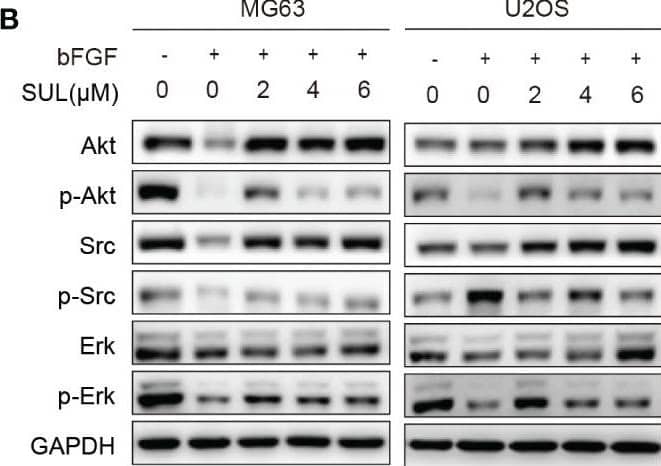
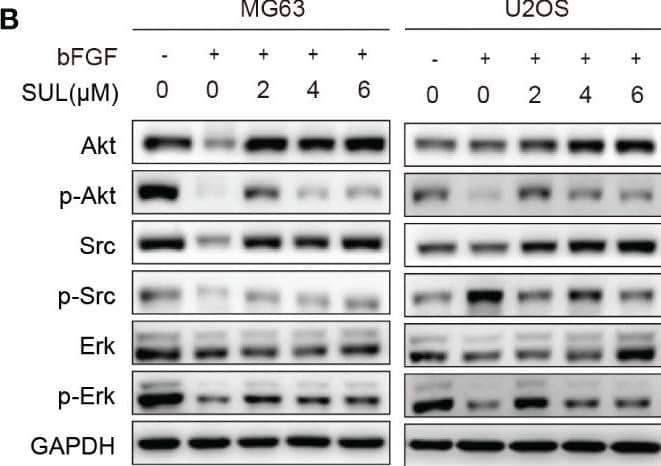
Detection of FGF basic/FGF2/bFGF by Western Blot Sulfatinib inhibits OS migration through suppression of epithelial–mesenchymal transition (EMT). (A) The protein levels of EMT and metastatic markers were detected by Western blotting after 24 h sulfatinib treatment. (B) Sulfatinib inhibits phosphorylation of migration-related signal pathway induced by bFGF. (C) Immunohistochemistry for EMT- related markers (FGFR1, p-FGFR1, N-cadherin, and E-cadherin) in tumor sections after treatment (scale bar, 100 μm). OS, osteosarcoma; bFGF, basic fibroblast growth factor. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37361567), licensed under a CC-BY license. Not internally tested by R&D Systems.
 View Larger
View Larger
Detection of FGF basic/FGF2/bFGF by Western Blot FGF2 is found at the outer surface of EVs secreted by dermal fibroblasts cultured in FGF2-containing medium. (A) FGF2, syntenin-1 and CD63 expression in DF cells and EV lysate (20 μg) was examined by western blot. Three isoforms of endogenous FGF2 were present in DF cells while FGF2-EVs contained the low molecular weight FGF2 isoform corresponding to recombinant FGF2. Original blots are shown in Suppl. Information. (B) Cells were cultured in the presence of His-tagged FGF2; western blot with 15 μg lysate and using anti-His antibody revealed the presence of His-FGF2 in secreted EVs. A shift in FGF2 size was also observed. Syntenin-1 was used as EV marker. Original blots are shown in Suppl. Information. (C) FGF2-EVs were loaded on a SEC column and collected fractions 8 and 9 (3E + 9p) were analyzed by western blot using antibodies for FGF2 and the EV markers CD63, CD81 and syntenin-1. Original blots are shown in Suppl. Information. For quantification, fractions were also analyzed by NTA. (D) FGF2 detection in FGF2-EVs by ELISA assay. Intact CTL- and FGF2-EVs (8E + 8p) were directly placed on FGF2 ELISA wells for external surface detection. FGF2-EVs were also treated with 0.1% triton and analyzed to detect internal FGF2. No FGF2 signal was detected in CTL-EVs. (E) FGF2 expression on FGF2-EVs was detected by flow cytometry. EVs were coupled to latex beads and labeled with FGF2 antibody before analysis by cytometry. Beads without EVs (beads) and beads labelled with goat IgG antibody (IgG) serve as negative controls. A representative plot is shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36550142), licensed under a CC-BY license. Not internally tested by R&D Systems.
 View Larger
View Larger
Detection of FGF basic/FGF2/bFGF by Western Blot FGF2 is found at the outer surface of EVs secreted by dermal fibroblasts cultured in FGF2-containing medium. (A) FGF2, syntenin-1 and CD63 expression in DF cells and EV lysate (20 μg) was examined by western blot. Three isoforms of endogenous FGF2 were present in DF cells while FGF2-EVs contained the low molecular weight FGF2 isoform corresponding to recombinant FGF2. Original blots are shown in Suppl. Information. (B) Cells were cultured in the presence of His-tagged FGF2; western blot with 15 μg lysate and using anti-His antibody revealed the presence of His-FGF2 in secreted EVs. A shift in FGF2 size was also observed. Syntenin-1 was used as EV marker. Original blots are shown in Suppl. Information. (C) FGF2-EVs were loaded on a SEC column and collected fractions 8 and 9 (3E + 9p) were analyzed by western blot using antibodies for FGF2 and the EV markers CD63, CD81 and syntenin-1. Original blots are shown in Suppl. Information. For quantification, fractions were also analyzed by NTA. (D) FGF2 detection in FGF2-EVs by ELISA assay. Intact CTL- and FGF2-EVs (8E + 8p) were directly placed on FGF2 ELISA wells for external surface detection. FGF2-EVs were also treated with 0.1% triton and analyzed to detect internal FGF2. No FGF2 signal was detected in CTL-EVs. (E) FGF2 expression on FGF2-EVs was detected by flow cytometry. EVs were coupled to latex beads and labeled with FGF2 antibody before analysis by cytometry. Beads without EVs (beads) and beads labelled with goat IgG antibody (IgG) serve as negative controls. A representative plot is shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36550142), licensed under a CC-BY license. Not internally tested by R&D Systems.
 View Larger
View Larger
Detection of FGF basic/FGF2/bFGF by Western Blot EV-bound FGF2 is protected from degradation. (A) Effect of EV binding on FGF2 thermal stability. FGF2 (10 ng/ml), alone or after incubation with EVs (8E + 8p) from different cell types for 1 h on ice, was placed at 37 °C for 24 h to challenge FGF2 thermal stability. Residual FGF2 activity was determined by (a) proliferation assay by BrDU incorporation and (b) ELISA assay to measure ESM-1 secretion. EVs from DF, MSC, HEK cells were tested. Results are mean ± SE of four independent experiments. Statistical significance was determined by one-way ANOVA (Dunnett’s multiple test), *< 0.05, **< 0.01, ***< 0.001, ****< 0.0001. (B) Effect of EV binding on FGF2 degradation by trypsin. FGF2 (10 ng), alone or after incubation with EVs from DF, MSC or HEK cells (8E + 8p) for 1 h on ice, was held at 37 °C with trypsin/EDTA (0.05%, diluted 1:2 v/v in 25 μl) for different time periods before loading on western blot to assess residual FGF2 protein. (a) Representative blot with CTL-EVs (b) Representative blot and quantification of FGF2 signal obtained after 10 min incubation with EVs from DF, MSC or HEK cells. Results are mean ± SE of 3–5 independent experiments. Statistical significance was determined by one-way ANOVA (Dunnett’s multiple test), *< 0.05. Original blots are shown in Suppl. Information. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36550142), licensed under a CC-BY license. Not internally tested by R&D Systems.
 View Larger
View Larger
Detection of FGF basic/FGF2/bFGF by Western Blot Sulfatinib inhibits OS migration through suppression of epithelial–mesenchymal transition (EMT). (A) The protein levels of EMT and metastatic markers were detected by Western blotting after 24 h sulfatinib treatment. (B) Sulfatinib inhibits phosphorylation of migration-related signal pathway induced by bFGF. (C) Immunohistochemistry for EMT- related markers (FGFR1, p-FGFR1, N-cadherin, and E-cadherin) in tumor sections after treatment (scale bar, 100 μm). OS, osteosarcoma; bFGF, basic fibroblast growth factor. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37361567), licensed under a CC-BY license. Not internally tested by R&D Systems.
 View Larger
View Larger
Detection of FGF basic/FGF2/bFGF by Western Blot FGF2 is found at the outer surface of EVs secreted by dermal fibroblasts cultured in FGF2-containing medium. (A) FGF2, syntenin-1 and CD63 expression in DF cells and EV lysate (20 μg) was examined by western blot. Three isoforms of endogenous FGF2 were present in DF cells while FGF2-EVs contained the low molecular weight FGF2 isoform corresponding to recombinant FGF2. Original blots are shown in Suppl. Information. (B) Cells were cultured in the presence of His-tagged FGF2; western blot with 15 μg lysate and using anti-His antibody revealed the presence of His-FGF2 in secreted EVs. A shift in FGF2 size was also observed. Syntenin-1 was used as EV marker. Original blots are shown in Suppl. Information. (C) FGF2-EVs were loaded on a SEC column and collected fractions 8 and 9 (3E + 9p) were analyzed by western blot using antibodies for FGF2 and the EV markers CD63, CD81 and syntenin-1. Original blots are shown in Suppl. Information. For quantification, fractions were also analyzed by NTA. (D) FGF2 detection in FGF2-EVs by ELISA assay. Intact CTL- and FGF2-EVs (8E + 8p) were directly placed on FGF2 ELISA wells for external surface detection. FGF2-EVs were also treated with 0.1% triton and analyzed to detect internal FGF2. No FGF2 signal was detected in CTL-EVs. (E) FGF2 expression on FGF2-EVs was detected by flow cytometry. EVs were coupled to latex beads and labeled with FGF2 antibody before analysis by cytometry. Beads without EVs (beads) and beads labelled with goat IgG antibody (IgG) serve as negative controls. A representative plot is shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36550142), licensed under a CC-BY license. Not internally tested by R&D Systems.
 View Larger
View Larger
Detection of FGF basic/FGF2/bFGF by Western Blot FGF2 is found at the outer surface of EVs secreted by dermal fibroblasts cultured in FGF2-containing medium. (A) FGF2, syntenin-1 and CD63 expression in DF cells and EV lysate (20 μg) was examined by western blot. Three isoforms of endogenous FGF2 were present in DF cells while FGF2-EVs contained the low molecular weight FGF2 isoform corresponding to recombinant FGF2. Original blots are shown in Suppl. Information. (B) Cells were cultured in the presence of His-tagged FGF2; western blot with 15 μg lysate and using anti-His antibody revealed the presence of His-FGF2 in secreted EVs. A shift in FGF2 size was also observed. Syntenin-1 was used as EV marker. Original blots are shown in Suppl. Information. (C) FGF2-EVs were loaded on a SEC column and collected fractions 8 and 9 (3E + 9p) were analyzed by western blot using antibodies for FGF2 and the EV markers CD63, CD81 and syntenin-1. Original blots are shown in Suppl. Information. For quantification, fractions were also analyzed by NTA. (D) FGF2 detection in FGF2-EVs by ELISA assay. Intact CTL- and FGF2-EVs (8E + 8p) were directly placed on FGF2 ELISA wells for external surface detection. FGF2-EVs were also treated with 0.1% triton and analyzed to detect internal FGF2. No FGF2 signal was detected in CTL-EVs. (E) FGF2 expression on FGF2-EVs was detected by flow cytometry. EVs were coupled to latex beads and labeled with FGF2 antibody before analysis by cytometry. Beads without EVs (beads) and beads labelled with goat IgG antibody (IgG) serve as negative controls. A representative plot is shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36550142), licensed under a CC-BY license. Not internally tested by R&D Systems.
 View Larger
View Larger
Detection of FGF basic/FGF2/bFGF by Western Blot EV-bound FGF2 is protected from degradation. (A) Effect of EV binding on FGF2 thermal stability. FGF2 (10 ng/ml), alone or after incubation with EVs (8E + 8p) from different cell types for 1 h on ice, was placed at 37 °C for 24 h to challenge FGF2 thermal stability. Residual FGF2 activity was determined by (a) proliferation assay by BrDU incorporation and (b) ELISA assay to measure ESM-1 secretion. EVs from DF, MSC, HEK cells were tested. Results are mean ± SE of four independent experiments. Statistical significance was determined by one-way ANOVA (Dunnett’s multiple test), *< 0.05, **< 0.01, ***< 0.001, ****< 0.0001. (B) Effect of EV binding on FGF2 degradation by trypsin. FGF2 (10 ng), alone or after incubation with EVs from DF, MSC or HEK cells (8E + 8p) for 1 h on ice, was held at 37 °C with trypsin/EDTA (0.05%, diluted 1:2 v/v in 25 μl) for different time periods before loading on western blot to assess residual FGF2 protein. (a) Representative blot with CTL-EVs (b) Representative blot and quantification of FGF2 signal obtained after 10 min incubation with EVs from DF, MSC or HEK cells. Results are mean ± SE of 3–5 independent experiments. Statistical significance was determined by one-way ANOVA (Dunnett’s multiple test), *< 0.05. Original blots are shown in Suppl. Information. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36550142), licensed under a CC-BY license. Not internally tested by R&D Systems.
 View Larger
View Larger
Detection of FGF basic/FGF2/bFGF by Western Blot FGF2 is found at the outer surface of EVs secreted by dermal fibroblasts cultured in FGF2-containing medium. (A) FGF2, syntenin-1 and CD63 expression in DF cells and EV lysate (20 μg) was examined by western blot. Three isoforms of endogenous FGF2 were present in DF cells while FGF2-EVs contained the low molecular weight FGF2 isoform corresponding to recombinant FGF2. Original blots are shown in Suppl. Information. (B) Cells were cultured in the presence of His-tagged FGF2; western blot with 15 μg lysate and using anti-His antibody revealed the presence of His-FGF2 in secreted EVs. A shift in FGF2 size was also observed. Syntenin-1 was used as EV marker. Original blots are shown in Suppl. Information. (C) FGF2-EVs were loaded on a SEC column and collected fractions 8 and 9 (3E + 9p) were analyzed by western blot using antibodies for FGF2 and the EV markers CD63, CD81 and syntenin-1. Original blots are shown in Suppl. Information. For quantification, fractions were also analyzed by NTA. (D) FGF2 detection in FGF2-EVs by ELISA assay. Intact CTL- and FGF2-EVs (8E + 8p) were directly placed on FGF2 ELISA wells for external surface detection. FGF2-EVs were also treated with 0.1% triton and analyzed to detect internal FGF2. No FGF2 signal was detected in CTL-EVs. (E) FGF2 expression on FGF2-EVs was detected by flow cytometry. EVs were coupled to latex beads and labeled with FGF2 antibody before analysis by cytometry. Beads without EVs (beads) and beads labelled with goat IgG antibody (IgG) serve as negative controls. A representative plot is shown. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36550142), licensed under a CC-BY license. Not internally tested by R&D Systems.
Reconstitution Calculator
Preparation and Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Background: FGF basic/FGF2/bFGF
FGF basic is a member of the FGF family of at least 23 related mitogenic proteins which show 35‑60% amino acid conservation. FGF acidic and basic, unlike the other members of the family, lack signal peptides and are apparently secreted by mechanisms other than the classical protein secretion pathway. FGF basic has been isolated from a number of sources, including neural tissue, pituitary, adrenal cortex, corpus luteum, and placenta. This factor contains four cysteine residues, but reduced FGF basic retains full biological activity, indicating that disulfide bonds are not required for this activity. A variety of forms of FGF basic are produced as a result of N-terminal extensions. These extensions affect localization of FGF basic in cellular compartments but do not affect biological activity. Binding of FGF to heparin or cell surface heparan sulfate proteoglycans is necessary for binding of FGF to high affinity FGF receptors. FGF acidic and basic appear to bind to the same high affinity receptors and show a similar range of biological activities. FGF basic stimulates the proliferation of all cells of mesodermal origin and many cells of neuroectodermal, ectodermal, and endodermal origin. FGF basic induces neuron differentiation, survival, and regeneration. FGF basic also modulates embryonic development and differentiation. These observed in vitro functions of FGF basic suggest FGF basic may play a role in vivo in the modulation of such normal processes as angiogenesis, wound healing and tissue repair, embryonic development and differentiation, and neuronal function and neural degeneration. Additionally, FGF basic may participate in the production of a variety of pathological conditions resulting from excessive cell proliferation and excessive angiogenesis.
- Coulier, F. et al. (1997) J. Mol. Evol. 44:43.
- Chen, C.H. et al. (2004) Curr. Vasc. Pharmacol. 2:33.
- Mohammadi, M. et al. (2005) Curr. Opin. Struct. Biol. 15:506.
- Fernig, D. et al. (1994) Prog. Growth Factor Res. 5:353.
Product Datasheets
Citations for Human FGF basic/FGF2/bFGF Antibody
R&D Systems personnel manually curate a database that contains references using R&D Systems products. The data collected includes not only links to publications in PubMed, but also provides information about sample types, species, and experimental conditions.
28
Citations: Showing 1 - 10
Filter your results:
Filter by:
-
Small Extracellular Vesicle‐Derived vWF Induces a Positive Feedback Loop between Tumor and Endothelial Cells to Promote Angiogenesis and Metastasis in Hepatocellular Carcinoma
Authors: Samuel Wan Ki Wong, Sze Keong Tey, Xiaowen Mao, Hiu Ling Fung, Zhi‐Jie Xiao, Danny Ka Ho Wong et al.
Advanced Science
Species: Human
Sample Types: Whole Cells
Applications: Neutralization -
XBP1‐elicited environment by chemotherapy potentiates repopulation of tongue cancer cells by enhancing miR‐22/lncRNA/KAT6B‐dependent NF‐ kappa B signalling
Authors: Xiaoting Jia, Ge Wang, Lihong Wu, Hao Pan, Li Ling, Jianlei Zhang et al.
Clinical and Translational Medicine
-
Fibroblast growth factor-2 bound to specific dermal fibroblast-derived extracellular vesicles is protected from degradation
Authors: I Petit, A Levy, S Estrach, CC Féral, AG Trentin, F Dingli, D Loew, J Qu, H Zhou, C Théry, C Prunier, D Aberdam, O Ferrigno
Scientific Reports, 2022-12-22;12(1):22131.
Species: Human
Sample Types: Cell Lysates, Whole Cells
Applications: Bioassay, Flow Cytometry, Western Blot -
Heme stimulates platelet mitochondrial oxidant production to induce targeted granule secretion
Authors: GK Annarapu, D Nolfi-Done, M Reynolds, Y Wang, L Kohut, B Zuckerbrau, S Shiva
Redox Biology, 2021-12-05;48(0):102205.
Species: Human
Sample Types: Cell Culture Supernates
Applications: Dot Blot -
Three subtypes of lung cancer fibroblasts define distinct therapeutic paradigms.
Authors: Haichuan H, Zofia P, Patricia H et al.
Cancer Cell.
-
A digital single-molecule nanopillar SERS platform for predicting and monitoring immune toxicities in immunotherapy
Authors: J Li, A Wuethrich, AAI Sina, HH Cheng, Y Wang, A Behren, PN Mainwaring, M Trau
Nature Communications, 2021-02-17;12(1):1087.
Species: Human
Sample Types: Serum
Applications: ELISA Capture -
Inhibition of protein glycosylation is a novel pro-angiogenic strategy that acts via activation of stress pathways
Authors: C Zhong, P Li, S Argade, L Liu, A Chilla', W Liang, H Xin, B Eliceiri, B Choudhury, N Ferrara
Nature Communications, 2020-12-10;11(1):6330.
Species: Mouse
Sample Types: In Vivo
Applications: In Vivo -
Metastatic Brain Tumors Disrupt the Blood-Brain Barrier and Alter Lipid Metabolism by Inhibiting Expression of the Endothelial Cell Fatty Acid Transporter Mfsd2a
Authors: S Tiwary, JE Morales, SC Kwiatkowsk, FF Lang, G Rao, JH McCarty
Sci Rep, 2018-05-29;8(1):8267.
Species: Human
Sample Types: Whole Cells
Applications: Neutralization -
Multicolor quantitative confocal imaging cytometry
Authors: DL Coutu, KD Kokkaliari, L Kunz, T Schroeder
Nat. Methods, 2017-11-13;15(1):39-46.
Species: Mouse
Sample Types: Whole Tissue
Applications: IHC -
Tumor-associated B-cells induce tumor heterogeneity and therapy resistance
Authors: R Somasundar, G Zhang, M Fukunaga-K, M Perego, C Krepler, X Xu, C Wagner, D Hristova, J Zhang, T Tian, Z Wei, Q Liu, K Garg, J Griss, R Hards, M Maurer, C Hafner, M Mayerhöfer, G Karanikas, A Jalili, V Bauer-Pohl, F Weihsengru, K Rappersber, J Koller, R Lang, C Hudgens, G Chen, M Tetzlaff, L Wu, DT Frederick, RA Scolyer, GV Long, M Damle, C Ellingswor, L Grinman, H Choi, BJ Gavin, M Dunagin, A Raj, N Scholler, L Gross, M Beqiri, K Bennett, I Watson, H Schaider, MA Davies, J Wargo, BJ Czerniecki, L Schuchter, D Herlyn, K Flaherty, M Herlyn, SN Wagner
Nat Commun, 2017-09-19;8(1):607.
Species: Human
Sample Types: Whole Cells
Applications: Neutralization -
Pericytes impair capillary blood flow and motor function after chronic spinal cord injury
Authors: Y Li, AM Lucas-Osma, S Black, MV Bandet, MJ Stephens, R Vavrek, L Sanelli, KK Fenrich, AF Di Narzo, S Dracheva, IR Winship, K Fouad, DJ Bennett
Nat. Med., 2017-05-01;0(0):.
Species: Rat
Sample Types: Whole Tissue
Applications: IHC -
Discovery of pan-VEGF inhibitory peptides directed to the extracellular ligand-binding domains of the VEGF receptors
Authors: Jussara S. Michaloski, Alexandre R. Redondo, Leila S. Magalhães, Caio C. Cambui, Ricardo J. Giordano
Science Advances
-
Stearoyl-CoA desaturase 1 and paracrine diffusible signals have a major role in the promotion of breast cancer cell migration induced by cancer-associated fibroblasts.
Authors: Angelucci C, Maulucci G, Colabianchi A, Iacopino F, D'Alessio A, Maiorana A, Palmieri V, Papi M, De Spirito M, Di Leone A, Masetti R, Sica G
Br J Cancer, 2015-04-16;112(10):1675-86.
Species: Human
Sample Types: Whole Cells
Applications: Bioassay -
Peroxynitrite upregulates angiogenic factors VEGF-A, BFGF, and HIF-1alpha in human corneal limbal epithelial cells.
Authors: Ashki N, Chan A, Qin Y, Wang W, Kiyohara M, Lin L, Braun J, Wadehra M, Gordon L
Invest Ophthalmol Vis Sci, 2014-03-19;55(3):1637-46.
Species: Human
Sample Types: Cell Lysates
Applications: Western Blot -
Endothelial heparan sulfate 6-o-sulfation levels regulate angiogenic responses of endothelial cells to fibroblast growth factor 2 and vascular endothelial growth factor.
Authors: Ferreras, Cristina, Rushton, Graham, Cole, Claire L, Babur, Muhammad, Telfer, Brian A, van Kuppevelt, Toin H, Gardiner, John M, Williams, Kaye J, Jayson, Gordon C, Avizienyte, Egle
J Biol Chem, 2012-08-27;287(43):36132-46.
Species: Human
Sample Types: Whole Tissue
Applications: IHC-Fr -
Inhibition of PAI-1 induces neutrophil-driven neoangiogenesis and promotes tissue regeneration via production of angiocrine factors in mice.
Blood, 2012-05-09;119(26):6382-93.
Species: Mouse
Sample Types: In Vivo
Applications: In Vivo -
Bone marrow stroma-secreted cytokines protect JAK2(V617F)-mutated cells from the effects of a JAK2 inhibitor
Authors: Taghi Manshouri, Zeev Estrov, Alfonso Quintás-Cardama, Jan Burger, Ying Zhang, Ana Livun et al.
Cancer Research
-
The fibroblast-derived paracrine factor neuregulin-1 has a novel role in regulating the constitutive color and melanocyte function in human skin.
Authors: Choi W, Wolber R, Gerwat W
J. Cell. Sci., 2010-08-24;123(0):3102-11.
Species: Human
Sample Types: Cell Lysates
Applications: Western Blot -
IFATS Series: FGF-2-induced HGF Secretion By Adipose-Derived Stromal Cells Inhibits Post-Injury Fibrogenesis Through A JNK-Dependent Mechanism.
Authors: Suga H, Eto H, Shigeura T, Inoue K, Aoi N, Kato H, Nishimura S, Manabe I, Gonda K, Yoshimura K
Stem Cells, 2009-01-01;0(0):.
Species: Mouse
Sample Types: In Vivo, Tissue Homogenates, Whole Tissue
Applications: IHC-P, Neutralization, Western Blot -
A CXCL5- and bFGF-dependent effect of PDGF-B-activated fibroblasts in promoting trafficking and differentiation of bone marrow-derived mesenchymal stem cells.
Authors: Nedeau AE, Bauer RJ, Gallagher K, Chen H, Liu ZJ, Velazquez OC
Exp. Cell Res., 2008-04-25;314(11):2176-86.
Species: Mouse
Sample Types: Whole Cells
Applications: Neutralization -
Temporally expressed PDGF and FGF-2 regulate embryonic coronary artery formation and growth.
Authors: Tomanek RJ, Hansen HK, Christensen LP
Arterioscler. Thromb. Vasc. Biol., 2008-04-17;28(7):1237-43.
Species: Avian
Sample Types: Whole Tissue
Applications: Neutralization -
Pancreatic stellate cells: partners in crime with pancreatic cancer cells.
Authors: Vonlaufen A, Joshi S, Qu C, Phillips PA, Xu Z, Parker NR, Toi CS, Pirola RC, Wilson JS, Goldstein D, Apte MV
Cancer Res., 2008-04-01;68(7):2085-93.
Species: Human
Sample Types: Whole Cells
Applications: Neutralization -
Agrin, a novel basement membrane component in human and rat liver, accumulates in cirrhosis and hepatocellular carcinoma.
Authors: Tatrai P, Dudas J, Batmunkh E, Máthé M, Zalatnai A, Schaff Z, Ramadori G, Kovalszky I
Lab. Invest., 2006-09-18;86(11):1149-60.
Species: Human
Sample Types: Whole Tissue
Applications: IHC-Fr -
Positive correlation between estradiol and vascular endothelial growth factor but not fibroblast growth factor-2 in normal human breast tissue in vivo.
Authors: Dabrosin C
Clin. Cancer Res., 2005-11-15;11(22):8036-41.
Species: Human
Sample Types: Whole Tissue
Applications: IHC-P -
FGFR2b signaling regulates ex vivo submandibular gland epithelial cell proliferation and branching morphogenesis.
Authors: Steinberg Z, Myers C, Heim VM, Lathrop CA, Rebustini IT, Stewart JS, Larsen M, Hoffman MP
Development, 2005-02-16;132(6):1223-34.
Species: Mouse
Sample Types: Whole Cells
Applications: Neutralization -
Differential role of platelet granular mediators in angiogenesis.
Authors: Brill A, Elinav H, Varon D
Cardiovasc. Res., 2004-08-01;63(2):226-35.
Species: Bovine
Sample Types: Whole Cells
Applications: Neutralization -
Proangiogenic properties of human myeloma cells: production of angiopoietin-1 and its potential relationship to myeloma-induced angiogenesis.
Authors: Giuliani N, Colla S, Lazzaretti M, Sala R, Roti G, Mancini C, Bonomini S, Lunghi P, Hojden M, Genestreti G, Svaldi M, Coser P, Fattori PP, Sammarelli G, Gazzola GC, Almici C, Caramatti C, Mangoni L, Rizzoli V
Blood, 2003-03-20;102(2):638-45.
Species: Human
Sample Types: Whole Cells
Applications: Neutralization -
The anti-HIV cytokine midkine binds the cell surface-expressed nucleolin as a low affinity receptor.
Authors: Said EA, Krust B, Nisole S, Svab J, Briand JP, Hovanessian AG
J. Biol. Chem., 2002-07-29;277(40):37492-502.
Species: Hamster
Sample Types: Whole Cells
Applications: ICC
FAQs
No product specific FAQs exist for this product, however you may
View all Antibody FAQsReviews for Human FGF basic/FGF2/bFGF Antibody
There are currently no reviews for this product. Be the first to review Human FGF basic/FGF2/bFGF Antibody and earn rewards!
Have you used Human FGF basic/FGF2/bFGF Antibody?
Submit a review and receive an Amazon gift card.
$25/€18/£15/$25CAN/¥75 Yuan/¥2500 Yen for a review with an image
$10/€7/£6/$10 CAD/¥70 Yuan/¥1110 Yen for a review without an image



