FRAT2 (Frequently Rearranged in Advanced T-cell lymphomas-2; also GSK-3-binding protein FRAT2) is an intracellular member of the GSK-3-binding protein family. Although its predicted MW is 28 kDa, due to its highly acidic nature, it runs anomalously at 35 kDa in SDS-PAGE. It is widely expressed, and plays a key role in the regulation of the Wnt signaling pathway. Typically, in the absence of stimulation, GSK-3 beta and beta -catenin associate with Axin, where GSK-3 beta phosphorylates beta -catenin, leading to its turnover. Upon Wnt stimulation, GSK-3 beta phosphorylation of beta -catenin is decoupled, resulting in beta -catenin mediated gene activation. FRAT2 has the same effect as Wnt stimulation on GSK-3 beta activity, serving as a non-Wnt activator of beta -catenin. It does so by promoting the dissociation of GSK-3 beta from axin. It also interacts with Diversin, a participant in JNK signaling. This suggests that FRAT2 plays a role in both Wnt/ beta -catenin and PCP signaling pathways. Human FRAT2 is 233 amino acids (aa) in length. It contains an N-terminal acidic domain (aa 1-54), a Pro-rich segment (aa 60-107) and a GSK-3 beta binding region (aa 171-196). Over aa 31‑233, human FRAT2 shares 73% aa sequence identity with both mouse FRAT2 and human FRAT1.

Key Product Details
Species Reactivity
Human
Applications
Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human FRAT2
Ser31-Leu233 (Ala83Thr)
Accession # O75474
Ser31-Leu233 (Ala83Thr)
Accession # O75474
Specificity
Detects human FRAT2 in direct ELISAs.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Human FRAT2 Antibody
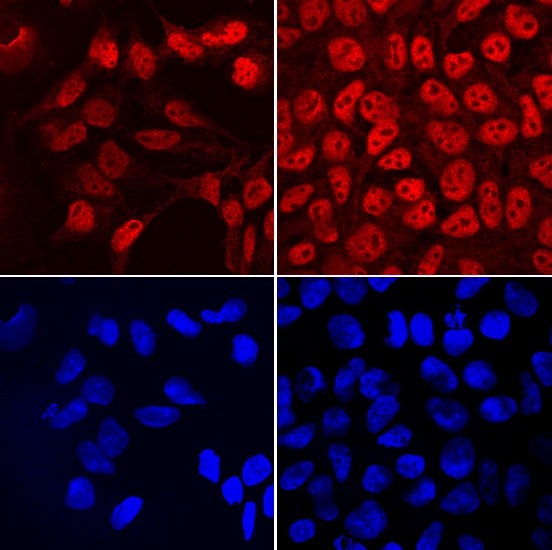
FRAT2 in NTera‑2 Human Cell Line.
FRAT2 was detected in immersion fixed NTera-2 human testicular embryonic carcinoma cell line cultured with (left panels) and without (right panels) 10µM retinoic acid for 4 days using Sheep Anti-Human FRAT2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF7980) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Sheep IgG Secondary Antibody (red, upper panels; Catalog # NL010) and counterstained with DAPI (blue, lower panels). Specific staining was localized to nuclei. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Applications for Human FRAT2 Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed NTera‑2 human testicular embryonic carcinoma cells with 10μM retinoic acid for 4 days
Sample: Immersion fixed NTera‑2 human testicular embryonic carcinoma cells with 10μM retinoic acid for 4 days
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: FRAT2
Long Name
Frequently Rearranged in Advanced T-cell Lymphomas 2
Alternate Names
frequently rearranged in advanced T-cell lymphomas 2FRAT-2, GSK-3 binding protein FRAT2, GSK-3-binding protein FRAT2, MGC10562
Gene Symbol
FRAT2
UniProt
Additional FRAT2 Products
Product Documents for Human FRAT2 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human FRAT2 Antibody
For research use only
Related Research Areas
Customer Reviews for Human FRAT2 Antibody
There are currently no reviews for this product. Be the first to review Human FRAT2 Antibody and earn rewards!
Have you used Human FRAT2 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
