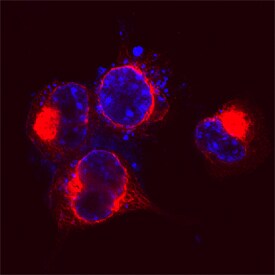
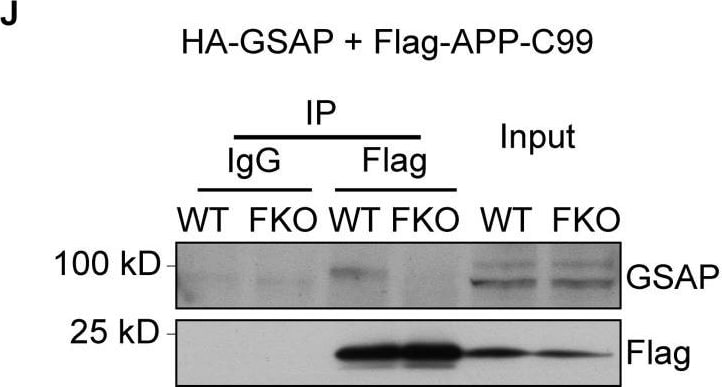
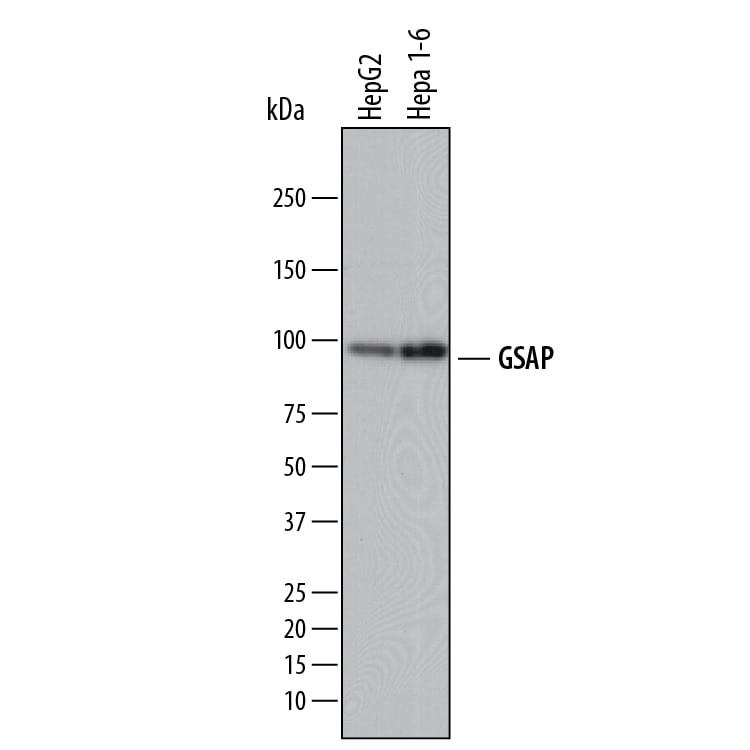
GSAP (GSAP/gamma-Secretase Activating Protein; also known is Pion) is a member of the GSAP family of proteins. It is expressed in neurons, and purportedly serves as a regulator for g-secretase processing of APP. Initially, it was thought that cytosolic g-secretase mediated the second cleavage step in APP processing. In the presence of GSAP, APP (now C99 after b-secretase cleavage) was preferentially cleaved between either Val40-Ile41, or Ala42-Thr43, generating Ab40 and Ab42, respectively. In the absence of GSAP, APP would be preferentially cleaved between L49-Val50. Notably, the presence of GSAP was shown to have no effect on g-secretase processing of Notch. Subsequent studies have introduced uncertainty into these relationships. While an absence of GSAP does apparently reduce Ab production, its presence may not have the regulatory effect once proposed. Mouse GSAP-16K (15-17 kDa) is 121 amino acids (aa) in length (aa 738-858) (SwissProt #:Q3TCV3), it presumably represents a proteolytic cleavage product of the large 95-100 kDa, 858 aa GSAP-FL termed also Pion/pigeon homolog protein. There are no readily identifiable structural motifs associated with the molecule. Mouse PION has two isoform variants associated with the gene. One possesses a Phe substitution for aa 172-858, while another possesses a six aa substitution for aa 527-858. Over aa 737-858, mouse GSAP shares 94% and 88% aa sequence identity with rat and human GSAP, respectively.