Ephrin-B2, also known as Htk-L, ELF-2, LERK-5, and NLERK-1 (1), is a member of the ephrin ligand family which binds members of the Eph receptor family. All ligands share a conserved extracellular sequence, which most likely corresponds to the receptor binding domain. This conserved sequence consists of approximately 125 amino acids and includes four invariant cysteines. The B-class ligands are transmembrane proteins which can become tyrosine phosphorylated upon receptor ligation. The cytoplasmic domains are approximately 80 amino acids long and are highly conserved, especially the last 33 amino acids. Several signaling molecules have been shown to interact with the cytoplasmic region, although specific signaling roles have yet to be elucidated. Ephrin-B2 has been shown to bind EphA4, EphB1, EphB2, EphB3, and EphB4 (2, 3). The extracellular domains of murine and human Ephrin-B2 share 98% amino acid identity. Only membrane-bound or Fc-clustered ligands are capable of activating the receptor in vitro. While soluble monomeric ligands bind the receptor, they do not induce receptor autophosphorylation and activation (2). In vivo, the ligands and receptors display reciprocal expression (3). It has been found that nearly all the receptors and ligands are expressed in developing and adult neural tissue (3). The Ephrin/Eph families also appear to play a role in angiogenesis (3).
Human/Mouse/Rat Ephrin‑B2 Antibody
R&D Systems | Catalog # AF496

Key Product Details
Species Reactivity
Validated:
Human, Mouse, Rat
Cited:
Human, Mouse, Rat, Frog - Xenopus (African Clawed Frog), Transgenic Mouse
Applications
Validated:
Western Blot, Flow Cytometry, Immunocytochemistry, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Flow Cytometry, Immunofluorescence, Immunocytochemistry, Immunoprecipitation, Functional Assay, Proximity Ligation Assay (PLA)
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse Ephrin-B2
Arg27-Ala227
Accession # AAA82934
Arg27-Ala227
Accession # AAA82934
Specificity
Detects mouse Ephrin-B2 in direct ELISAs and Western blots. In direct ELISAs and Western blots, approximately 5% cross-reactivity with recombinant mouse Ephrin-B1 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human/Mouse/Rat Ephrin‑B2 Antibody
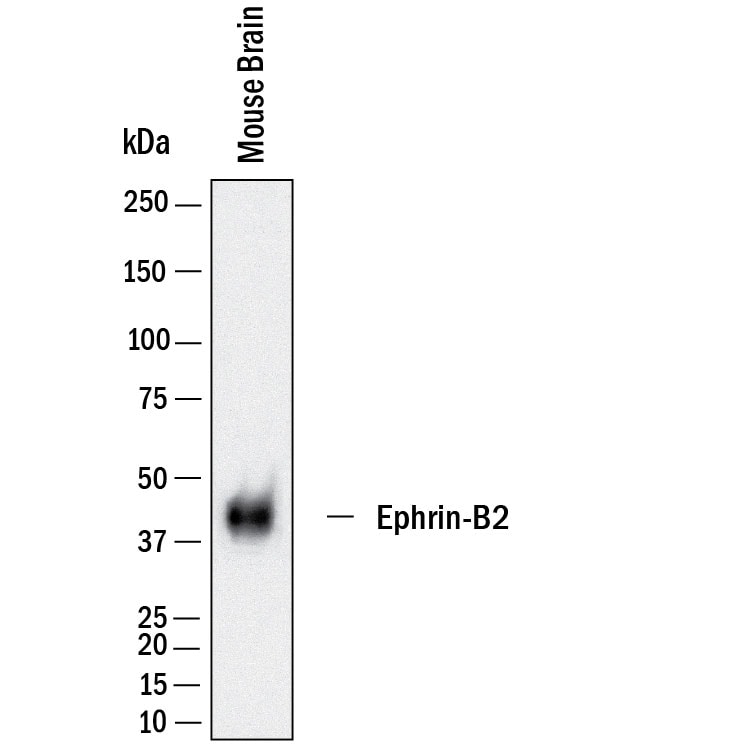
Detection of Mouse Ephrin‑B2 by Western Blot.
Western blot shows lysates of mouse brain tissue. PVDF membrane was probed with 2 µg/mL of Goat Anti-Human/Mouse/Rat Ephrin-B2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF496) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF019). Specific bands were detected for Ephrin-B2 at approximately 40-45 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.Detection of Ephrin‑B2 in SH‑SY5Y Human Cell Line by Flow Cytometry.
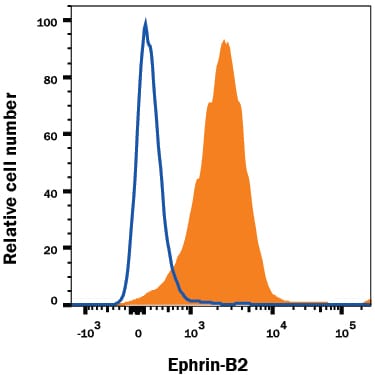
SH-SY5Y human neuroblastoma cell line was stained with Goat Anti-Human/Mouse/Rat Ephrin-B2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF496, filled histogram) or isotype control antibody (AB-108-C, open histogram), followed by Phycoerythrin-conjugated Anti-Goat IgG Secondary Antibody (F0107).Ephrin‑B2 in Rat Hippo-campal Neurons.
Ephrin‑B2 was detected in immersion fixed rat hippocampal neurons using 2 µg/mL Goat Anti-Human/Mouse/Rat Ephrin‑B2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF496) for 3 hours at room temperature. Cells were stained (red) and counterstained (green). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Ephrin‑B2 in SH‑SY5Y Human Cell Line.
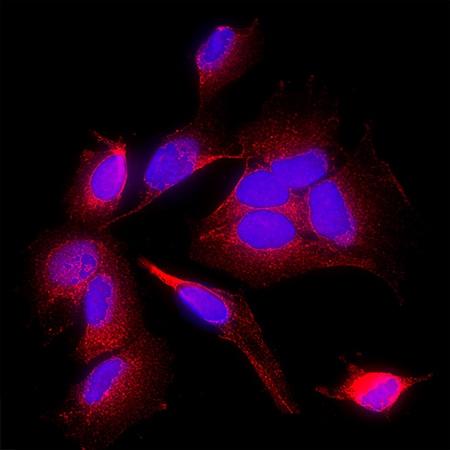
Ephrin‑B2 was detected in immersion fixed SH‑SY5Y human neuroblastoma cell line using Goat Anti-Human/Mouse/Rat Ephrin‑B2 Antigen Affinity-purified Polyclonal Antibody (Catalog # af496) at 15 µg/ml for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to the cell surface. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Ephrin-B2 by Western Blot
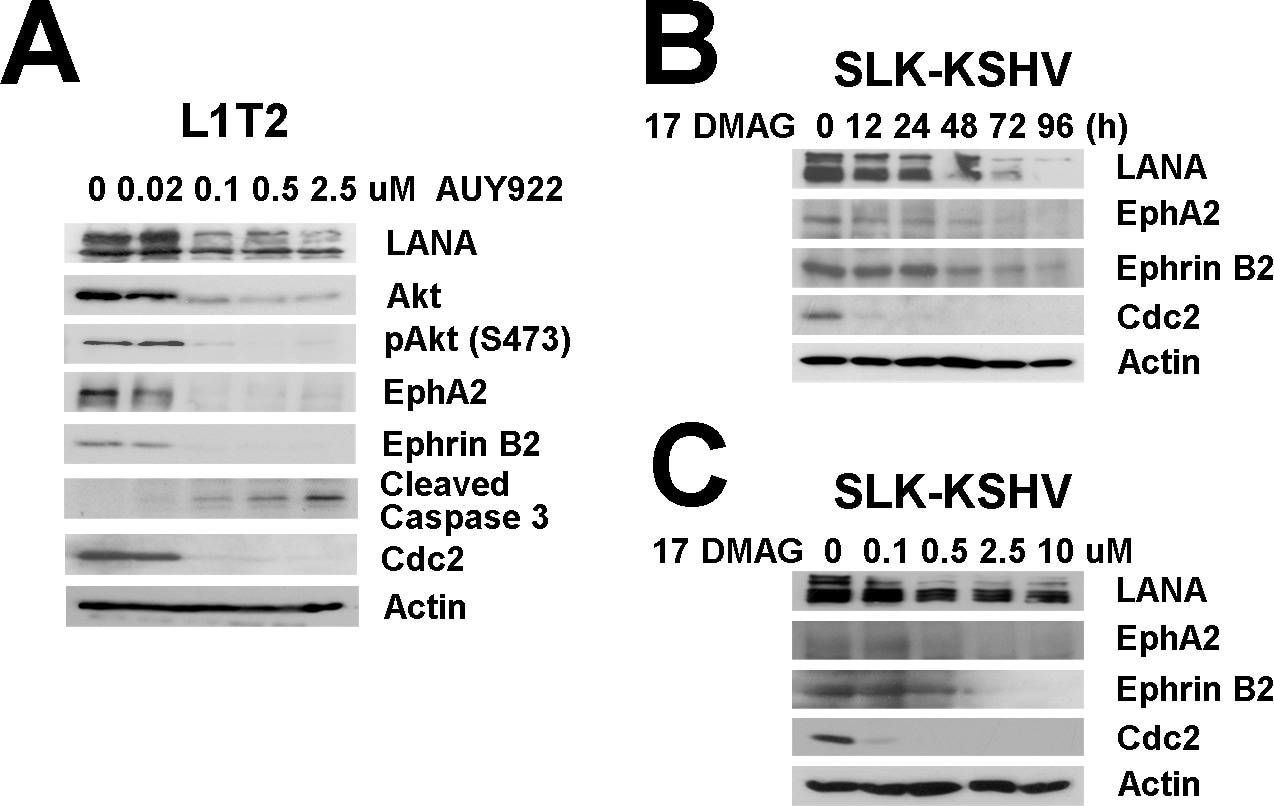
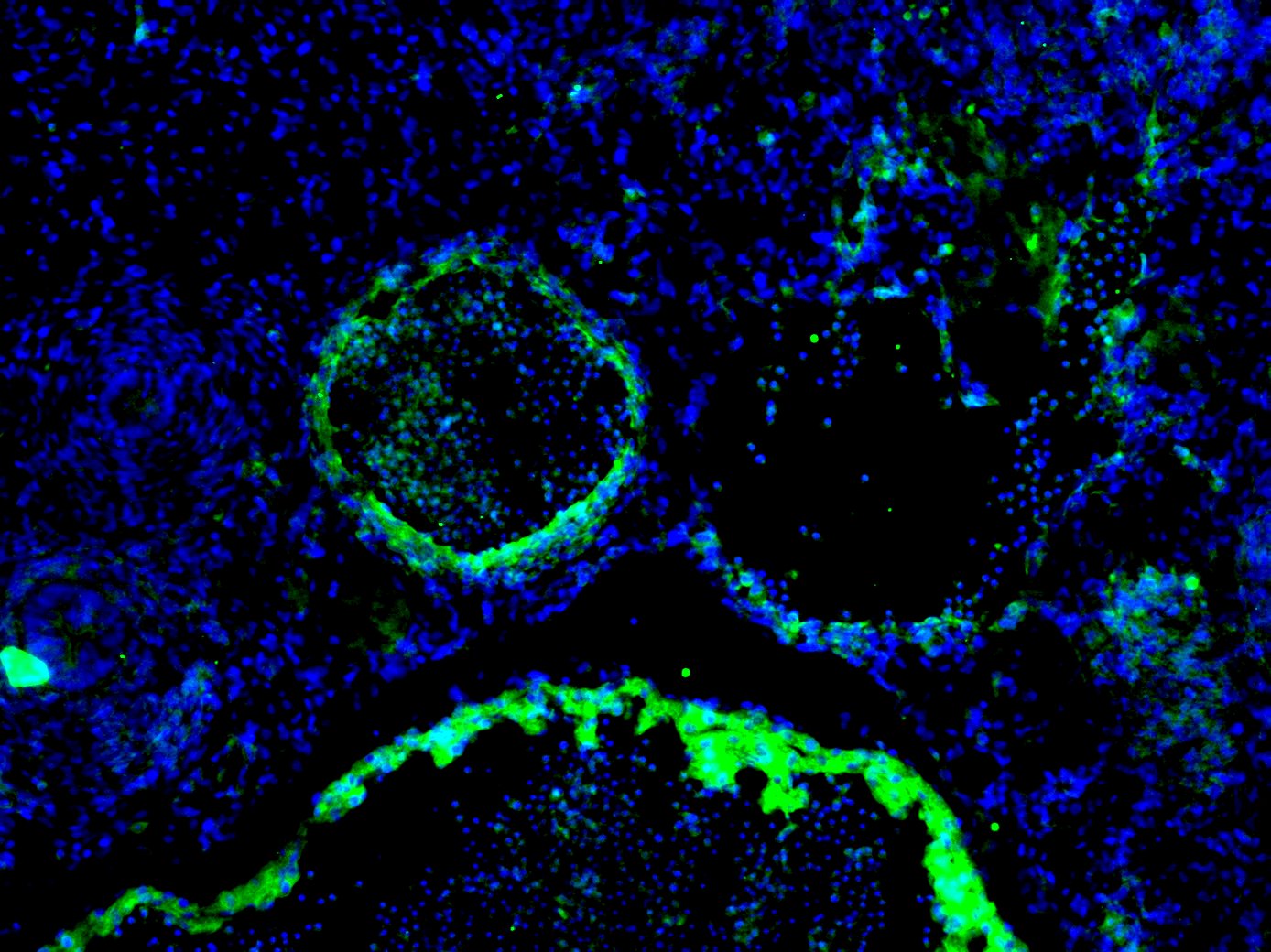
Effects of Hsp90 inhibitors on LANA in KSHV-infected endothelial cells.(A) L1T2 cells were treated with AUY922 at concentrations of 0, 0.02, 0.1, 0.5, and 2.5 µM for 48 hours, whole cell lysates were immunoblotted with anti-LANA, anti-Hsp90 antibodies, anti-EphA2, anti-Ephrin-B2, and anti-Akt (total), and anti-pAkt (S473) antibodies separately. Apoptosis was evaluated with anti-cleaved PARP and anti-cleaved Caspase-3 antibodies, Cdc2 and beta -Actin were used as controls. (B–C) SLK-KSHV were treated with 0.5 µM 17-DMAG for 0, 12, 24, 48, 72 and 96 hours, or at concentrations of 0, 0.1, 0.5, 2.5 and 10 µM for 48 hours separately, whole cell lysates were immunoblotted with anti-LANA antibody, anti-EphA2, anti-EphrinB2, Cdc2 and beta -Actin were used as controls. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/23209418), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Ephrin-B2 by Immunohistochemistry
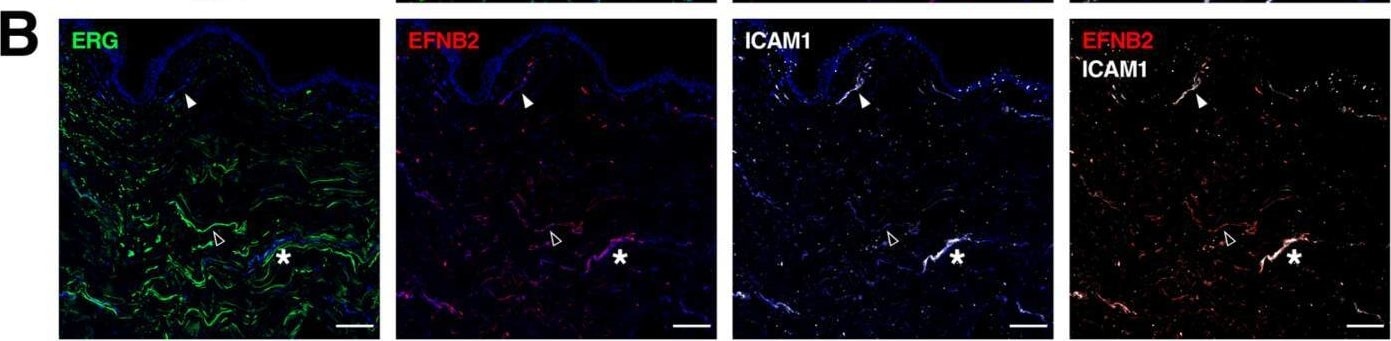
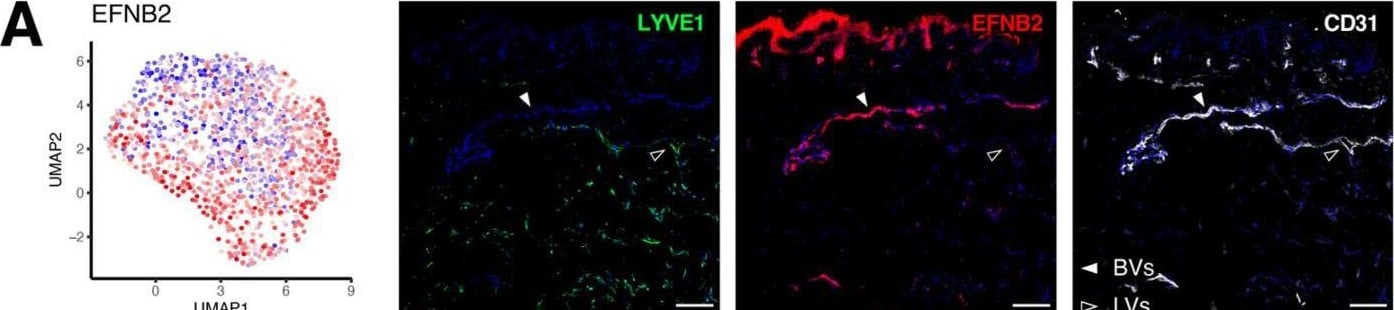
Gene signatures peculiar to the venular compartment. (A) EFNB2 expression measured by scRNA-seq (left) and immunofluorescence staining (right) denoting its abundance across different BEC subpopulations and its exclusive expression in human dermal BVs (white arrowheads). (B) Representative confocal images of skin sections (upper) and whole-mount skin blocks (lower) stained for ERG (green), EFNB2 (red) and ICAM1 (white). The unique composition of EFNB2 and ICAM1 expression enables the discrimination of arterioles (empty arrowheads), post-capillary venules (*) and venules (white arrowheads). (C) EGR2 expression is most enriched in the post-capillary venule cluster and is restricted to BVs (white arrowheads). (D) Representative section (upper) and whole-mount staining (lower) of ERG (green), EGR2 (red) and ICAM1 (white), highlighting EGR2+ICAM1+ BVs. Scale bars: 100 µm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35406678), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Ephrin-B2 by Immunohistochemistry
Gene signatures peculiar to the venular compartment. (A) EFNB2 expression measured by scRNA-seq (left) and immunofluorescence staining (right) denoting its abundance across different BEC subpopulations and its exclusive expression in human dermal BVs (white arrowheads). (B) Representative confocal images of skin sections (upper) and whole-mount skin blocks (lower) stained for ERG (green), EFNB2 (red) and ICAM1 (white). The unique composition of EFNB2 and ICAM1 expression enables the discrimination of arterioles (empty arrowheads), post-capillary venules (*) and venules (white arrowheads). (C) EGR2 expression is most enriched in the post-capillary venule cluster and is restricted to BVs (white arrowheads). (D) Representative section (upper) and whole-mount staining (lower) of ERG (green), EGR2 (red) and ICAM1 (white), highlighting EGR2+ICAM1+ BVs. Scale bars: 100 µm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35406678), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse/Rat Ephrin‑B2 Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
0.25 µg/106 cells
Sample: SH‑SY5Y human neuroblastoma cell line
Sample: SH‑SY5Y human neuroblastoma cell line
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed cultured rat embryonic hippocampal neurons and fixed SH-SY5Y human neuroblastoma cell line
Sample: Immersion fixed cultured rat embryonic hippocampal neurons and fixed SH-SY5Y human neuroblastoma cell line
Western Blot
2 µg/mL
Sample: Mouse brain tissue
Sample: Mouse brain tissue
Reviewed Applications
Read 3 reviews rated 4.3 using AF496 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Ephrin-B2
References
- Eph Nomenclature Committee [letter]. (1997) Cell 90:403.
- Flanagan, J.G. and P. Vanderhaeghen (1998) Annu. Rev. Neurosci. 21:309.
- Pasquale, E.B. (1997) Curr. Opin. Cell Biol. 9:608.
Alternate Names
EFNB2, ELF-2, EphrinB2, Htk-L, LERK-5, NLERK-1
Gene Symbol
EFNB2
UniProt
Additional Ephrin-B2 Products
Product Documents for Human/Mouse/Rat Ephrin‑B2 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse/Rat Ephrin‑B2 Antibody
For research use only
Related Research Areas
Citations for Human/Mouse/Rat Ephrin‑B2 Antibody
Customer Reviews for Human/Mouse/Rat Ephrin‑B2 Antibody (3)
4.3 out of 5
3 Customer Ratings
Have you used Human/Mouse/Rat Ephrin‑B2 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
3 of
3 reviews
Showing All
Filter By:
-
Application: Western BlotSample Tested: Cell Lysates and Cell culture supernatantSpecies: HumanVerified Customer | Posted 03/16/2021
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: E12.5 mouse embryo fixed in 4% PFASpecies: MouseVerified Customer | Posted 12/16/2020Dilution used - 1:200. The staining was done on an E12.5 transverse mouse section fixed overnight with 4% PFA. Stained using standard IF technique. Attached picture shows an artery and a vein and it is clearly seen that the artery seen is stained positive whereas the adjacent vein is negative for EphrinB2 (as it should be).
-
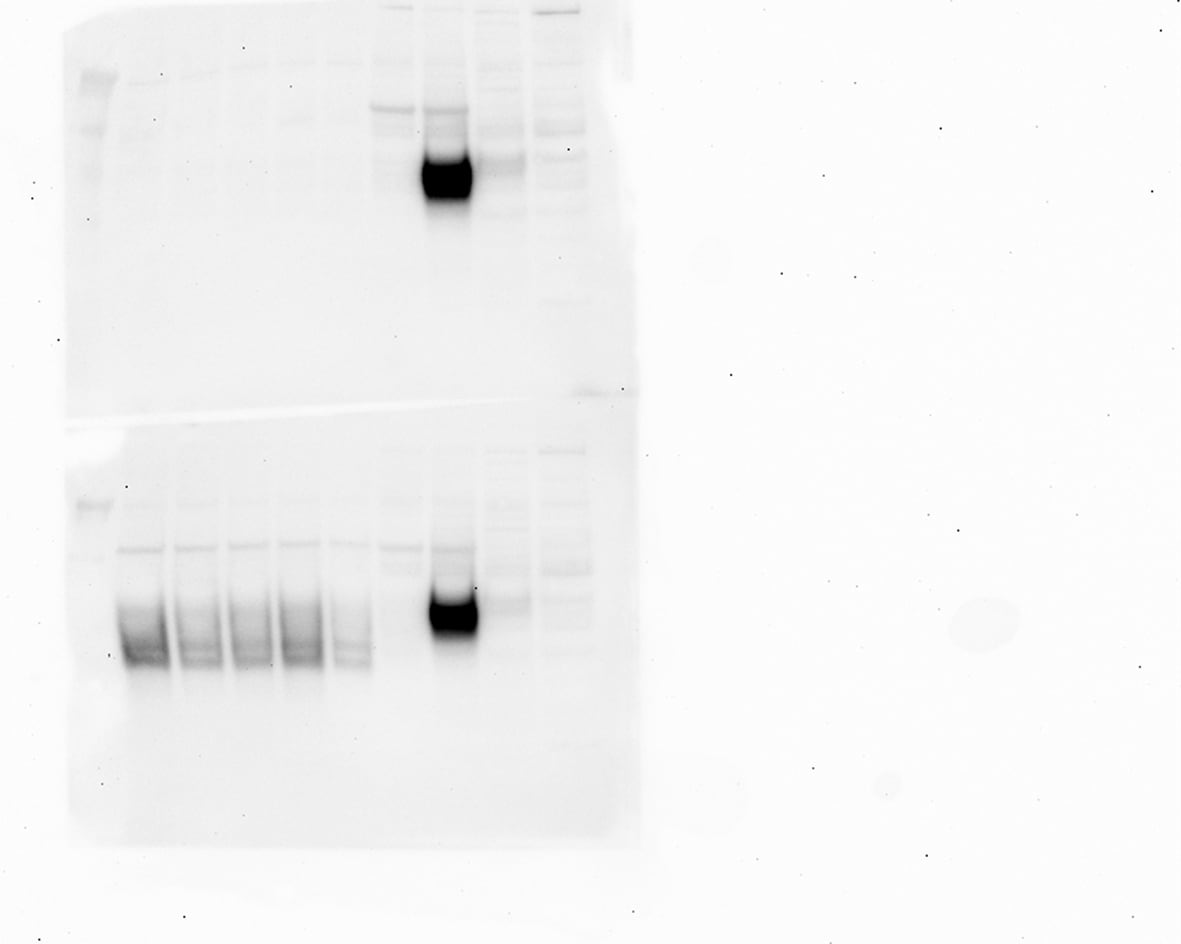
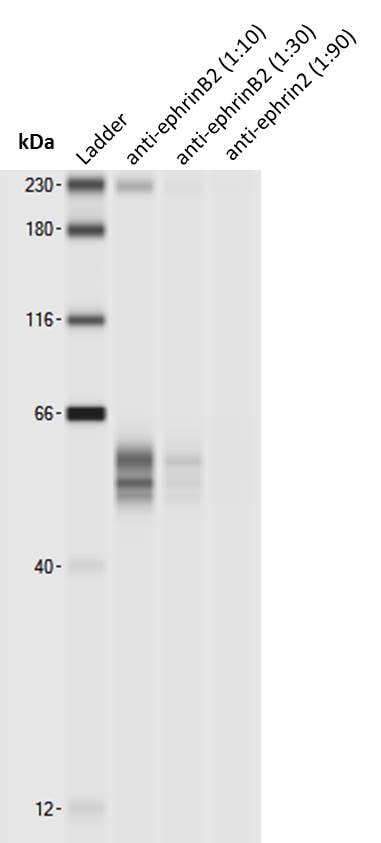
Application: Simple WesternSample Tested: mouse brain lysateSpecies: MouseVerified Customer | Posted 07/12/2017Simple Western lane view shows 53 and 58kDa signals for ephrinB2. Note that higher concentrations of this antibody may lead to an unspecific signal corresponding to the size of the internal standard (230 kDa, leftmost lane).Simple Western: anti-ephrinB2 antibody [biotechne AF496] Simple Western lane view shows 53 and 58kDa signals for ephrinB2 in 0.5 mg/ml of mouse brain lysate (lysis buffer from ARY014 proteome profiler kit). This run was performed under reducing conditions using the 12-230 kDa separation system in combination with a secondary anti-goat-HRP antibody (biotechne, HAF109, 1:50). Note that higher concentrations of this antibody may lead to an unspecific signal corresponding to the size of the internal standard (230 kDa, leftmost lane).
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...