SCD-1 (Stearoyl-CoA desaturase 1; also Acyl-CoA desaturase, fatty acid desaturase, and Delta-9 desaturase) is a 37-40 kDa member of the fatty acid desaturase family of enzymes. It is an ER-embedded protein that is expressed by multiple cell types, including adipocytes, hepatocytes, macrophages, endothelial and sebaceous gland cells. SCD-1 catalyzes the formation of monounsaturated fatty acids from saturated fatty acids. It does so by generating a double bond between the C9 and C10 carbons of dietary and/or endogenously synthesized fatty acids. This creates either palmitoleic or oleic acid, two fatty acids that are optimally suited for either storage or inclusion into phospholipids. It also removes a potential source of inflammation, as saturated fatty acids are known to activate TLRs with the subsequent onset of inflammation. Human SCD-1 is a 4-transmembrane (TM), 359 amino acid (aa) protein. It contains a 71 aa cytoplasmic N-terminus, followed by two TM segments (aa 72-119) and an extended cytoplasmic region (aa 120-216) that possesses three utilized Ser/Thr phosphorylation sites, two additional TM segments (aa 217-273), and a C-terminal cytoplasmic tail (aa 274-359) that contains most of the catalytic region. There is one potential isoform variant that shows a 13 aa substitution for aa 295-359. Over aa 141-221, human SCD-1 shares 95% aa sequence identity with mouse SCD-1.

Key Product Details
Validated by
Knockout/Knockdown, Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Knockout Validated, Western Blot, Immunoprecipitation
Cited:
Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human SCD-1
Ala141-Gly221
Accession # O00767
Ala141-Gly221
Accession # O00767
Specificity
Detects human SCD-1 in direct ELISAs and Western blots. In direct ELISAs, less than 5% cross-reactivity with recombinant human SCD-5 and recombinant mouse SCD-1 is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Human SCD‑1 Antibody
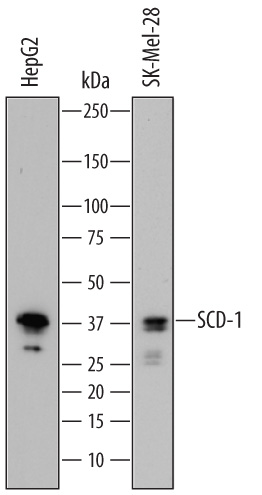
Detection of Human SCD‑1 by Western Blot.
Western blot shows lysates of HepG2 human hepatocellular carcinoma cell line and SK-Mel-28 human malignant melanoma cell line. PVDF membrane was probed with 0.5 µg/mL of Sheep Anti-Human SCD-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF7550) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (HAF016). A specific band was detected for SCD-1 at approximately 38 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Western Blot Shows Human SCD‑1 Specificity by Using Knockout Cell Line.
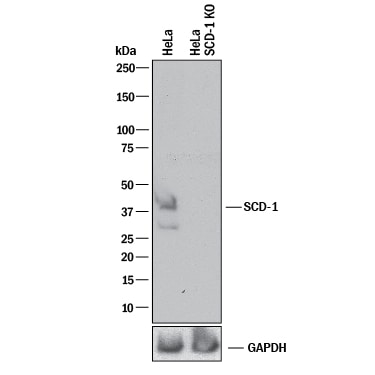
Western blot shows lysates of HeLa human cervical epithelial carcinoma parental cell line and SCD-1 knockout HeLa cell line (KO). PVDF membrane was probed with 0.5 µg/mL of Sheep Anti-Human SCD-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF7550) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (HAF016). A specific band was detected for SCD-1 at approximately 41 kDa (as indicated) in the parental HeLa cell line, but is not detectable in knockout HeLa cell line. GAPDH (AF5718) is shown as a loading control. This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Western Blot Shows Human SCD-1 Specificity Using Knockout Cell Line.
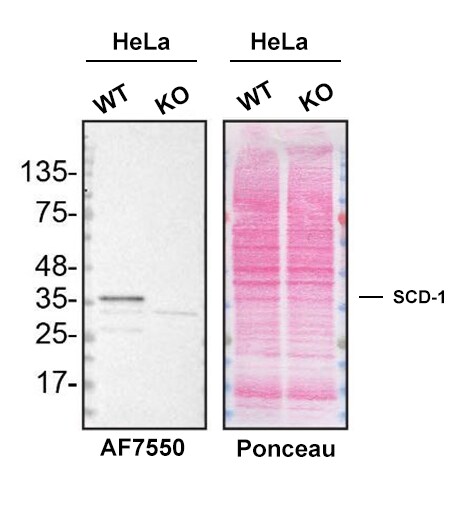
Western blot shows lysates of HeLa human cervical epithelial carcinoma parental cell line and SCD-1 knockout HeLa cell line (KO). Nitrocellulose membrane was probed with 1 µg/mL of Sheep Anti-Human SCD-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF7550) followed by HRP-conjugated anti-sheep IgG Secondary Antibody. A specific band was detected for SCD-1 at approximately 36 kDa (as indicated) in the parental HeLa cell line, but is not detectable in knockout HeLa cell line. The Ponceau stained transfer of the blot is shown. This experiment was conducted under reducing conditions. Image, protocol, and testing courtesy of YCharOS Inc. See ycharos.com for additional details.SCD-1 Specificity is Shown by Immunocytochemistry in Knockout Cell Line.
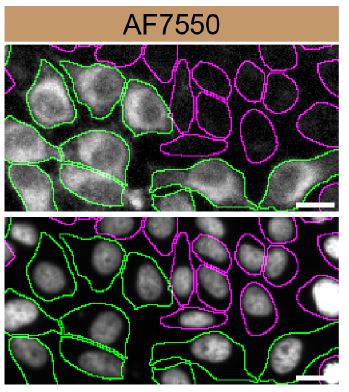
HeLa WT and SCD-1 KO cells were labelled with a green or a far-red fluorescent dye, respectively. Cells were stained with Sheep Anti-Human SCD-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF7550) followed by incubation with an anti-sheep Alexa-fluor 555 coupled secondary antibody (upper panel). DAPI-only counterstained cells shown on a lower panel. Acquisition of the blue (nucleus-DAPI), green (identification of WT cells), red (antibody staining) and far-red (identification of KO cells) channels was performed. Representative images of the blue and red (grayscale) channels are shown. WT and KO cells are outlined with green and magenta dashed line, respectively. Primary antibody concentration used: 1 µg/mL. Image, protocol and testing courtesy of YCharOS Inc. (ycharos.com).Detection of SCD-1 by Immunoprecipitation.
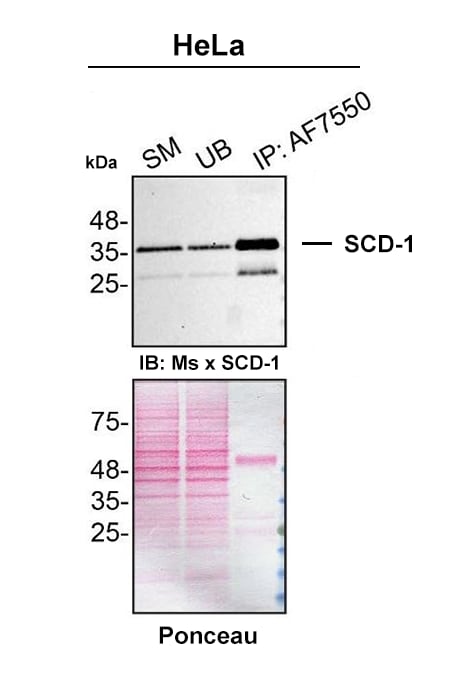
PMA-treated HeLa lysates were prepared and immunoprecipitation was performed using 2.0 µg of Sheep Anti-Human SCD-1 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF7550) pre-coupled to Dynabeads protein G. Immunoprecipitated SCD-1 was detected with Mouse Anti-SCD-1 Antibody.The Ponceau stained transfer of the blot is shown. SM=4% starting material; UB=4% unbound fraction; IP=immunoprecipitate; HC=antibody heavy chain. Image, protocol and testing courtesy of YCharOS Inc. (ycharos.com).Applications for Human SCD‑1 Antibody
Application
Recommended Usage
Immunoprecipitation
2 µg/1 mg cell lysate
Sample: Cell lysate of HeLa human cervical epithelial carcinoma cell line
Sample: Cell lysate of HeLa human cervical epithelial carcinoma cell line
Knockout Validated
SCD‑1
is specifically detected in HeLa human cervical epithelial carcinoma parental cell line but is not detectable in
SCD‑1 knockout HeLa cell line.
Western Blot
0.5 µg/mL
Sample: HepG2 human hepatocellular carcinoma cell line and SK‑Mel‑28 human malignant melanoma cell line
Sample: HepG2 human hepatocellular carcinoma cell line and SK‑Mel‑28 human malignant melanoma cell line
Reviewed Applications
Read 1 review rated 5 using AF7550 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: SCD-1
Long Name
Stearoyl CoA-desaturase 1, Isoform of the Delta9 Desaturase
Alternate Names
SCD1
Gene Symbol
SCD
UniProt
Additional SCD-1 Products
Product Documents for Human SCD‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human SCD‑1 Antibody
For research use only
Related Research Areas
Citations for Human SCD‑1 Antibody
Customer Reviews for Human SCD‑1 Antibody (1)
5 out of 5
1 Customer Rating
Have you used Human SCD‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Simple WesternSample Tested: Human mesenchymal stem cell whole cell lysateSpecies: HumanVerified Customer | Posted 06/24/20150.2 ug/mL hMSC lysate run with 10 ug/mL SCD-1 antibody (A7550)
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- Immunoprecipitation Protocol
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...