Signal regulatory protein gamma (SIRP gamma, designated CD172g), also called SIRP beta 2, is a monomeric 45-47 kDa type I transmembrane glycoprotein belonging to the SIRP/SHPS (CD172) family of the Ig superfamily (1 ‑ 5). SIRP members are “paired receptors” with homology in the extracellular domain but variability in the C‑terminus and signaling function (1, 2). The 387 amino acid (aa) SIRP gamma sequence contains a 28 aa potential signal sequence, a 332 aa extracellular domain (ECD) with four potential N‑glycosylation sites, a 23 aa transmembrane domain and a 4 aa cytoplasmic sequence. SIRP gamma contains one V-type Ig‑like domain that contains a J‑like sequence and two C1-type Ig‑like domains within its ECD (1, 2). Isoforms that lack one (isoform 2, 276 aa) or two (isoform 3, 170 aa) membrane-proximal C‑type Ig-like domains have been described (5). Within the ECD, human SIRP gamma isoform 1 shares 78% aa identity with human SIRP beta 1, and appears to have structurally similar orthologs only in rhesus monkey and chimpanzee (100% and 91% aa identity, respectively) (2). SIRP gamma is the only SIRP known to be expressed on T cells, CD56bright NK cells and activated NK cells; it is not expressed on myeloid cells (5, 6). It shows adhesion to CD47, but at lower affinity than SIRP alpha (6). Expression of SIRP gamma on T cells suggests a role as an accessory protein interacting with CD47‑expressing antigen presenting cells (5, 6). Unlike SIRP alpha that has cytoplasmic ITIM domains, and SIRP beta 1 that interacts with DAP-12, SIRP gamma does not contain any obvious signaling mechanism (1, 2, 6). However, SIRP gamma -mediated adhesion appears to promote antigen-specific T cell proliferation and costimulate T cell activation (5).

Key Product Details
Species Reactivity
Applications
Label
Antibody Source
Product Specifications
Immunogen
Val64-Ser364, (Val263Ala) and (Ser286Leu)
Accession # Q9P1W8
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human SIRP gamma /CD172g Antibody
Detection of Human SIRP gamma /CD172g by Western Blot.
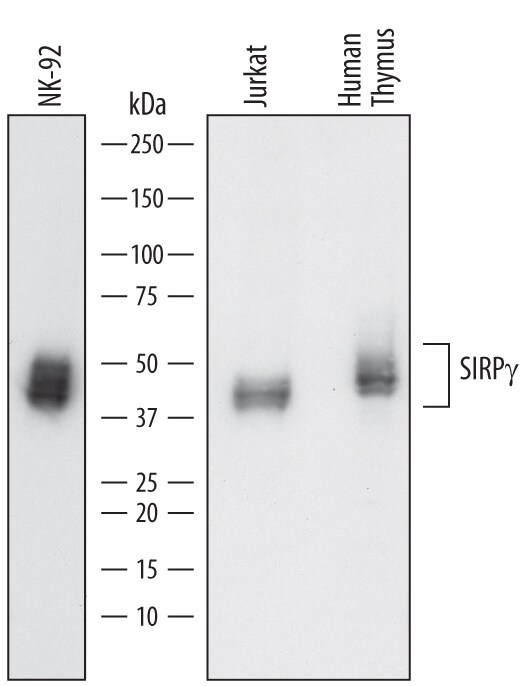
Western blot shows lysates of NK-92 human natural killer lymphoma cell line, Jurkat human acute T cell leukemia cell line, and human thymus tissue. PVDF membrane was probed with 0.5 µg/mL of Sheep Anti-Human SIRP gamma/CD172g Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4486) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (HAF016). Specific bands were detected for SIRP gamma/CD172g at approximately 45-50 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of SIRP gamma /CD172g in Human Blood Monocytes by Flow Cytometry.
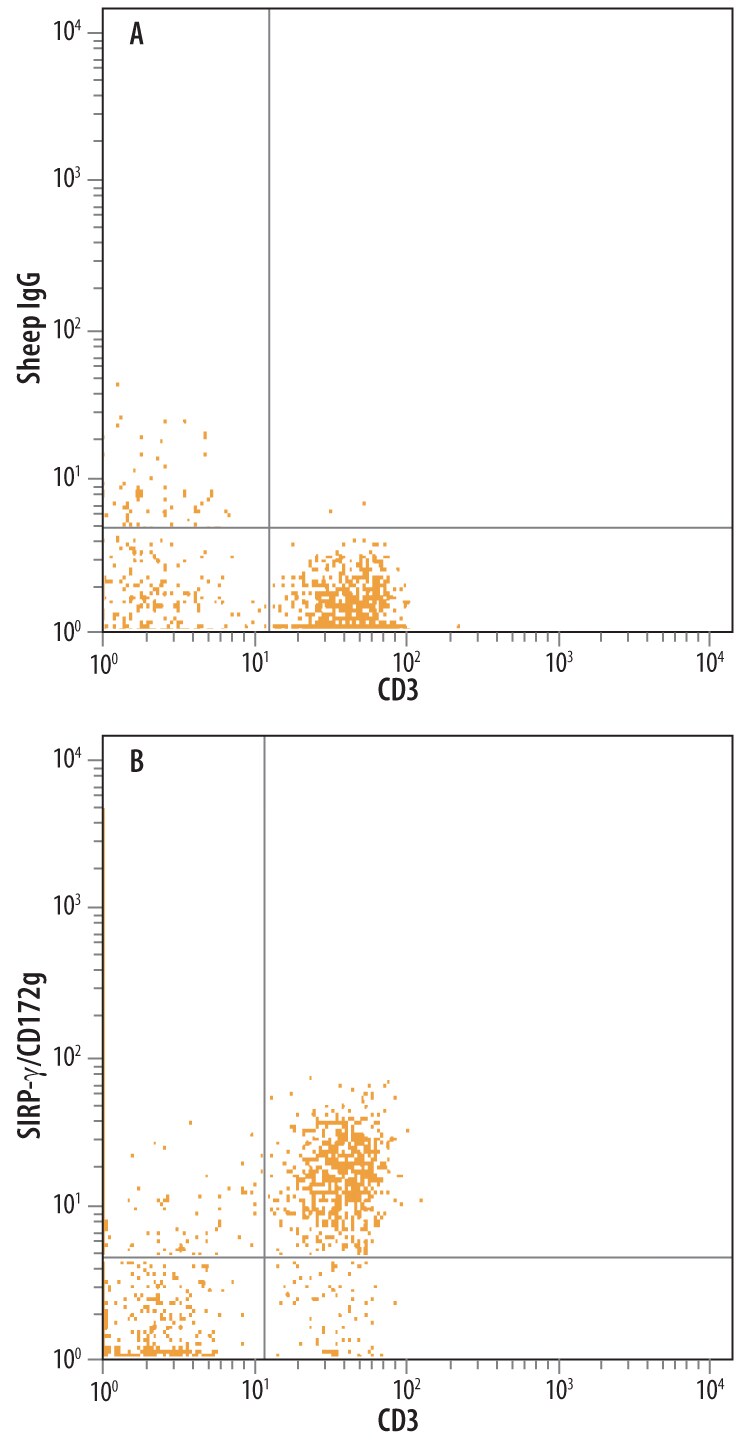
Human peripheral blood monocytes were stained with Sheep Anti-Human SIRP gamma/CD172g Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4486) followed by Phycoerythrin-conjugated Anti-Sheep IgG Secondary Antibody (F0126) and Mouse Anti-Human CD3e APC-conjugated Monoclonal Antibody (FAB100A). Quadrant markers were set based on control antibody staining (Catalog # 5-001-A).Applications for Human SIRP gamma /CD172g Antibody
CyTOF-ready
Flow Cytometry
Sample: Human peripheral blood monocytes
Western Blot
Sample: NK‑92 human natural killer lymphoma cell line, Jurkat human acute T cell leukemia cell line, and human thymus tissue
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Sterile PBS to a final concentration of 0.2 mg/mL. For liquid material, refer to CoA for concentration.
Formulation
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: SIRP gamma/CD172g
References
- Barclay, A.N. & M.H. Brown (2006) Nat. Rev. Immunol. 6:457.
- vanBeek, E.M. et al. (2005) J. Immunol. 175:7781.
- van den Berg, T.K. et al. (2005) J. Immunol. 175:7788.
- Ichigotani, Y. et al. (2000) J. Hum. Genet. 45:378.
- Piccio, L. et al. (2005) Blood 105:2421.
- Brooke, G. et al. (2004) J. Immunol. 173:2562.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional SIRP gamma/CD172g Products
Product Documents for Human SIRP gamma /CD172g Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human SIRP gamma /CD172g Antibody
For research use only
Related Research Areas
Customer Reviews for Human SIRP gamma /CD172g Antibody
There are currently no reviews for this product. Be the first to review Human SIRP gamma /CD172g Antibody and earn rewards!
Have you used Human SIRP gamma /CD172g Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Cellular Response to Hypoxia Protocols
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars