Vesicle-associated membrane protein (VAMP)-associated protein B (VAP-B; also VAMP-B) is an ~30 kDa ubiquitously expressed type IV transmembrane protein belonging to the VAP family (1, 2). It is found in endoplasmic reticulum (ER), Golgi and other membranes as a homodimer or a heterodimer with VAP-A, probably associating through a GxxxG motif in the transmembrane regions (1, 2). Human VAP-B cDNA encodes 243 amino acids (aa) that include a 222 aa cytoplasmic domain and a 21 aa C-terminal membrane anchor. The cytoplasmic domain contains a mobile sperm protein (MSP) domain (aa 7‑124) and a coiled-coil region (aa 159‑196). Human VAP-B shares 90%, 89%, 96%, 96% and 94% aa identity with mouse, rat, canine, bovine and porcine VAP-B, respectively. VAP-A and VAP-B MSP domains recruit FFAT (two phenylalanines in an acidic tract)-motif-containing proteins to the cytosolic surface of ER membranes (2‑4). FFAT proteins mediate many of the effects of VAPs on regulation of membrane transport, phospholipid biosynthesis, microtubule organization, and the unfolded protein response (2, 3). VAPs also interact with some SNARE and viral proteins (2). A human polymorphism of VAP-B, P56S, is found in three familial motor neuron diseases, notably the amylotrophic lateral sclerosis variant ALS8 (2). It produces a non-functional protein that can dimerize with and inhibit function of normal VAP-B, leading formation of intracellular aggregates and increased ER-stress-induced death of motor neurons (5‑7). It can also promote cleavage and secretion of soluble VAP-B, which can then function as a ligand for EPH receptors (8). A naturally occurring 99 aa isoform of VAP-B that diverges at aa 71 within the MSP domain is termed VAP-C (1, 9). It also appears to be a negative regulator of VAP-A and VAP-B (9). While VAP-B is used by hepatitis C virus (HCV) for its propagation, VAP-C inhibits HCV propagation (9).


Key Product Details
Species Reactivity
Human
Applications
Immunohistochemistry, Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Sheep IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human VAP-B
Ala2-Pro132
Accession # O95292
Ala2-Pro132
Accession # O95292
Specificity
Detects human VAP-B in direct ELISAs and Western blots. In direct ELISAs, approximately 60% cross-reactivity with recombinant rat VAP-B is observed and less than 5% cross-reactivity with recombinant human VAP-A is observed.
Clonality
Polyclonal
Host
Sheep
Isotype
IgG
Scientific Data Images for Human VAP‑B Antibody
Detection of Human VAP-B by Western Blot.
Western blot shows lysates of human skeletal muscle tissue. PVDF membrane was probed with 1 µg/mL of Sheep Anti-Human VAP-B Antigen Affinity-purified Polyclonal Antibody (Catalog # AF5855) followed by HRP-conjugated Anti-Sheep IgG Secondary Antibody (Catalog # HAF016). A specific band was detected for VAP-B at approximately 30 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 8.VAP‑B in Human Brain.
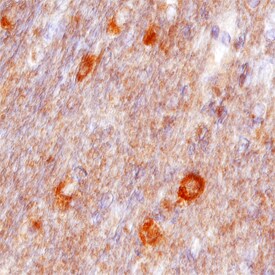
VAP-B was detected in immersion fixed paraffin-embedded sections of human brain (cerebellum) using Sheep Anti-Human VAP-B Antigen Affinity-purified Polyclonal Antibody (Catalog # AF5855) at 10 µg/mL overnight at 4 °C. Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013). Tissue was stained using the Anti-Sheep HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS019) and counter-stained with hematoxylin (blue). Specific staining was localized to neuronal cell bodies. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Applications for Human VAP‑B Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human brain (cerebellum) subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013)
Sample: Immersion fixed paraffin-embedded sections of human brain (cerebellum) subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (Catalog # CTS013)
Western Blot
1 µg/mL
Sample: Human skeletal muscle tissue
Sample: Human skeletal muscle tissue
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: VAP-B
References
- Nishimura, Y. et al. (1999) Biochem. Biophys. Res. Commun. 254:21.
- Lev, S. et al. (2008) Trends Cell Biol. 18:282.
- Peretti, D. et al. (2008) Mol. Biol. Cell 19:3871.
- Kaiser, S.E. et al. (2005) Structure 13:1035.
- Prosser, D.C. et al. (2008) J. Cell Sci. 121:3052.
- Gkogkas, C. et al. (2008) Hum. Mol. Genet. 17:1517.
- Suzuki, H. et al. (2009) J. Neurochem. 108:973.
- Tsuda, H. et al. (2008) Cell 133:963.
- Kukihara, H. et al. (2009) J. Virol. 83:7959.
Long Name
VAMP [Vesicle-associated Membrane Protein]-associated Protein B and C
Alternate Names
ALS8, VAMP-B, VAMP-C, VAP-C, VAPB
Gene Symbol
VAPB
UniProt
Additional VAP-B Products
Product Documents for Human VAP‑B Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human VAP‑B Antibody
For research use only
Customer Reviews for Human VAP‑B Antibody
There are currently no reviews for this product. Be the first to review Human VAP‑B Antibody and earn rewards!
Have you used Human VAP‑B Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...